Abstract
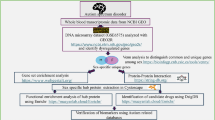
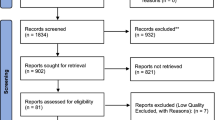
Autism spectrum disorder (ASD) is a hereditary heterogeneous neurodevelopmental disorder characterized by social and speech dysplasia. We collected the expression profiles of ASD in GSE26415, GSE42133 and GSE123302 from the gene expression omnibus (GEO) database, as well as methylation data of GSE109905. Differentially expressed genes (DEGs) between ASD and controls were obtained by differential expression analysis. Enrichment analysis identified the biological functions and signaling pathways involved by common genes in three groups of DEGs. Protein-protein interaction (PPI) networks were used to identify genes with the highest connectivity as key genes. In addition, we identified methylation markers by associating differentially methylated positions. Key methylation markers were identified using the least absolute shrink and selection operator (LASSO) model. Receiver operating characteristic curves and nomograms were used to identify the diagnostic role of key methylation markers for ASD. A total of 57 common genes were identified in the three groups of DEGs. These genes were mainly enriched in Sphingolipid metabolism and PPAR signaling pathway. In the PPI network, we identified seven key genes with higher connectivity, and used qRT-PCR experiments to verify the expressions. In addition, we identified 31 methylation markers and screened 3 key methylation markers (RUNX2, IMMP2L and MDM2) by LASSO model. Their methylation levels were closely related to the diagnostic effects of ASD. Our analysis identified RUNX2, IMMP2L and MDM2 as possible diagnostic markers for ASD. Identifying different biomarkers and risk genes will contribute to the diagnosis of ASD and the development of new clinical and drug treatments.





Similar content being viewed by others
Data availability
The data used in this study can be found in GEO database, including GSE26415, GSE42133, GSE123302 and GSE109905.
References
Amini A, Namvarpour Z, Namvarpour M, Raoofi A (2020) Risperidone accelerates bone loss in rats with autistic-like deficits induced by maternal lipopolysaccharides exposure. Life Sci 258:118197. https://doi.org/10.1016/j.lfs.2020.118197
Arnold S, Foley KR, Hwang YIJ, Richdale AL, Uljarevic M, Lawson LP et al (2019) Cohort profile: the Australian longitudinal study of adults with autism (ALSAA). BMJ Open 9(12):e030798. https://doi.org/10.1136/bmjopen-2019-030798
Barone R, Rizzo R, Tabbi G, Malaguarnera M, Frye RE, Bastin J (2019) Nuclear peroxisome proliferator-activated receptors (PPARs) as therapeutic targets of resveratrol for autism spectrum disorder. Int J Mol Sci 20(8):1878. https://doi.org/10.3390/ijms20081878
Ben-David E, Shifman S (2013) Combined analysis of exome sequencing points toward a major role for transcription regulation during brain development in autism. Mol Psychiatry 18(10):1054–1056. https://doi.org/10.1038/mp.2012.148
Benitez-Burraco A, Murphy E (2016) The oscillopathic nature of language deficits in autism: from genes to language evolution. Front Hum Neurosci 10:120. https://doi.org/10.3389/fnhum.2016.00120
Benitez-Burraco A, Lattanzi W, Murphy E (2016) Language impairments in ASD resulting from a failed domestication of the human brain. Front Neurosci 10:373. https://doi.org/10.3389/fnins.2016.00373
Chater-Diehl E, Ejaz R, Cytrynbaum C, Siu MT, Turinsky A, Choufani S, Goodman SJ, Abdul-Rahman O, Bedford M, Dorrani N, Engleman K, Flores-Daboub J, Genevieve D, Mendoza-Londono R, Meschino W, Perrin L, Safina N, Townshend S, Scherer SW, Anagnostou E, Piton A, Deardorff M, Brudno M, Chitayat D, Weksberg R (2019) New insights into DNA methylation signatures: SMARCA2 variants in Nicolaides-Baraitser syndrome. BMC Med Genet 12(1):105. https://doi.org/10.1186/s12920-019-0555-y
Chow ML, Pramparo T, Winn ME, Barnes CC, Li HR, Weiss L, Fan JB, Murray S, April C, Belinson H, Fu XD, Wynshaw-Boris A, Schork NJ, Courchesne E (2012) Age-dependent brain gene expression and copy number anomalies in autism suggest distinct pathological processes at young versus mature ages. PLoS Genet 8(3):e1002592. https://doi.org/10.1371/journal.pgen.1002592
Courchesne E, Campbell K, Solso S (2011) Brain growth across the life span in autism: age-specific changes in anatomical pathology. Brain Res 1380:138–145. https://doi.org/10.1016/j.brainres.2010.09.101
Dong D, Zielke HR, Yeh D, Yang P (2018) Cellular stress and apoptosis contribute to the pathogenesis of autism spectrum disorder. Autism Res 11(7):1076–1090. https://doi.org/10.1002/aur.1966
Elsabbagh M, Johnson MH (2010) Getting answers from babies about autism. Trends Cogn Sci 14(2):81–87. https://doi.org/10.1016/j.tics.2009.12.005
Fatemi SH, Aldinger KA, Ashwood P, Bauman ML, Blaha CD, Blatt GJ, Chauhan A, Chauhan V, Dager SR, Dickson PE, Estes AM, Goldowitz D, Heck DH, Kemper TL, King BH, Martin LA, Millen KJ, Mittleman G, Mosconi MW, Persico AM, Sweeney JA, Webb SJ, Welsh JP (2012) Consensus paper: pathological role of the cerebellum in autism. Cerebellum 11(3):777–807. https://doi.org/10.1007/s12311-012-0355-9
Fogel BL, Wexler E, Wahnich A, Friedrich T, Vijayendran C, Gao F, Parikshak N, Konopka G, Geschwind DH (2012) RBFOX1 regulates both splicing and transcriptional networks in human neuronal development. Hum Mol Genet 21(19):4171–4186. https://doi.org/10.1093/hmg/dds240
Ghaleiha A, Rasa SM, Nikoo M, Farokhnia M, Mohammadi MR, Akhondzadeh S (2015) A pilot double-blind placebo-controlled trial of pioglitazone as adjunctive treatment to risperidone: effects on aberrant behavior in children with autism. Psychiatry Res 229(1–2):181–187. https://doi.org/10.1016/j.psychres.2015.07.043
Grigg I, Ivashko-Pachima Y, Hait TA, Korenkova V, Touloumi O, Lagoudaki R et al (2020) Tauopathy in the young autistic brain: novel biomarker and therapeutic target. Transl Psychiatry 10(1):228. https://doi.org/10.1038/s41398-020-00904-4
Guang S, Pang N, Deng X, Yang L, He F, Wu L, Chen C, Yin F, Peng J (2018) Synaptopathology involved in autism spectrum disorder. Front Cell Neurosci 12:470. https://doi.org/10.3389/fncel.2018.00470
Guo X, Simas T, Lai MC, Lombardo MV, Chakrabarti B, Ruigrok ANV et al (2019) Enhancement of indirect functional connections with shortest path length in the adult autistic brain. Hum Brain Mapp 40(18):5354–5369. https://doi.org/10.1002/hbm.24777
Keenan RJ, Freymann DM, Stroud RM, Walter P (2001) The signal recognition particle. Annu Rev Biochem 70:755–775. https://doi.org/10.1146/annurev.biochem.70.1.755
Kreilaus F, Chesworth R, Eapen V, Clarke R, Karl T (2019) First behavioural assessment of a novel Immp2l knockdown mouse model with relevance for Gilles de la Tourette syndrome and autism spectrum disorder. Behav Brain Res 374:112057. https://doi.org/10.1016/j.bbr.2019.112057
Li L, Martinez SS, Hu W, Liu Z, Tjian R (2015) A specific E3 ligase/deubiquitinase pair modulates TBP protein levels during muscle differentiation. Elife 4:e08536. https://doi.org/10.7554/eLife.08536
Maestrini E, Pagnamenta AT, Lamb JA, Bacchelli E, Sykes NH, Sousa I et al (2010) High-density SNP association study and copy number variation analysis of the AUTS1 and AUTS5 loci implicate the IMMP2L-DOCK4 gene region in autism susceptibility. Mol Psychiatry 15(9):954–968. https://doi.org/10.1038/mp.2009.34
Muhle R, Trentacoste SV, Rapin I (2004) The genetics of autism. Pediatrics 113(5):e472–e486. https://doi.org/10.1542/peds.113.5.e472
Murphy CM, Wilson CE, Robertson DM, Ecker C, Daly EM, Hammond N et al (2016) Autism spectrum disorder in adults: diagnosis, management, and health services development. Neuropsychiatr Dis Treat 12:1669–1686. https://doi.org/10.2147/NDT.S65455
Naviaux JC, Schuchbauer MA, Li K, Wang L, Risbrough VB, Powell SB, Naviaux RK (2014) Reversal of autism-like behaviors and metabolism in adult mice with single-dose antipurinergic therapy. Transl Psychiatry 4:e400. https://doi.org/10.1038/tp.2014.33
Pan PY, Tammimies K, Bolte S (2020) The association between somatic health, autism Spectrum disorder, and autistic traits. Behav Genet 50(4):233–246. https://doi.org/10.1007/s10519-019-09986-3
Ravarani CNJ, Flock T, Chavali S, Anandapadamanaban M, Babu MM, Balaji S (2020) Molecular determinants underlying functional innovations of TBP and their impact on transcription initiation. Nat Commun 11(1):2384. https://doi.org/10.1038/s41467-020-16182-z
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK (2015) Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43(7):e47. https://doi.org/10.1093/nar/gkv007
Stathopoulos S, Gaujoux R, Lindeque Z, Mahony C, Van Der Colff R, Van Der Westhuizen F, O’Ryan C (2020) DNA methylation associated with mitochondrial dysfunction in a south African autism Spectrum disorder cohort. Autism Res 13(7):1079–1093. https://doi.org/10.1002/aur.2310
Tian Y, Morris TJ, Webster AP, Yang Z, Beck S, Feber A, Teschendorff AE (2017) ChAMP: updated methylation analysis pipeline for Illumina BeadChips. Bioinformatics 33(24):3982–3984. https://doi.org/10.1093/bioinformatics/btx513
Tsai NP, Wilkerson JR, Guo W, Maksimova MA, DeMartino GN, Cowan CW, Huber KM (2012) Multiple autism-linked genes mediate synapse elimination via proteasomal degradation of a synaptic scaffold PSD-95. Cell 151(7):1581–1594. https://doi.org/10.1016/j.cell.2012.11.040
Velmeshev D, Schirmer L, Jung D, Haeussler M, Perez Y, Mayer S, Bhaduri A, Goyal N, Rowitch DH, Kriegstein AR (2019) Single-cell genomics identifies cell type-specific molecular changes in autism. Science 364(6441):685–689. https://doi.org/10.1126/science.aav8130
Wang W, Nag S, Zhang X, Wang MH, Wang H, Zhou J, Zhang R (2015) Ribosomal proteins and human diseases: pathogenesis, molecular mechanisms, and therapeutic implications. Med Res Rev 35(2):225–285. https://doi.org/10.1002/med.21327
Waye MMY, Cheng HY (2018) Genetics and epigenetics of autism: a review. Psychiatry Clin Neurosci 72(4):228–244. https://doi.org/10.1111/pcn.12606
Wong CC, Meaburn EL, Ronald A, Price TS, Jeffries AR, Schalkwyk LC et al (2014) Methylomic analysis of monozygotic twins discordant for autism spectrum disorder and related behavioural traits. Mol Psychiatry 19(4):495–503. https://doi.org/10.1038/mp.2013.41
Yoon SH, Choi J, Lee WJ, Do JT (2020) Genetic and epigenetic etiology underlying autism spectrum disorder. J Clin Med 9(4):966. https://doi.org/10.3390/jcm9040966
Zhang Y, Liu Y, Zarrei M, Tong W, Dong R, Wang Y, Zhang H, Yang X, MacDonald JR, Uddin M, Scherer SW, Gai Z (2018) Association of IMMP2L deletions with autism spectrum disorder: a trio family study and meta-analysis. Am J Med Genet B Neuropsychiatr Genet 177(1):93–100. https://doi.org/10.1002/ajmg.b.32608
Acknowledgements
The authors thank all the participants included in this study.
Funding
This work was not supported by any funds.
Author information
Authors and Affiliations
Contributions
Bei Zhang and Xiaoyuan Hu designed and managed the study. Bei Zhang and Yuefei Li contributed significantly to the data analyses. Xiaoyuan Hu and Yongkang Ni performed methylation analysis. Bei Zhang and Lin Xue wrote the paper. Lin Xue reviewed and edited. All authors contributed to and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics statement
This study was performed at the Fourth People’s Hospital of Urumqi and was approved by the ethics review board of the Fourth People’s Hospital of Urumqi (No: 20200306). Patients gave their approval to be enrolled in this study in an informed written consent. The patient studies were conducted in accordance with the ethical guidelines of the Declaration of Helsinki.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, B., Hu, X., Li, Y. et al. Identification of methylation markers for diagnosis of autism spectrum disorder. Metab Brain Dis 37, 219–228 (2022). https://doi.org/10.1007/s11011-021-00805-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00805-5