Abstract
Purpose
Patients who undergo surgery for lung cancer experience a variety of symptoms, such as pain and coughing, which interfere with their postoperative daily functions. However, there may be differences between the perception of symptoms among healthcare providers and patients. This study aimed to investigate patients’ experiences after lung cancer surgery and analyze whether the perception of postoperative symptoms among the healthcare providers differed from that reported by patients.
Methods
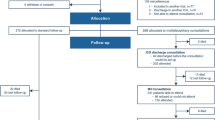
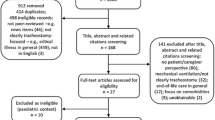
Semi-structured qualitative interviews involving 39 patients who underwent lung cancer surgery at the Sichuan Cancer Hospital were conducted between November 2018 and October 2019. In addition, 22 healthcare providers from the Department of Thoracic Surgery at the hospital answered open-ended questions about their perception of symptoms related to lung cancer surgery. The types and frequencies of symptoms reported by the patients and healthcare providers were compared.
Results
The most frequent patient-reported symptoms were pain (967 times, 39 patients, 100%), coughing (904 times, 37 patients, 94.87%), shortness of breath (491 times, 35 patients, 89.74%), Disturbed sleep (412 times, 34 patients, 87.18%), and interference while walking (347 times, 36 patients, 92.31%). Of the patient-reported symptoms, the first four were perceived by the healthcare providers, while they interpreted interference while walking as fatigue.
Conclusion
Although the healthcare providers and patients had a certain consensus on the main symptoms, there were differences in perception. Healthcare providers need to pay more attention to postoperative interference while walking.

Similar content being viewed by others
Data availability
The authors are willing to share data, analytic methods, and study materials related to this article with other researchers, provided that they will not be used for commercial or profit purposes. The corresponding author of this article can be contacted by email regarding the research materials needed while stating the reason for their use. The authors will be glad to provide relevant materials after approval and discussion.
Code availability
Inter-rater reliability tests and qualitative data were analyzed using NVivo 12 (QSR International. 2020. http://www.qsrinternational.com).
References
Chen W, Zheng R, Baade PD et al (2016) Cancer statistics in China, 2015. CA Cancer J Clin 66:115–132. https://doi.org/10.3322/caac.21338
National Health Commission of the People’s Republic of China (2019) Chinese guidelines for diagnosis and treatment of primary lung cancer 2018 (English version). Chin J Cancer Res 31:1–28
Kim BJ, Caudle AS, Gottumukkala V, Aloia TA (2016) The impact of postoperative complications on a timely return to intended oncologic therapy (RIOT): the role of enhanced recovery in the cancer journey. Int Anesthesiol Clin 54:e33–e46. https://doi.org/10.1097/AIA.0000000000000113
Fernandez FG, Kosinski AS, Furnary AP et al (2018) Differential effects of operative complications on survival after surgery for primary lung cancer. J Thorac Cardiovasc Surg 155:1254-1264.e1. https://doi.org/10.1016/j.jtcvs.2017.09.149
Barbera L, Atzema C, Sutradhar R et al (2013) Do patient-reported symptoms predict emergency department visits in cancer patients? A population-based analysis. Annal Emer Med 61:427-437.e5. https://doi.org/10.1016/j.annemergmed.2012.10.010
Efficace F, Cartoni C, Niscola P et al (2012) Predicting survival in advanced hematologic malignancies: do patient-reported symptoms matter? Eur J Haematol 89:410–416. https://doi.org/10.1111/ejh.12004
Neville A, Lee L, Antonescu I et al (2014) Systematic review of outcomes used to evaluate enhanced recovery after surgery. Br J Surg 101:159–170. https://doi.org/10.1002/bjs.9324
Maessen JM, Dejong CH, Kessels AG, von Meyenfeldt MF; Enhanced Recovery After Surgery Group (2008) Length of stay: an inappropriate readout of the success of enhanced recovery programs. World J Surg 32:971–975.https://doi.org/10.1007/s00268-007-9404-9
Bruera E, Sweeney C, Calder K, Palmer L, Benisch-Tolley S (2001) Patient preferences versus physician perceptions of treatment decisions in cancer care. J Clin Oncol 19:2883–2885. https://doi.org/10.1200/JCO.2001.19.11.2883
Butow PN, Brown RF, Cogar S, Tattersall MH, Dunn SM (2002) Oncologists’ reactions to cancer patients’ verbal cues. Psychooncology 11:47–58. https://doi.org/10.1002/pon.556
Chang VT, Hwang SS, Feuerman M, Kasimis BS (2000) Symptom and quality of life survey of medical oncology patients at a veterans affairs medical center: a role for symptom assessment. Cancer 88:1175–1183. https://doi.org/10.1002/(sici)1097-0142(20000301)88:5%3c1175::aid-cncr30%3e3.0.co;2-n
Newell S, Sanson-Fisher RW, Girgis A, Bonaventura A (1998) How well do medical oncologists’ perceptions reflect their patients’ reported physical and psychosocial problems? Data from a survey of five oncologists. Cancer 83:1640–1651
Farrell C, Beaver K, Heaven C, Maguire P (2001) Identifying the concerns of women undergoing chemotherapy treatment for cancer. Eur J Cancer 37:S390. https://doi.org/10.1016/S0959-8049(01)81894-3
McIntyre P. Side effects of targeted treatments: clinicians’ perceptions, patients’ realities. Cancer World. https://bit.ly/2Hz0JpA. Accessed 1 March 2015
Basch E, Iasonos A, McDonough T et al (2006) Patient versus clinician symptom reporting using the National Cancer Institute Common Terminology Criteria for Adverse Events: results of a questionnaire-based study. Lancet Oncol 7:903–909. https://doi.org/10.1016/S1470-2045(06)70910-X
Petersen MA, Larsen H, Pedersen L, Sonne N, Groenvold M (2006) Assessing health-related quality of life in palliative care: comparing patient and physician assessments. Eur J Cancer 42:1159–1166. https://doi.org/10.1016/j.ejca.2006.01.032
Basch E (2017) Patient-reported outcomes - harnessing patients’ voices to improve clinical care. N Engl J Med 376:105–108. https://doi.org/10.1056/NEJMp16112
US FDA (2009) Guidance for industry patient-report outcome measures: use in medical product development to support labeling claims. In: US Department of health and Human Services (ed) Rockville MD. https://www.fda.gov/downloads/drugs/guidances/ucm193282.pdf. Accessed 01 Oct 2020
US FDA (2015) Clinical outcome assessment (COA): glossary of terms. https://www.fda.gov/Drugs/DevelopmentApprovalProcess/DrugDevelopmentToolsQualificationProgram/ucm284077.htm. Accessed 01 Oct 2020
Calvert M, Kyte D, Mercieca-Bebber R et al (2018) Guidelines for inclusion of patient-reported outcomes in clinical trial protocols: the SPIRIT-PRO extension. JAMA 319:483–494. https://doi.org/10.1001/jama.2017.21903
Shi Q, Mendoza TR, Wang XS, Cleeland CS (2016) Using a symptom-specific instrument to measure patient-reported daily functioning in patients with cancer. Eur J Cancer 67:83–90. https://doi.org/10.1016/j.ejca.2016.07.027
Basch E, Deal AM, Dueck AC et al (2017) Overall survival results of a trial assessing patient-reported outcomes for symptom monitoring during routine cancer treatment. JAMA 318:197–198. https://doi.org/10.1001/jama.2017.7156
Glaser BG, Strauss AL (1967) Discovery of grounded theory: strategies for qualitative research. Routledge, New York.https://doi.org/10.4324/9780203793206
Dhillon HM, Bell ML, van der Ploeg HP et al (2017) Impact of physical activity on fatigue and quality of life in people with advanced lung cancer: a randomized controlled trial. Ann Oncol 28:1889–1897. https://doi.org/10.1093/annonc/mdx205
Park J, Neuman HB, Bennett AV et al (2014) Patient expectations of functional outcomes after rectal cancer surgery: a qualitative study. Dis Colon Rectum 57:151–157. https://doi.org/10.1097/DCR.0000000000000036
Shi Q, Wang XS, Vaporciyan AA et al (2016) Patient-reported symptom interference as a measure of postsurgery functional recovery in lung cancer. J Pain Symptom Manage 52:822–831. https://doi.org/10.1016/j.jpainsymman.2016.07.005
Bendixen M, Jørgensen OD, Kronborg C, Andersen C, Licht PB (2016) Postoperative pain and quality of life after lobectomy via video-assisted thoracoscopic surgery or anterolateral thoracotomy for early stage lung cancer: a randomised controlled trial. Lancet Oncol 17:836–844. https://doi.org/10.1016/S1470-2045(16)00173-X
Wei X, Yu H, Dai W, Mu Y, Wang Y, Liao J, Peng L, Han Y, Li Q, Shi Q (2021) Patient-reported outcomes of video-assisted thoracoscopic surgery versus thoracotomy for locally advanced lung cancer: a longitudinal cohort study. Ann Surg Oncol. https://doi.org/10.1245/s10434-021-09981-1
Van der Ploeg APT, Ayez N, Akkersdijk GP, van Rossem CC, de Rooij PD (2020) Postoperative pain after lobectomy: robot-assisted, video-assisted and open thoracic surgery. J Robot Surg 14:131–136. https://doi.org/10.1007/s11701-019-00953-y
Apfelbaum JL, Chen C, Mehta SS, Gan TJ (2003) Postoperative pain experience: results from a national survey suggest postoperative pain continues to be undermanaged. Anesth Analg 97:534–540, table of contents. https://doi.org/10.1213/01.ane.0000068822.10113.9e
Lin R, Che G (2018) Risk factors of cough in non-small cell lung cancer patients after video-assisted thoracoscopic surgery. J Thorac Dis 10:5368–5375. https://doi.org/10.21037/jtd.2018.08.54
Sawada S, Suehisa H, Yamashita M (2012) Inhalation of corticosteroid and β-agonist for persistent cough following pulmonary resection. Gen Thorac Cardiovasc Surg 60:285–288. https://doi.org/10.1007/s11748-011-0910-1
Kim KI, Shin S, Kim K, Lee J (2016) Efficacy and safety of Maekmoondong-tang for chronic dry cough: a study protocol for a randomized controlled trial. BMC Complement Altern Med 16:46. https://doi.org/10.1186/s12906-016-1028-x
Wahls SA (2012) Causes and evaluation of chronic dyspnea. Am Fam Physician 86:173–182
Fagundes CP, Shi Q, Vaporciyan AA et al (2015) Symptom recovery after thoracic surgery: measuring patient-reported outcomes with the MD Anderson Symptom Inventory. J Thorac Cardiovasc Surg 150(613–619):e612. https://doi.org/10.1016/j.jtcvs.2015.05.057
Basch E, Deal AM, Kris MG et al (2016) Symptom monitoring with patient-reported outcomes during routine cancer treatment: a randomized controlled trial. J Clin Oncol 34:557–565. https://doi.org/10.1200/JCO.2015.63.0830
Basch E, Jia X, Heller G et al (2009) Adverse symptom event reporting by patients vs clinicians: relationships with clinical outcomes. J Natl Cancer Inst 101:1624–1632. https://doi.org/10.1093/jnci/djp386
Calvert M, Kyte D, Price G, Valderas JM, Hjollund NH (2019) Maximising the impact of patient reported outcome assessment for patients and society. BMJ 364:k5267. https://doi.org/10.1136/bmj.k5267
U.S. Department of Health and Human Services FDA Center for Drug Evaluation and Research, U.S. Department of Health and Human Services FDA Center for Biologics Evaluation and Research, U.S. Department of Health and Human Services FDA Center for Devices and Radiological Health (2006) Guidance for industry: patient-reported outcome measures: use in medical product development to support labeling claims: draft guidance. Health Qual Life Outcomes 4:79. https://doi.org/10.1186/1477-7525-4-79
Acknowledgements
We would like to thank Wenhan Li from the Department of Thoracic Surgery, Sichuan Cancer Hospital & Institute, Sichuan Cancer Center, School of Medicine, University of Electronic Science and Technology of China, for her contributions in collecting data for this study.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81872506).
Author information
Authors and Affiliations
Contributions
Study conception and designing: all authors.
Acquisition, analysis, or interpretation of data: all authors.
Drafting of the abstract: Xing Wei.
Critical revision of the article for important intellectual content: all authors.
Final approval of the version to be published: all authors.
Statistical analysis: HongfanYu.
Funding acquisition: Qiuling Shi.
Administrative, technical, or material support: Qiang Li and Qiuling Shi.
Study supervision: Qiang Li and Qiuling Shi.
Xing Wei and Hongfan Yu had full access to all the study data and take responsibility for the integrity of the data and accuracy of the data analysis.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Ethics Committee of Sichuan Cancer Hospital (approval number: SCCHEC-02–2018-043).
Consent to participate
Written informed consent was obtained from individual participants.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Disclaimer
The authors have full control of all primary data and agree to allow the journal to review the data if requested.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix 1: Lending questions for qualitative semi-structured interviews
Appendix 1: Lending questions for qualitative semi-structured interviews
-
1.
Please recall your symptoms from the day of the operation to today.
If the patient is unable to describe in detail, use the following guide questions:
-
A.
What are the symptoms you experienced after surgery?
-
B.
Do these symptoms occur together or in sequence?
-
C.
Do XX symptoms affect your actions?
-
D.
When did XX’s symptoms subside?
-
E.
Which symptom do you think is the most serious? Why is that?
-
F.
What kind of activity limitation after surgery makes you feel most uncomfortable, such as walking, going to the bathroom, and washing?
-
2.
Do you think your feelings during the operation and hospitalization are fully described?
-
3.
Do you have any other feelings that you wish to tell us?
Rights and permissions
About this article
Cite this article
Wei, X., Yu, H., Dai, W. et al. Discrepancy in the perception of symptoms among patients and healthcare providers after lung cancer surgery. Support Care Cancer 30, 1169–1179 (2022). https://doi.org/10.1007/s00520-021-06506-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-021-06506-0