Abstract
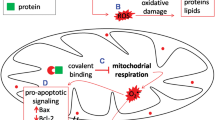
Drug-induced liver injury (DILI) caused by the ingestion of medications, herbs, chemicals or dietary supplements, is a clinically widespread health problem. The underlying mechanism of DILI is the formation of reactive metabolites, which trigger mitochondrial oxidative stress and the opening of mitochondrial permeability transition (MPT) pores through direct toxicity or immune response, leading to cell inflammation, apoptosis, and necrosis. Traditionally, mitochondria play an indispensable role in maintaining the physiological and biochemical functions of cells by producing ATP and mediating intracellular signal transduction; drugs can typically stimulate the mitochondria and, in the case of sustained stress, can eventually cause impairment of mitochondrial function and metabolic activity. Meanwhile, the mitochondrial stress response, as an adaptive protective mechanism, occurs when mitochondrial homeostasis is threatened. In this review, we summarize the relevant frontier researches of the protective effects of mitochondrial stress response in DILI as well as the potential related mechanisms, thus providing some thoughts for the clinical treatment of DILI.


Similar content being viewed by others
References
Vakifahmetoglu-Norberg H, Ouchida AT, Norberg E (2017) The role of mitochondria in metabolism and cell death. Biochem Biophys Res Commun 482:426–431. https://doi.org/10.1016/j.bbrc.2016.11.088
Mittler R (2017) ROS are good. Trends Plant Sci 22:11–19. https://doi.org/10.1016/j.tplants.2016.08.002
Garcia-Cortes M, Robles-Diaz M, Stephens C, Ortega-Alonso A, Lucena MI, Andrade RJ (2020) Drug induced liver injury: an update. Arch Toxicol 94:3381–3407. https://doi.org/10.1007/s00204-020-02885-1
Aithal GP (2019) Drug-induced liver injury. Medicine 47:734–739. https://doi.org/10.1016/j.mpmed.2019.08.005
Teschke R (2018) Top-ranking drugs out of 3312 drug-induced liver injury cases evaluated by the Roussel Uclaf causality assessment method. Expert Opin Drug Metab Toxicol 14:1169–1187. https://doi.org/10.1080/17425255.2018.1539077
Han D, Dara L, Win S, Than TA, Yuan L, Abbasi SQ, Liu ZX, Kaplowitz N (2013) Regulation of drug-induced liver injury by signal transduction pathways: critical role of mitochondria. Trends Pharmacol Sci 34:243–253. https://doi.org/10.1016/j.tips.2013.01.009
Jaeschke H (2018) Mitochondrial dysfunction as a mechanism of drug-induced hepatotoxicity: current understanding and future perspectives. J Clin Transl Res. https://doi.org/10.18053/jctres.04.201801.005
Duan L, Woolbright BL, Jaeschke H, Ramachandran A (2020) Late Protective effect of netrin-1 in the murine acetaminophen hepatotoxicity model. Toxicol Sci 175:168–181. https://doi.org/10.1093/toxsci/kfaa041
Jaeschke H, Akakpo JY, Umbaugh DS, Ramachandran A (2020) Novel therapeutic approaches against acetaminophen-induced liver injury and acute liver failure. Toxicol Sci 174:159–167. https://doi.org/10.1093/toxsci/kfaa002
Wang J, Li M, Zhang W, Gu A, Dong J, Li J, Shan A (2018) Protective effect of N-acetylcysteine against oxidative stress induced by zearalenone via mitochondrial apoptosis pathway in SIEC02 cells. Toxins (Basel) 10:1–17. https://doi.org/10.3390/toxins10100407
Li F, Qiu Y, Xia F, Sun H, Liao H, Xie A, Lee J, Lin P, Wei M, Shao Y et al (2020) Dual detoxification and inflammatory regulation by ceria nanozymes for drug-induced liver injury therapy. Nano Today. https://doi.org/10.1016/j.nantod.2020.100925
Wang SF, Chen S, Tseng LM, Lee HC (2020) Role of the mitochondrial stress response in human cancer progression. Exp Biol Med 245:861–878. https://doi.org/10.1177/1535370220920558
Xue F, Hua Z (2017) Signaling pathway of mitochondrial stress. Front Lab Med 1:40–42. https://doi.org/10.1016/j.flm.2017.02.009
D’Amico D, Sorrentino V, Auwerx J (2017) Cytosolic proteostasis networks of the mitochondrial stress response. Trends Biochem Sci 42:712–725. https://doi.org/10.1016/j.tibs.2017.05.002
Hill S, Van Remmen H (2014) Mitochondrial stress signaling in longevity: a new role for mitochondrial function in aging. Redox Biol 2:936–944. https://doi.org/10.1016/j.redox.2014.07.005
Lin S, Xing H, Zang T, Ruan X, Wo L, He M (2018) Sirtuins in mitochondrial stress: indispensable helpers behind the scenes. Ageing Res Rev 44:22–32. https://doi.org/10.1016/j.arr.2018.03.006
Seli E, Wang T, Horvath TL (2019) Mitochondrial unfolded protein response: a stress response with implications for fertility and reproductive aging. Fertil Steril 111:197–204. https://doi.org/10.1016/j.fertnstert.2018.11.048
Hill S, Sataranatarajan K, Van Remmen H (2018) Role of signaling molecules in mitochondrial stress response. Front Genet 9:225. https://doi.org/10.3389/fgene.2018.00225
Anderson NS, Haynes CM (2020) Folding the mitochondrial UPR into the integrated stress response. Trends Cell Biol 30:428–439. https://doi.org/10.1016/j.tcb.2020.03.001
Fessler E, Eckl EM, Schmitt S, Mancilla IA, Meyer-Bender MF, Hanf M, Philippou-Massier J, Krebs S, Zischka H, Jae LT (2020) A pathway coordinated by DELE1 relays mitochondrial stress to the cytosol. Nature 579:433–437. https://doi.org/10.1038/s41586-020-2076-4
Quiros PM, Prado MA, Zamboni N, D’Amico D, Williams RW, Finley D, Gygi SP, Auwerx J (2017) Multi-omics analysis identifies ATF4 as a key regulator of the mitochondrial stress response in mammals. J Cell Biol 216:2027–2045. https://doi.org/10.1083/jcb.201702058
Tauber D, Parker R (2019) 15-Deoxy-Delta(12,14)-prostaglandin J2 promotes phosphorylation of eukaryotic initiation factor 2alpha and activates the integrated stress response. J Biol Chem 294:6344–6352. https://doi.org/10.1074/jbc.RA118.007138
Topf U, Wrobel L, Chacinska A (2016) Chatty mitochondria: keeping balance in cellular protein homeostasis. Trends Cell Biol 26:577–586. https://doi.org/10.1016/j.tcb.2016.03.002
Callegari S, Dennerlein S (2018) Sensing the stress: a role for the UPR(mt) and UPR(am) in the quality control of mitochondria. Front Cell Dev Biol 6:31. https://doi.org/10.3389/fcell.2018.00031
Coyne LP, Chen XJ (2018) mPOS is a novel mitochondrial trigger of cell death—implications for neurodegeneration. FEBS Lett 592:759–775. https://doi.org/10.1002/1873-3468.12894
Kim HE, Grant AR, Simic MS, Kohnz RA, Nomura DK, Durieux J, Riera CE, Sanchez M, Kapernick E, Wolff S et al (2016) Lipid biosynthesis coordinates a mitochondrial-to-cytosolic stress response. Cell 166:1539-1552e1516. https://doi.org/10.1016/j.cell.2016.08.027
Abeliovich H, Dengjel J (2016) Mitophagy as a stress response in mammalian cells and in respiring S. cerevisiae. Biochem Soc Trans 44:541–545. https://doi.org/10.1042/BST20150278
Eisner V, Picard M, Hajnoczky G (2018) Mitochondrial dynamics in adaptive and maladaptive cellular stress responses. Nat Cell Biol 20:755–765. https://doi.org/10.1038/s41556-018-0133-0
Girerd S, Tosca L, Herault O, Vignon C, Biard D, Aggoune D, Dkhissi F, Bonnet ML, Sorel N, Desterke C et al (2018) Superoxide dismutase 2 (SOD2) contributes to genetic stability of native and T315I-mutated BCR-ABL expressing leukemic cells. Biochem Biophys Res Commun 498:715–722. https://doi.org/10.1016/j.bbrc.2018.03.023
Felson DT (2016) Safety of nonsteroidal antiinflammatory drugs. N Engl J Med 375:2595–2596. https://doi.org/10.1056/NEJMe1614257
Walters KM, Woessner KM (2016) An overview of nonsteroidal antiinflammatory drug reactions. Immunol Allergy Clin North Am 36:625–641. https://doi.org/10.1016/j.iac.2016.06.001
Lee WM (2017) Acetaminophen (APAP) hepatotoxicity—isn’t it time for APAP to go away? J Hepatol 67:1324–1331. https://doi.org/10.1016/j.jhep.2017.07.005
Hamilton LA, Collins-Yoder A, Collins RE (2016) Drug-induced liver injury. AACN Adv Crit Care 27:430–440. https://doi.org/10.4037/aacnacc2016953
McGill MR, Jaeschke H (2013) Metabolism and disposition of acetaminophen: recent advances in relation to hepatotoxicity and diagnosis. Pharm Res 30:2174–2187. https://doi.org/10.1007/s11095-013-1007-6
Chao X, Wang H, Jaeschke H, Ding WX (2018) Role and mechanisms of autophagy in acetaminophen-induced liver injury. Liver Int 38:1363–1374. https://doi.org/10.1111/liv.13866
Ramachandran A, Jaeschke H (2019) Acetaminophen hepatotoxicity: a mitochondrial perspective. Adv Pharmacol 85:195–219. https://doi.org/10.1016/bs.apha.2019.01.007
Iorga A, Dara L, Kaplowitz N (2017) Drug-induced liver injury: cascade of events leading to cell death, apoptosis or necrosis. Int J Mol Sci 18:1–25. https://doi.org/10.3390/ijms18051018
Jetten MJ, Ruiz-Aracama A, Coonen ML, Claessen SM, van Herwijnen MH, Lommen A, van Delft JH, Peijnenburg AA, Kleinjans JC (2016) Interindividual variation in gene expression responses and metabolite formation in acetaminophen-exposed primary human hepatocytes. Arch Toxicol 90:1103–1115. https://doi.org/10.1007/s00204-015-1545-2
Attanasio S, Gernoux G, Ferriero R, De Cegli R, Carissimo A, Nusco E, Campione S, Teckman J, Mueller C, Piccolo P et al (2020) CHOP-c-JUN complex plays a critical role in liver proteotoxicity induced by mutant Z alpha1 antitrypsin. bioRxiv. https://doi.org/10.1101/2020.05.04.076752
Melber A, Haynes CM (2018) UPR(mt) regulation and output: a stress response mediated by mitochondrial-nuclear communication. Cell Res 28:281–295. https://doi.org/10.1038/cr.2018.16
Shan S, Shen Z, Song F (2018) Autophagy and acetaminophen-induced hepatotoxicity. Arch Toxicol 92:2153–2161. https://doi.org/10.1007/s00204-018-2237-5
Williams JA, Ding WX (2020) Role of autophagy in alcohol and drug-induced liver injury. Food Chem Toxicol 136:111075. https://doi.org/10.1016/j.fct.2019.111075
Baulies A, Ribas V, Nunez S, Torres S, Alarcon-Vila C, Martinez L, Suda J, Ybanez MD, Kaplowitz N, Garcia-Ruiz C et al (2015) Lysosomal cholesterol accumulation sensitizes to acetaminophen hepatotoxicity by impairing mitophagy. Sci Rep 5:18017. https://doi.org/10.1038/srep18017
Ni HM, McGill MR, Chao X, Du K, Williams JA, Xie Y, Jaeschke H, Ding WX (2016) Removal of acetaminophen protein adducts by autophagy protects against acetaminophen-induced liver injury in mice. J Hepatol 65:354–362. https://doi.org/10.1016/j.jhep.2016.04.025
Shan S, Shen Z, Zhang C, Kou R, Xie K, Song F (2019) Mitophagy protects against acetaminophen-induced acute liver injury in mice through inhibiting NLRP3 inflammasome activation. Biochem Pharmacol 169:113643. https://doi.org/10.1016/j.bcp.2019.113643
McWilliams TG, Muqit MM (2017) PINK1 and Parkin: emerging themes in mitochondrial homeostasis. Curr Opin Cell Biol 45:83–91. https://doi.org/10.1016/j.ceb.2017.03.013
Wang H, Ni HM, Chao X, Ma X, Rodriguez YA, Chavan H, Wang S, Krishnamurthy P, Dobrowsky R, Xu DX et al (2019) Double deletion of PINK1 and Parkin impairs hepatic mitophagy and exacerbates acetaminophen-induced liver injury in mice. Redox Biol 22:101148. https://doi.org/10.1016/j.redox.2019.101148
Du K, Ramachandran A, McGill MR, Mansouri A, Asselah T, Farhood A, Woolbright BL, Ding WX, Jaeschke H (2017) Induction of mitochondrial biogenesis protects against acetaminophen hepatotoxicity. Food Chem Toxicol 108:339–350. https://doi.org/10.1016/j.fct.2017.08.020
Jaeschke H, Duan L, Nguyen N, Ramachandran A (2019) Mitochondrial damage and biogenesis in acetaminophen-induced liver injury. Liver Res 3:150–156. https://doi.org/10.1016/j.livres.2019.10.002
Zhang T, Zhang Q, Guo J, Yuan H, Peng H, Cui L, Yin J, Zhang L, Zhao J, Li J et al (2016) Non-cytotoxic concentrations of acetaminophen induced mitochondrial biogenesis and antioxidant response in HepG2 cells. Environ Toxicol Pharmacol 46:71–79. https://doi.org/10.1016/j.etap.2016.06.030
Ramachandran A, Jaeschke H (2020) A mitochondrial journey through acetaminophen hepatotoxicity. Food Chem Toxicol 140:111282. https://doi.org/10.1016/j.fct.2020.111282
Liu X, Zhao H, Luo C, Du D, Huang J, Ming Q, Jin F, Wang D, Huang W (2019) Acetaminophen responsive miR-19b modulates SIRT1/Nrf2 signaling pathway in drug-induced hepatotoxicity. Toxicol Sci 170:476–488. https://doi.org/10.1093/toxsci/kfz095
Lazarska KE, Dekker SJ, Vermeulen NPE, Commandeur JNM (2018) Effect of UGT2B7*2 and CYP2C8*4 polymorphisms on diclofenac metabolism. Toxicol Lett 284:70–78. https://doi.org/10.1016/j.toxlet.2017.11.038
den Braver MW, Zhang Y, Venkataraman H, Vermeulen NP, Commandeur JN (2016) Simulation of interindividual differences in inactivation of reactive para-benzoquinone imine metabolites of diclofenac by glutathione S-transferases in human liver cytosol. Toxicol Lett 255:52–62. https://doi.org/10.1016/j.toxlet.2016.05.015
Dara L, Liu ZX, Kaplowitz N (2016) Mechanisms of adaptation and progression in idiosyncratic drug induced liver injury, clinical implications. Liver Int 36:158–165. https://doi.org/10.1111/liv.12988
Kang SW, Haydar G, Taniane C, Farrell G, Arias IM, Lippincott-Schwartz J, Fu D (2016) AMPK activation prevents and reverses drug-induced mitochondrial and hepatocyte injury by promoting mitochondrial fusion and function. PLoS ONE 11:e0165638. https://doi.org/10.1371/journal.pone.0165638
Wink S, Hiemstra SW, Huppelschoten S, Klip JE, van de Water B (2018) Dynamic imaging of adaptive stress response pathway activation for prediction of drug induced liver injury. Arch Toxicol 92:1797–1814. https://doi.org/10.1007/s00204-018-2178-z
Dragovic G, Nikolic K, Dimitrijevic B, Jevtovic D, Salemovic D, Tomanovic N, Boricic I (2019) Severe hepatotoxicity induced by efavirenz in a treatment-naive, low body mass index HIV-infected, female patient with no hepatitis and other virus co-infections. Ultrastruct Pathol 43:220–223. https://doi.org/10.1080/01913123.2019.1673862
Chwiki S, Campos MM, McLaughlin ME, Kleiner DE, Kovacs JA, Morse CG, Abu-Asab MS (2017) Adverse effects of antiretroviral therapy on liver hepatocytes and endothelium in HIV patients: an ultrastructural perspective. Ultrastruct Pathol 41:186–195. https://doi.org/10.1080/01913123.2017.1282066
Ganta KK, Mandal A, Chaubey B (2017) Depolarization of mitochondrial membrane potential is the initial event in non-nucleoside reverse transcriptase inhibitor efavirenz induced cytotoxicity. Cell Biol Toxicol 33:69–82. https://doi.org/10.1007/s10565-016-9362-9
Polo M, Alegre F, Moragrega AB, Gibellini L, Marti-Rodrigo A, Blas-Garcia A, Esplugues JV, Apostolova N (2017) Lon protease: a novel mitochondrial matrix protein in the interconnection between drug-induced mitochondrial dysfunction and endoplasmic reticulum stress. Br J Pharmacol 174:4409–4429. https://doi.org/10.1111/bph.14045
Apostolova N, Gomez-Sucerquia LJ, Gortat A, Blas-Garcia A, Esplugues JV (2011) Autophagy as a rescue mechanism in efavirenz-induced mitochondrial dysfunction: a lesson from hepatic cells. Autophagy 7:1402–1404. https://doi.org/10.4161/auto.7.11.17653
Borsa M, Ferreira PL, Petry A, Ferreira LG, Camargo MM, Bou-Habib DC, Pinto AR (2015) HIV infection and antiretroviral therapy lead to unfolded protein response activation. Virol J 12:77. https://doi.org/10.1186/s12985-015-0298-0
Alegre F, Moragrega AB, Polo M, Marti-Rodrigo A, Esplugues JV, Blas-Garcia A, Apostolova N (2018) Role of p62/SQSTM1 beyond autophagy: a lesson learned from drug-induced toxicity in vitro. Br J Pharmacol 175:440–455. https://doi.org/10.1111/bph.14093
De Bruyn T, Stieger B, Augustijns PF, Annaert PP (2016) Clearance prediction of HIV protease inhibitors in man: role of hepatic uptake. J Pharm Sci 105:854–863. https://doi.org/10.1002/jps.24564
Ganta KK, Chaubey B (2019) Endoplasmic reticulum stress leads to mitochondria-mediated apoptosis in cells treated with anti-HIV protease inhibitor ritonavir. Cell Biol Toxicol 35:189–204. https://doi.org/10.1007/s10565-018-09451-7
Gibellini L, De Biasi S, Pinti M, Nasi M, Riccio M, Carnevale G, Cavallini GM, Sala de Oyanguren FJ, O’Connor JE, Mussini C et al (2012) The protease inhibitor atazanavir triggers autophagy and mitophagy in human preadipocytes. AIDS 26:2017–2026. https://doi.org/10.1097/QAD.0b013e328359b8be
Polus A, Bociaga-Jasik M, Czech U, Goralska J, Cialowicz U, Chojnacka M, Polus M, Jurowski K, DEMBINSKA-KIEC A, (2017) The human immunodeficiency virus (HIV1) protease inhibitor sanquinavir activates autophagy and removes lipids deposited in lipid droplets. J Physiol Pharmacol 68:283–293
Chetty S, Ramesh M, Singh-Pillay A, Soliman ME (2017) Recent advancements in the development of anti-tuberculosis drugs. Bioorg Med Chem Lett 27:370–386. https://doi.org/10.1016/j.bmcl.2016.11.084
Sanjay S, Girish C, Toi PC, Bobby Z (2021) Gallic acid attenuates isoniazid and rifampicin-induced liver injury by improving hepatic redox homeostasis through influence on Nrf2 and NF-κB signalling cascades in wistar rats. J Pharm Pharmacol 73:1–14. https://doi.org/10.1093/jpp/rgaa048
Zhang T, Ikejima T, Li L, Wu R, Yuan X, Zhao J, Wang Y, Peng S (2017) Impairment of mitochondrial biogenesis and dynamics involved in isoniazid-induced apoptosis of HepG2 cells was alleviated by p38 MAPK pathway. Front Pharmacol 8:753. https://doi.org/10.3389/fphar.2017.00753
Jia ZL, Cen J, Wang JB, Zhang F, Xia Q, Wang X, Chen XQ, Wang RC, Hsiao CD, Liu KC et al (2019) Mechanism of isoniazid-induced hepatotoxicity in zebrafish larvae: activation of ROS-mediated ERS, apoptosis and the Nrf2 pathway. Chemosphere 227:541–550. https://doi.org/10.1016/j.chemosphere.2019.04.026
Liang Y, Zhou T, Chen Y, Lin D, Jing X, Peng S, Zheng D, Zeng Z, Lei M, Wu X et al (2017) Rifampicin inhibits rotenone-induced microglial inflammation via enhancement of autophagy. Neurotoxicology 63:137–145. https://doi.org/10.1016/j.neuro.2017.09.015
Lee EH, Baek SY, Park JY, Kim YW (2020) Rifampicin activates AMPK and alleviates oxidative stress in the liver as mediated with Nrf2 signaling. Chem Biol Interact 315:108889. https://doi.org/10.1016/j.cbi.2019.108889
Chaudhury A, Duvoor C, Reddy Dendi VS, Kraleti S, Chada A, Ravilla R, Marco A, Shekhawat NS, Montales MT, Kuriakose K et al (2017) Clinical review of antidiabetic drugs: implications for type 2 diabetes mellitus management. Front Endocrinol (Lausanne) 8:6. https://doi.org/10.3389/fendo.2017.00006
Miralles-Linares F, Puerta-Fernandez S, Bernal-Lopez MR, Tinahones FJ, Andrade RJ, Gomez-Huelgas R (2012) Metformin-induced hepatotoxicity. Diabetes Care 35:e21. https://doi.org/10.2337/dc11-2306
Nanjan MJ, Mohammed M, Prashantha Kumar BR, Chandrasekar MJN (2018) Thiazolidinediones as antidiabetic agents: a critical review. Bioorg Chem 77:548–567. https://doi.org/10.1016/j.bioorg.2018.02.009
Hur KY, Lee MS (2015) New mechanisms of metformin action: focusing on mitochondria and the gut. J Diabetes Investig 6:600–609. https://doi.org/10.1111/jdi.12328
Park J, Joe Y, Ryter SW, Surh YJ, Chung HT (2019) Similarities and distinctions in the effects of metformin and carbon monoxide in immunometabolism. Mol Cells 42:292–300. https://doi.org/10.14348/molcells.2019.0016
Song YM, Lee WK, Lee YH, Kang ES, Cha BS, Lee BW (2016) Metformin restores Parkin-mediated mitophagy, suppressed by cytosolic p53. Int J Mol Sci 17:1–14. https://doi.org/10.3390/ijms17010122
Geng Y, Hernandez Villanueva A, Oun A, Buist-Homan M, Blokzijl H, Faber KN, Dolga A, Moshage H (2020) Protective effect of metformin against palmitate-induced hepatic cell death. Biochim Biophys Acta Mol Basis Dis 1866:165621. https://doi.org/10.1016/j.bbadis.2019.165621
Lee YH, Chung MC, Lin Q, Boelsterli UA (2008) Troglitazone-induced hepatic mitochondrial proteome expression dynamics in heterozygous Sod2(+/−) mice: two-stage oxidative injury. Toxicol Appl Pharmacol 231:43–51. https://doi.org/10.1016/j.taap.2008.03.025
Bjornsson ES (2017) Hepatotoxicity of statins and other lipid-lowering agents. Liver Int 37:173–178. https://doi.org/10.1111/liv.13308
Karahalil B, Hare E, Koc G, Uslu I, Senturk K, Ozkan Y (2017) Hepatotoxicity associated with statins. Arh Hig Rada Toksikol 68:254–260. https://doi.org/10.1515/aiht-2017-68-2994
Mollazadeh H, Atkin SL, Butler AE, Ruscica M, Sirtori CR, Sahebkar A (2018) The effect of statin therapy on endoplasmic reticulum stress. Pharmacol Res 137:150–158. https://doi.org/10.1016/j.phrs.2018.10.006
Bao S, Lin J, Xie M, Wang C, Nie X (2021) Simvastatin affects Nrf2/MAPK signaling pathway and hepatic histological structure change in Gambusia affinis. Chemosphere 269:128725. https://doi.org/10.1016/j.chemosphere.2020.128725
Farag MM, Mohamed MB, Youssef EA (2015) Assessment of hepatic function, oxidant/antioxidant status, and histopathological changes in rats treated with atorvastatin: effect of dose and acute intoxication with acetaminophen. Hum Exp Toxicol 34:828–837. https://doi.org/10.1177/0960327114559991
Guixe-Muntet S, de Mesquita FC, Vila S, Hernandez-Gea V, Peralta C, Garcia-Pagan JC, Bosch J, Gracia-Sancho J (2017) Cross-talk between autophagy and KLF2 determines endothelial cell phenotype and microvascular function in acute liver injury. J Hepatol 66:86–94. https://doi.org/10.1016/j.jhep.2016.07.051
Andres AM, Hernandez G, Lee P, Huang C, Ratliff EP, Sin J, Thornton CA, Damasco MV, Gottlieb RA (2014) Mitophagy is required for acute cardioprotection by simvastatin. Antioxid Redox Signal 21:1960–1973. https://doi.org/10.1089/ars.2013.5416
Ashrafizadeh M, Ahmadi Z, Farkhondeh T, Samarghandian S (2020) Modulatory effects of statins on the autophagy: a therapeutic perspective. J Cell Physiol 235:3157–3168. https://doi.org/10.1002/jcp.29227
Schirrmacher V (2019) From chemotherapy to biological therapy: a review of novel concepts to reduce the side effects of systemic cancer treatment (Review). Int J Oncol 54:407–419. https://doi.org/10.3892/ijo.2018.4661
Guo X, Li W, Hu J, Zhu EC, Su Q (2020) Hepatotoxicity in patients with solid tumors treated with PD-1/PD-L1 inhibitors alone, PD-1/PD-L1 inhibitors plus chemotherapy, or chemotherapy alone: systematic review and meta-analysis. Eur J Clin Pharmacol 76:1345–1354. https://doi.org/10.1007/s00228-020-02903-2
Song J, Zhao W, Lu C, Shao X (2019) LATS2 overexpression attenuates the therapeutic resistance of liver cancer HepG2 cells to sorafenib-mediated death via inhibiting the AMPK-Mfn2 signaling pathway. Cancer Cell Int 19:60. https://doi.org/10.1186/s12935-019-0778-1
Dirks-Naylor AJ, Kouzi SA, Bero JD, Phan DT, Taylor HN, Whitt SD, Mabolo R (2014) Doxorubicin alters the mitochondrial dynamics machinery and mitophagy in the liver of treated animals. Fundam Clin Pharmacol 28:633–642. https://doi.org/10.1111/fcp.12073
Yan C, Li TS (2018) Dual role of mitophagy in cancer drug resistance. Anticancer Res 38:617–621. https://doi.org/10.21873/anticanres.12266
Funding
Funding for this study was provided by National Nature Science Foundation of China (Grant No. 81100281), Hubei Provincial Natural Science Foundation of China (Grant No. 2020CFB371), and Medical and Health Research Project from Yichang Science and Technology Bureau (Grant No. A15301-35).
Author information
Authors and Affiliations
Contributions
WH, XY, JZ, QY and CZ contributed to the writing, review, and editing of the manuscript. JZ made the figures and tables. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zheng, J., Yuan, Q., Zhou, C. et al. Mitochondrial stress response in drug-induced liver injury. Mol Biol Rep 48, 6949–6958 (2021). https://doi.org/10.1007/s11033-021-06674-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06674-6