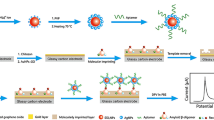
Abstract
Molecularly imprinted polymers (MIP) consist of a molecular recognition technology with applicability in different areas, including forensic chemistry. Among the forensic applications, the use of MIP in biological fluid analysis has gained prominence. Biological fluids are complex samples that generally require a pre-treatment to eliminate interfering agents to improve the results of the analyses. In this review, we address the development of this molecular imprinting technology over the years, highlighting the forensic applications of molecularly imprinted polymers in biological sample preparation for analysis of stimulant drugs such as cocaine, amphetamines, and nicotine.
Graphical abstract






Similar content being viewed by others
Abbreviations
- AIBN:
-
2,2′-Azobisisobutironitrile
- AMP:
-
Amphetamine
- BE:
-
Benzoylecgonine
- BSA:
-
Bovine serum albumin
- CE:
-
Cocaethylene
- COC:
-
Cocaine
- EGDMA:
-
Ethylene glycol dimethacrylate
- EME:
-
Ecgonine methyl ester
- FM:
-
Functional monomer
- GC:
-
Gas chromatography
- GC–MS:
-
Gas chromatography coupled to mass spectrometry
- GDMA:
-
Glycerol dimethacrylate
- HEMA:
-
Hydroxyethyl methacrylate
- HPLC:
-
High-pressure liquid chromatography
- HPLC–MS/MS:
-
High-pressure liquid chromatography coupled to tandem mass spectrometry
- IMS:
-
Ion-mobility spectrometry
- LC–MS:
-
Liquid chromatography coupled to mass spectrometry
- LLE:
-
Liquid–liquid extraction
- LSD:
-
Lysergic acid diethylamide
- MAA:
-
Methacrylic acid
- MAMP:
-
Methamphetamine
- MDMA:
-
3,4-Methylenedioxymethamphetamine
- MEPS:
-
Microextraction by packed sorbent
- MIP:
-
Molecularly imprinted polymer
- MISPE:
-
Molecularly imprinted solid-phase extraction
- NCT:
-
Nicotine
- NIP:
-
Non-imprinted polymer
- PSI–MS:
-
Paper spray ionization tandem mass spectrometry
- RAM:
-
Restricted access materials
- RAMIP:
-
Restricted access molecularly imprinted polymer
- SC:
-
Synthetic cathinones
- SPE:
-
Solid-phase extraction
- SPME:
-
Solid-phase microextraction
- TRIM:
-
Trimethacrylate trimethylolpropane
- μ-SPE:
-
Micro-solid-phase extraction
References
Tarley CRT, Sotomayor MDPT, Kubota LT. Polímeros biomiméticos em química analítica. Parte 1: Preparo e aplicações de MIP em técnicas de extração e separação. Quim Nova. 2005;28(6):1076–86 Available from: http://www.scielo.br/pdf/qn/v28n6/26840.pdf.
Wulff G Molecular recognition in polymers prepared by imprinting with templates. In: Polymeric Reagents and Catalysts. 1986. p. 186–229.
Figueiredo EC, Dias ACB, Arruda MAZ. Impressão molecular: Uma estratégia promissora na elaboração de matrizes para a liberação controlada de fármacos. Braz J Pharm Sci. 2008;44(3):361–75.
Borges KB, de Figueiredo EC, Queiroz MEC. Preparo de Amostras para Análise de Compostos Orgânicos. 1st ed. Rio de Janeiro: LTC; 2015. p. 288.
Gama MR, Bottoli CBG. Molecularly imprinted polymers for bioanalytical sample preparation. J Chromatogr B Anal Technol Biomed Life Sci. 2017;1043:107–21. https://doi.org/10.1016/j.jchromb.2016.09.045.
Herrera-Chacón A, Cetó X, del Valle M. Molecularly imprinted polymers - towards electrochemical sensors and electronic tongues [internet]. Analytical and bioanalytical chemistry. Springer Science and Business Media Deutschland GmbH; 2021 [cited 2021 Jul 7]. p. 1–24. Available from: https://doi.org/10.1007/s00216-021-03313-8.
Ye L, Cormack PAG, Mosbach K Molecularly imprinted monodisperse microspheres for competitive radioassay.
Sorribes-Soriano A, Esteve-Turrillas FA, Armenta S, de la Guardia M, Herrero-Martínez JM. Cocaine abuse determination by ion mobility spectrometry using molecular imprinting. J Chromatogr A. 2017;1481:23–30. https://doi.org/10.1016/j.chroma.2016.12.041.
Marfà J, Pupin RR, Sotomayor M, Pividori MI. Magnetic-molecularly imprinted polymers in electrochemical sensors and biosensors. Anal Bioanal Chem [Internet]. 2021 Jun 23 [cited 2021 Jul 7];1–17. Available from: https://link.springer.com/10.1007/s00216-021-03461-x
Djozan D, Ebrahimi B. Preparation of new solid phase micro extraction fiber on the basis of atrazine-molecular imprinted polymer: application for GC and GC/MS screening of triazine herbicides in water, rice and onion. Anal Chim Acta. 2008;616(2):152–9.
Sánchez-González J, Tabernero MJ, Bermejo AM, Bermejo-Barrera P, Moreda-Piñeiro A. Porous membrane-protected molecularly imprinted polymer micro-solid-phase extraction for analysis of urinary cocaine and its metabolites using liquid chromatography - tandem mass spectrometry. Anal Chim Acta. 2015;898:50–9.
Kamari K, Taheri A. Preparation and evaluation of magnetic core–shell mesoporous molecularly imprinted polymers for selective adsorption of amitriptyline in biological samples. J Taiwan Inst Chem Eng. 2018;86:230–9.
Chen L, Wang X, Lu W, Wu X, Li J. Molecular imprinting: perspectives and applications. Chem Soc Rev. 2016;45(8):2137–211. https://doi.org/10.1039/C6CS00061D.
Jardim ICSF. Extração em Fase Sólida : Fundamentos Teóricos e Novas Estratégias para Preparação de Fases Sólidas. Sci Chromatogr. 2010;2(1):13–25.
Majors RE. Overview of sample preparation. LCGC [Internet]. 1991 [cited 2020 Jul 31];9(1):16–20. Available from: http://www.chromatographyonline.com/overview-sample-preparation
Li G, Row KH. Recent applications of molecularly imprinted polymers (MIPs) on micro-extraction techniques. Sep Purif Rev. 2018;47(1):1–18. https://doi.org/10.1080/15422119.2017.1315823.
Abdel-Rehim M. Microextraction by packed sorbent (MEPS): a tutorial. Anal Chim Acta. Elsevier. 2011;701:119–28.
Nascimento RF, Melo DQ, Lima ACA, Barros AL, Vidal CB, Raulino GSC. (2014) Equilíbrio de adsorção. Adsorção aspectos teóricos e aplicações ambientais. Fortaleza. 256 p.
Thibert V, Legeay P, Chapuis-Hugon F, Pichon V. Molecularly imprinted polymer for the selective extraction of cocaine and its metabolites, benzoylecgonine and ecgonine methyl ester, from biological fluids before LC-MS analysis. J Chromatogr B Anal Technol Biomed Life Sci. 2014;949–950:16–23.
Wulff G, Sarhan A. The use of polymers with enzyme-analogous structures for the resoltution of racemates. 11th ed. Bonn: Angewandte Chemie International Edition; 1972. https://doi.org/10.1002/anie.197203341.
Wulff G, Vesper W, Grobe-Einsler R, Sarhan A. (1977) Enzyme-analogue built polymers, 4. Die Makromol Chemie 178:2799–816.
Wulff G, Grobe-Einsler R, Vesper W, Sarhan A. Enzyme-analogue built polymers, 5*. Die Makromol Chemie. 1977;178:2817–25.
Andersson LI, Mosbach K. Enantiomeric resolution on molecularly imprinted polymers prepared with only non-covalent and non-ionic interactions. J Chromatogr [Internet]. 1990 1 [cited 2020 May 4];516(2):313–22. Available from: https://www.sciencedirect.com/science/article/pii/S0021967301892736
Panasyuk TL, Mirsky VM, Piletsky SA, Wolfbeis OS. Electropolymerized molecularly imprinted polymers as receptor layers in capacitive chemical sensors. Anal Chem. 1999;71(20):4609–13.
Tai D-F, Lin C-Y, Wu T-Z, Huang J-H, Shu P-Y. (2006) Artificial receptors in serologic tests for the early diagnosis of dengue virus infection. Clin Chem [Internet]. [cited 2020 May 22];52(8):1486–91. Available from: https://academic.oup.com/clinchem/article/52/8/1486/5626915
Patra S, Roy E, Madhuri R, Sharma PK. Nano-iniferter based imprinted sensor for ultra trace level detection of prostate-specific antigen in both men and women. Biosens Bioelectron. 2015;66:1–10.
Denderz N, Lehotay J, Čižmárik J, Cibulková Z, Šimon P. Thermodynamic study of molecularly imprinted polymer used as the stationary phase in high performance liquid chromatography. J Chromatogr A. 2012;1235:77–83.
da Silva AS, de Oliveira HL, da Silva ATM, do Nascimento TA, Borges MMC, Silva RCDS, et al. Assessment of the performance of solid phase extraction based on pipette tip employing a hybrid molecularly imprinted polymer as an adsorbent for enantioselective determination of albendazole sulfoxide. J Chromatogr Sci. 2019;57(7):671–8.
de Faria HD, de Carvalho ALC, Santos MG, Barbosa AF, Figueiredo EC. New advances in restricted access materials for sample preparation: a review. Anal Chim Acta. 2017;959:43–65.
Mendes TV, Franqui LS, Santos MG, Wisniewski C, Figueiredo EC. Synthesis and characterization of a new magnetic restricted access molecularly imprinted polymer for biological sample preparation. Mater Today Commun. 2020;24:101002.
Moraes GDOI, Da Silva LMR, Dos Santos-Neto ÁJ, Florenzano FH, Figueiredo EC. A new restricted access molecularly imprinted polymer capped with albumin for direct extraction of drugs from biological matrices: the case of chlorpromazine in human plasma. Anal Bioanal Chem. 2013;405(24):7687–96.
Oliveira HL, de Teixeira LS, Dinali LAF, Pires BC, Simões NS, Borges KB. Microextraction by packed sorbent using a new restricted molecularly imprinted polymer for the determination of estrogens from human urine samples. Microchem J. 2019;150:104162. https://doi.org/10.1016/j.microc.2019.104162.
Pereira E, Cáceres C, Rivera F, Rivas B, Sáez P. Preparation of molecularly imprinted polymers for diphenylamine removal from organic gunshot residues. J Chil Chem Soc. 2015;59(4):2731–6.
Zarei AR, Zafarghandi RS. Selective determination of 2,4,6-trinitrotoluene in water samples based on magnetic imprinted nanoparticles via grafting polymerization. J Braz Chem Soc. 2015;26(4):741–7.
Jackson R, Petrikovics I, Lai EPC, Yu JCC. Molecularly imprinted polymer stir bar sorption extraction and electrospray ionization tandem mass spectrometry for determination of 2-aminothiazoline-4- carboxylic acid as a marker for cyanide exposure in forensic urine analysis. Anal Methods. 2010;2(5):552–7.
Peñuela-Pinto O, Armenta S, Esteve-Turrillas FA, de la Guardia M. Selective determination of clenbuterol residues in urine by molecular imprinted polymer—ion mobility spectrometry. Microchem J. 2017;134:62–7.
Koster EHM, Crescenzi C, Den Hoedt W, Ensing K, De Jong GJ. Fibers coated with molecularly imprinted polymers for solid-phase microextraction. Anal Chem. 2001;73(13):3140–5.
Hu Y, Feng S, Gao F, Li-Chan ECY, Grant E, Lu X. Detection of melamine in milk using molecularly imprinted polymers-surface enhanced Raman spectroscopy. Food Chem. 2015;176:123–9. https://doi.org/10.1016/j.foodchem.2014.12.051.
Alizadeh T, Ganjali MR, Zare M, Norouzi P. Selective determination of chloramphenicol at trace level in milk samples by the electrode modified with molecularly imprinted polymer. Food Chem. 2012;130(4):1108–14.
Hassan AHA, Moura SL, Ali FHM, Moselhy WA, del Pilar Taboada Sotomayor M, Pividori MI. Electrochemical sensing of methyl parathion on magnetic molecularly imprinted polymer. Biosens Bioelectron. 2018;118:181–7.
Dai C-M, Geissen S-U, Zhang Y-L, Zhang Y-J, Zhou X-F. Selective removal of diclofenac from contaminated water using molecularly imprinted polymer microspheres. Environ Pollut. 2011;159(6):1660–6.
De Barros LA, Pereira LA, Custódio R, Rath S. A novel computational approach for development of highly selective fenitrothion imprinted polymer: theoretical predictions and experimental validations. J Braz Chem Soc. 2014;25(4):619–28.
Nestić M, Babić S, Pavlović DM, Sutlović D. Molecularly imprinted solid phase extraction for simultaneous determination of δ9-tetrahydrocannabinol and its main metabolites by gas chromatography-mass spectrometry in urine samples. Forensic Sci Int. 2013;231(1–3):317–24.
Masteri-Farahani M, Mashhadi-Ramezani S, Mosleh N. Molecularly imprinted polymer containing fluorescent graphene quantum dots as a new fluorescent nanosensor for detection of methamphetamine. Spectrochim Acta - Part A Mol Biomol Spectrosc. 2020;229:118021.
Ti L, Richardson L, DeBeck K, Nguyen P, Montaner J, Wood E, et al. The impact of engagement in street-based income generation activities on stimulant drug use cessation among people who inject drugs. Drug Alcohol Depend. 2014;141:58–64.
Fonseca Pego AM, de Souza Eller Franco de Oliveira SCW, Franco de Oliveira T, Leyton V, Miziara I, Yonamine M. Cocaine toxicological findings in cases of violent death in Sao Paulo city - Brazil. J Forensic Legal Med. 2018;60:3–8.
de Silva ATM, Bessa CDPB, de Borges WS, Borges KB. Bioanalytical methods for determining ecstasy components in biological matrices: a review. TrAC - Trends Ana Chem. Elsevier B.V. 2018;108:323–46.
Aria M, Cuccurullo C. Bibliometrix: an R-tool for comprehensive science mapping analysis. J Inf Secur. 2017;11(4):959–75.
Caligiorne SM, Marinho PA. Cocaína: aspectos históricos, toxicológicos e analíticos - uma revisão. Rev Crim e Med Leg. 2016;1(1):34–45.
UNODC -United Nations Office on Drugs and Crime. (2019) World drug report 2019. [cited 2020 Jul 23]. Available from: (United Nations publication, Sales No. E.19.XI.8).
Randall B (2004) Disposition of toxic drugs and chemicals in man, seven edition. Biomed Publ
Dean RA, Christian CD, Sample RHB, Bosron WF. (1991) Human liver cocaine esterases: ethanol-mediated formation of ethylcocaine. FASEB J
da Cruz RA, do Carmo Santos Guedes M. (2013) Cocaína: Aspectos Toxicológico E Analítico. Rev Eletrônica FACP. 1–15.
Feitosa RS, Sodré FF, Maldaner AO. Drogas de abuso em águas naturais e residuárias urbanas: Ocorrência, determinação e aplicações forenses. Quim Nova. 2013;36(2):291–305.
Bujak R, Gadzała-Kopciuch R, Nowaczyk A, Raczak-Gutknecht J, Kordalewska M, Struck-Lewicka W, et al. (2016) New sorbent materials for selective extraction of cocaine and benzoylecgonine from human urine samples. J Pharm Biomed Anal
Sánchez-González J, Jesús Tabernero M, María Bermejo A, Bermejo-Barrera P, Moreda-Piñeiro A. Development of magnetic molecularly imprinted polymers for solid phase extraction of cocaine and metabolites in urine before high performance liquid chromatography - tandem mass spectrometry. Talanta. 2016;147:641–9.
Schramm W, Smith RH, Craig PA. (1993) Methods of simplified saliva collection for the measurement of drugs of abuse, therapeutic drugs, and other molecules. Ann N Y Acad Sci
Chiappin S, Antonelli G, Gatti R, De Palo EF. (2007) Saliva specimen: a new laboratory tool for diagnostic and basic investigation. Clin Chim Acta
Bueno LHP, da Silva RHA, Azenha AV, de Souza Dias MC, De Martinis BS. (2014) Oral fluid as an alternative matrix to determine ethanol for forensic purposes. Forensic Sci Int
Kintz P, Cirimele V, Ludes B. (2000) Detection of cannabis in oral fluid (saliva) and forehead wipes (sweat) from impaired drivers. J Anal Toxicol
Samyn N, De Boeck G, Verstraete AG. (2002) The use of oral fluid and sweat wipes for the detection of drugs of abuse in drivers. J Forensic Sci
Verstraete AG. (2005) Oral fluid testing for driving under the influence of drugs: history, recent progress and remaining challenges. Forensic Sci Int
Drummer OH. (2008) Introduction and review of collection techniques and applications of drug testing of oral fluid. In: Therapeutic Drug Monitoring
Vindenes V, Yttredal B, Øiestad EL, Waal H, Bernard JP, Mørland JG, et al. (2011) Oral fluid is aviable alternative for monitoring drug abuse: detection of drugs in oral fluid by liquid chromatography-tandem mass spectrometry and comparison to the results from urine samples from patients treated with methadone or buprenorphine. J Anal Toxicol
Cone EJ. (1990) Testing human hair for drugs of abuse. i. individual dose and time profiles of morphine and codeine in plasma, saliva, urine, and beard compared to drug-induced effects on pupils and behavior. J Anal Toxicol
Cone EJ. (1993) Saliva testing for drugs of abuse. Ann N Y Acad Sci
Clauwaert K, Decaestecker T, Mortier K, Lambert W, Deforce D, Van Peteghem C, et al. (2004) The determination of cocaine, benzoylecgonine, and cocaethylene in small-volume oral fluid samples by liquid chromatography-quadrupole-time-of- flight mass spectrometry. J Anal Toxicol
De Jager L, Andrews, ARJ. (2002) Development of a screening method for cocaine and cocaine metabolites in saliva using hollow fiber membrane solvent microextraction. Anal Chim Acta
Sousa DVM, Pereira F V., Nascentes CC, Moreira JS, Boratto VHM, Orlando RM. (2020) Cellulose cone tip as a sorbent material for multiphase electrical field-assisted extraction of cocaine from saliva and determination by LC-MS/MS. Talanta
Meng P, Wang Y. (2010) Small volume liquid extraction of amphetamines in saliva. Forensic Sci Int
Smith FP. (1981) Detection of amphetamine in bloodstains, semen, seminal stains, saliva, and saliva stains. Forensic Sci Int
Amiri-Aref M, Raoof JB, Ojani R. (2013) Electrocatalytic oxidation and selective determination of an opioid analgesic methadone in the presence of acetaminophen at a glassy carbon electrode modified with functionalized multi-walled carbon nanotubes: application for human urine, saliva and pharm. Colloids Surf B Biointerfaces
Yonamine M, Tawil N, De Moraes Moreau RL, Alves Silva O. (2003) Solid-phase micro-extraction–gas chromatography–mass spectrometry and headspace-gas chromatography of tetrahydrocannabinol, amphetamine, methamphetamine, cocaine and ethanol in saliva samples. J Chromatogr B Anal Technol Biomed Life Sci
Fucci N, De Giovanni N, Chiarotti M, Scarlata S. (2001) SPME-GC analysis of THC in saliva samples collected with “EPITOPE” device. Forensic Sci Int
Maseda C, Hama K, Fukui Y, Matsubara K, Takahashi S, Akane A. (1986) Detection of Δ9-THC in saliva by capillary GC ECD after marihuana smoking. Forensic Sci Int
Schnoll R, Paul Wileyto E, Gross R, Hitsman B, Hawk LW, Cinciripini P, et al. (2020) Evaluation of nicotine patch adherence measurement using self-report and saliva cotinine among abstainers in a smoking cessation trial. Drug Alcohol Depend
Parzynski CS, Jaszyna-Gasior M, Franken FH, Moolchan ET. (2008) Measuring nicotine intake among highly-dependent adolescent smokers: comparability of saliva and plasma cotinine concentrations. Pharmacol Biochem Behav
Kardani F, Daneshfar A, Sahrai R. (2010) Determination of nicotine, anabasine, and cotinine in urine and saliva samples using single-drop microextraction. J Chromatogr B Anal Technol Biomed Life Sci
Sorribes-Soriano A, Esteve-Turrillas FA, Armenta S, Montoya A, Herrero-Martínez JM, de la Guardia M. Magnetic molecularly imprinted polymers for the selective determination of cocaine by ion mobility spectrometry. J Chromatogr A. 2018;1545:22–31.
Tavares LS, Carvalho TC, Romão W, Vaz BG, Chaves AR. Paper spray tandem mass spectrometry based on molecularly imprinted polymer substrate for cocaine analysis in oral fluid. J Am Soc Mass Spectrom. 2018;29(3):566–72.
Sánchez-González J, García-Carballal S, Cabarcos P, Tabernero MJ, Bermejo-Barrera P, Moreda-Piñeiro A. Determination of cocaine and its metabolites in plasma by porous membrane-protected molecularly imprinted polymer micro-solid-phase extraction and liquid chromatography-tandem mass spectrometry. J Chromatogr A. 2016;1451:15–22.
Wackerlig J, Lieberzeit PA. (2015) Molecularly imprinted polymer nanoparticles in chemical sensing - synthesis, characterisation and application. Sensors and Actuators, B: Chemical
Sánchez-González J, Barreiro-Grille T, Cabarcos P, Tabernero MJ, Bermejo-Barrera P, Moreda-Piñeiro A. Magnetic molecularly imprinted polymer based - micro-solid phase extraction of cocaine and metabolites in plasma followed by high performance liquid chromatography - tandem mass spectrometry. Microchem J. 2016;127:206–12.
Thibert V, Legeay P, Chapuis-Hugon F, Pichon V. Synthesis and characterization of molecularly imprinted polymers for the selective extraction of cocaine and its metabolite benzoylecgonine from hair extract before LC-MS analysis. Talanta. 2012;88:412–9.
Matsui J, Doblhoff-Dier O, Takeuchi T. 2-(Trifluoromethyl)acrylic acid: a novel functional monomer in non-covalent molecular imprinting. Anal Chim Acta. 1997;343(1–2):1–4.
Matsui J, Takeuchi T. A molecularly imprinted polymer rod as nicotine selective affinity media prepared with 2-(trifluoromethyl)acrylic acid. Anal Commun [Internet]. 1997 [cited 2021 Feb 1];34(7):199–200. Available from: https://pubs.rsc.org/en/content/articlehtml/1997/ac/a703189k
Matsui J, Kaneko A, Miyoshi Y, Yokoyama K, Tamiya E, Takeuchi T. A molecularly imprinted nicotine-selective polymer. Anal Lett [Internet]. 1996 [cited 2021 1];29(12):2071–8. Available from: https://www.tandfonline.com/doi/abs/10.1080/00032719608002231
Zander Å, Findlay P, Renner T, Sellergren B, Swietlow A. Analysis of nicotine and its oxidation products in nicotine chewing gum by a molecularly imprinted solid-phase extraction. Anal Chem [Internet]. 1998 [cited 2021 Feb 1];70(15):3304–14. Available from: https://pubs.acs.org/sharingguidelines
Mullett WM, Lai EPC, Sellergren B. Determination of nicotine in tobacco by molecularly imprinted solid phase extraction with differential pulsed elution. Anal Commun [Internet]. 1999 [cited 2021 Feb 1];36(6):217–20. Available from: https://pubs.rsc.org/en/content/articlehtml/1999/ac/a902509j
Sambe H, Hoshina K, Moaddel R, Wainer IW, Haginaka J. Uniformly-sized, molecularly imprinted polymers for nicotine by precipitation polymerization. J Chromatogr A. 2006;1134(1–2):88–94.
Liu Y, Liu X, Wang J. Molecularly imprinted solid-phase extraction sorbent for removal of nicotine from tobacco smoke. Anal Lett [Internet]. 2003 [cited 2021 Feb 1];36(8):1631–45. Available from: https://www.tandfonline.com/doi/abs/10.1081/AL-120021554
Huang XH, Song JJ, Li H, Gong MT, Zhang Y. Selective removal of nicotine from the main stream smoke by using a surface-imprinted polymer monolith as adsorbent. J Hazard Mater. 2019;365:53–63.
Horemans F, Weustenraed A, Spivak D, Cleij TJ. (2012) Towards water compatible MIPs for sensing in aqueous media. J Mol Recognit. [cited 2021 Feb 1];25(6):344–51. Available from: http://doi.wiley.com/10.1002/jmr.2191
Lu C, Li H, Xu M, Wang S, Li G, Zhong W, et al. (2015) Preparation of nicotine-imprinted monolith by in-situ surface imprinting onto internal hole surface of macroporous silica for selective enrichment and separation of nicotine in environmental water sample. Sep Sci Technol [Internet]. [cited 2021 1];50(14):2124–33. Available from: https://www.tandfonline.com/doi/abs/10.1080/01496395.2015.1041977
Xie J, Zhou B, Zhang T, Zeng X, Yang M, Wang W, et al. (2018) Preparation of nicotine surface molecularly imprinted polymers for selective solid-phase extraction of nicotine from zero-level refill liquids of electronic cigarettes. Anal Methods [Internet]. [cited 2021 Feb 1];10(29):3637–44. Available from: https://pubs.rsc.org/en/content/articlehtml/2018/ay/c8ay00616d
Nogami H, Nakahori Y, Murashima T, Matsui J. (2017) Nicotine-selective polymeric adsorbent obtained by molecular imprinting with excess use of itaconic acid. Chromatography [Internet]. [cited 2021 Feb 1];38(1):15–21. Available from: https://www.jstage.jst.go.jp/article/jpchrom/38/1/38_2016.017/_article
Abdelkader MSA, Lockwood B, Sansongsak P. (2010) Uptake of nicotine from suspension culture of Nicotiana tabacum by molecularly imprinted polymers. J Pharm Pharmacol [Internet]. 1 [cited 2021 Feb 1];62(5):633–7. Available from: http://doi.wiley.com/10.1211/jpp.62.05.0011
Cegłowski M, Smoluch M, Reszke E, Silberring J, Schroeder G. (2017) Molecularly imprinted polymers as selective adsorbents for ambient plasma mass spectrometry. Anal Bioanal Chem [Internet]. [cited 2021 Feb 1];409(13):3393–405. Available from: https://link.springer.com/article/10.1007/s00216-017-0281-2
Yang J, Hu Y, Cai JB, Zhu XL, Su QD, Hu YQ, et al. (2007) Selective hair analysis of nicotine by molecular imprinted solid-phase extraction: An application for evaluating tobacco smoke exposure. Food Chem Toxicol [Internet]. [cited 2021 Feb 1];45(6):896–903. Available from: https://pubmed.ncbi.nlm.nih.gov/17222493/
Figueiredo EC, de Oliveira DM, de Siqueira MEPB, Arruda MAZ. On-line molecularly imprinted solid-phase extraction for the selective spectrophotometric determination of nicotine in the urine of smokers. Anal Chim Acta. 2009 Mar 2;635(1):102–7.
Franqui LS, Santos MG, Virtuoso LS, Maia PP, Figueiredo EC. (2015) Synthesis and characterization of a magnetic molecularly imprinted polymer for the selective extraction of nicotine and cotinine from urine samples followed by GC-MS analysis. Anal Methods [Internet]. 7 [cited 2021 Feb 1];7(21):9237–44. Available from: www.rsc.org/methods
Hashemi SH, Keykha F. (2019) Application of the response surface methodology in the optimization of modified molecularly imprinted polymer based pipette-Tip micro-solid phase extraction for spectrophotometric determination of nicotine in seawater and human plasma. Anal Methods [Internet]. [cited 2021 Feb 1];11(42):5405–12. Available from: https://pubs.rsc.org/en/content/articlehtml/2019/ay/c9ay01496a
Ruela ALM, Figueiredo EC, Pereira GR. Molecularly imprinted polymers as nicotine transdermal delivery systems. Chem Eng J. 2014;248:1–8.
Ruela ALM, de Figueiredo EC, Carvalho FC, de Araújo MB, Pereira GR. Adsorption and release of nicotine from imprinted particles synthesised by precipitation polymerisation: optimising transdermal formulations. Eur Polym J. 2018 Mar 1;100:67–76.
Heal DJ, Smith SL, Gosden J, Nutt DJ. (2013) Amphetamine, past and present - a pharmacological and clinical perspective. [Internet]. Vol. 27, Journal of Psychopharmacology. SAGE PublicationsSage UK: London, England [cited 2021 Jan 28]. p. 479–96. Available from: http://journals.sagepub.com/doi/10.1177/0269881113482532
Díaz-Liñán MC, García-Valverde MT, Lucena R, Cárdenas S, López-Lorente AI. Dual-template molecularly imprinted paper for the determination of drugs of abuse in saliva samples by direct infusion mass spectrometry. Microchem J. 2021;160:105686.
De La Torre R, Farré M, Navarro M, Pacifici R, Zuccaro P, Pichini S. (2004) Clinical pharmacokinetics of amfetamine and related substances: monitoring in conventional and non-conventional matrices. [Internet]. Vol. 43, Clinical Pharmacokinetics. Springer; [cited 2021 Jan 28]. p. 157–85. Available from: https://link.springer.com/article/10.2165/00003088-200443030-00002
Robitaille C, Collin J. (2016) Prescription psychostimulant use among young adults: a narrative review of qualitative studies. Subst Use Misuse [Internet]. [cited 2021 Jan 28];51(3):357–69. Available from: https://www.tandfonline.com/doi/abs/10.3109/10826084.2015.1110170
Chan B, Freeman M, Kondo K, Ayers C, Montgomery J, Paynter R, et al. (2019) Pharmacotherapy for methamphetamine/amphetamine use disorder—a systematic review and meta-analysis. Addiction [Internet]. [cited 2021 Jan 28];114(12):2122–36. Available from: https://onlinelibrary.wiley.com/doi/10.1111/add.14755
Djozan D, Farajzadeh MA, Sorouraddin SM, Baheri T. (2011) Synthesis and application of high selective monolithic fibers based on molecularly imprinted polymer for SPME of trace methamphetamine. Chromatographia [Internet]. [cited 2021 Jan 28];73(9–10):975–83. Available from: https://link.springer.com/article/10.1007/s10337-011-1984-8
Djozan D, Farajzadeh MA, Sorouraddin SM, Baheri T. Molecularly imprinted-solid phase extraction combined with simultaneous derivatization and dispersive liquid-liquid microextraction for selective extraction and preconcentration of methamphetamine and ecstasy from urine samples followed by gas chromatography. J Chromatogr A. 2012;1248:24–31.
Djozan D, Farajzadeh MA, Sorouraddin SM, Baheri T. (2012) Determination of methamphetamine, amphetamine and ecstasy by inside-needle adsorption trap based on molecularly imprinted polymer followed by GC-FID determination. Microchim Acta [Internet]. [cited 2021 Jan 28];179(3–4):209–17. Available from: www.merck.com
El-Beqqali A, Andersson LI, Jeppsson AD, Abdel-Rehim M. Molecularly imprinted polymer-sol-gel tablet toward micro-solid phase extraction: II. Determination of amphetamine in human urine samples by liquid chromatography–tandem mass spectrometry. J Chromatogr B Anal Technol Biomed Life Sci. 2017;1063:130–5.
Kumazawa T, Hasegawa C, Hara K, Uchigasaki S, Lee X-P, Seno H, et al. (2012) Molecularly imprinted solid-phase extraction for the selective determination of methamphetamine, amphetamine, and methylenedioxyphenylalkylamine designer drugs in human whole blood by gas chromatography-mass spectrometry. J Sep Sci [Internet]. 1 [cited 2021 Jan 28];35(5–6):726–33. Available from: http://doi.wiley.com/10.1002/jssc.201100924
Murakami T, Iwamuro Y, Ishimaru R, Chinaka S, Hasegawa H. (2018) Molecularly imprinted polymer solid-phase extraction of synthetic cathinones from urine and whole blood samples. J Sep Sci [Internet]. [cited 2021 Jan 28];41(24):4506–14. Available from: https://doi.org/10.1002/jssc.201800874.
Sorribes-Soriano A, Esteve-Turrillas FA, Armenta S, Amorós P, Herrero-Martínez JM. Amphetamine-type stimulants analysis in oral fluid based on molecularly imprinting extraction. Anal Chim Acta. 2019;1052:73–83.
Acknowledgements
The authors would like to thank the Brazilian agencies CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico), FAPES (Fundação de Amparo à Pesquisa do Estado do Espírito Santo), FAPEMIG (Fundação de Amparo à Pesquisa do Estado de Minas Gerais), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES, Finance Code 001) for financial support. This study was financed in part by the project involving the Rede Mineira de Química (RQ-MG) supported by FAPEMIG (Project: REDE-113/10; Project: CEX-RED-0010-14).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ferreira, J.B., de Jesus Macrino, C., Dinali, L.A.F. et al. Molecularly imprinted polymers as a selective sorbent for forensic applications in biological samples—a review. Anal Bioanal Chem 413, 6013–6036 (2021). https://doi.org/10.1007/s00216-021-03596-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-021-03596-x