Abstract
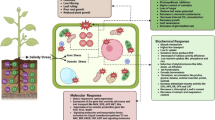
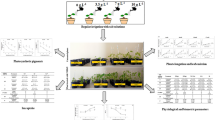
As a novel antioxidant, sodium nitroprusside (SNP) can be used to reduce the adverse effects of various abiotic stresses, especially drought stress. Drought stress is a major problem in the vegetative and reproductive stages of “Mexican lime” (Citrus aurantifolia (Christ.) Swingle), which is known as a main horticultural plant. The aim of the current investigation was to study the impact of SNP on biochemical, morphological, and physiological characteristics of “Mexican lime” under drought stress in vitro condition. This study was performed as a factorial experiment based on a completely randomized design with four replications. Drought stress was induced by using polyethylene glycol (PEG 6000) in four levels (0, 1, 2, and 3%) in Murashige and Skoog (MS) medium. To evaluate the effect of SNP in ameliorating drought stress, various concentrations (0, 25, 50, and 100 μM) of SNP were supplemented to MS medium. The results showed that drought stress led to a reduction in shoot number, shoot length, leaf number, and fresh and dry weight. The application of 25 μM SNP resulted in a favorable impact on these parameters. Also, the highest level of total proline content, electrolyte leakage, and antioxidant enzyme activities were observed under severe drought stress induced by 3% PEG (− 0.3 MPa water potentials).



Similar content being viewed by others
References
Aazami M, Torabi M, Jalili E (2010) In vitro response of promising tomato genotypes for tolerance to osmotic stress. Afr J Biotechnol 9:4014–4017. https://doi.org/10.5897/AJB10.1452
Ali AS, Elozeiri AA (2017) Metabolic processes during seed germination. Adv Seed Biol 3:141–166. https://doi.org/10.5772/intechopen.70653
Al-Jebory EI (2012) Effect of water stress on carbohydrate metabolism during Pisum sativum seedlings growth. Euph J Agric Sci 4:1–12
Amiri E, Shahsavar AR (2010) Response of lime seedlings (Citrus aurantifolia L.) to exogenous spermidine treatments under drought stress. Aust J Basic Appl Sci 4:4483–4489. 10.1.1.1061.941
Antoniou C, Xenofontos R, Chatzimichail G, Christou A, Kashfi K, Fotopoulos V (2020) Exploring the potential of nitric oxide and hydrogen sulfide (NOSH)-releasing synthetic compounds as novel priming agents against drought stress in Medicago sativa plants. Biomolecules 10:120. https://doi.org/10.3390/biom10010120
Arun M, Naing AH, Jeon SM, Ai TN, Aye T, Kim CK (2017) Sodium nitroprusside stimulates growth and shoot regeneration in chrysanthemum. Hortic Environ Biotechnol 58:78–84. https://doi.org/10.1007/s13580-017-0070-z
Bates LS, Waldren RP, Teare I (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bhuyan MB, Hasanuzzaman M, Parvin K, Mohsin SM, Al Mahmud J, Nahar K, Fujita M (2020) Nitric oxide and hydrogen sulfide: two intimate collaborators regulating plant defense against abiotic stress. Plant Growth Regul 90:409–424. https://doi.org/10.1007/s10725-020-00594-4
Boon E, Zimmerman E, St-Arnaud M, Hijri M (2013) Allelic differences within and among sister spores of the arbuscular mycorrhizal fungus Glomus etunicatum suggest segregation at sporulation. PLoS ONE 8:e83301. https://doi.org/10.1007/s11627-013-9526-8
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Cakmak I, Marschner H (1992) Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase, and glutathione reductase in bean leaves. Plant Physiol 98:1222–1227. https://doi.org/10.1104/pp.98.4.1222
Camilios-Neto D, Bonato P, Wassem R, Tadra-Sfeir MZ, Brusamarello-Santos LC, Valdameri G, Donatti L, Faoro H, Weiss VA, Chubatsu LS (2014) Dual RNA-seq transcriptional analysis of wheat roots colonized by Azospirillum brasilense reveals up-regulation of nutrient acquisition and cell cycle genes. BMC Genomics 15:378. https://doi.org/10.1186/1471-2164-15-378
Carimi F, Zottini M, Costa A, Cattelan I, De Michele R, Terzi M, Lo Schiavo F (2005) NO signalling in cytokinin-induced programmed cell death. Plant Cell Environ 28:1171–1178. https://doi.org/10.1111/j.1365-3040.2005.01355.x
Cechin I, Cardoso GS, Fumis TdF, Corniani N (2015) Nitric oxide reduces oxidative damage induced by water stress in sunflower plants. Bragantia 74:200–206. https://doi.org/10.1590/1678-4499.353
Chen D, Wang S, Cao B, Cao D, Leng G, Li H, Deng X (2016) Genotypic variation in growth and physiological response to drought stress and re-watering reveals the critical role of recovery in drought adaptation in maize seedlings. Front Plant Sci 6:1241. https://doi.org/10.3389/fpls.2015.01241
Chun SC, Paramasivan M, Chandrasekaran M (2018) Proline accumulation influenced by osmotic stress in arbuscular mycorrhizal symbiotic plants. Front Microbiol 9:2525. https://doi.org/10.3389/fmicb.2018.02525
Demiral H, Baykul E, Gezer MD, Erkoc S, Engin A, Baykul MC (2014) Preparation and characterization of activated carbon from chestnut shell and its adsorption characteristics for lead. Sep Sci Technol 49:2711–2720. https://doi.org/10.1080/01496395.2014.942742
El-Tayeb M (2005) Response of barley grains to the interactive e. ect of salinity and salicylic acid. Plant Growth Regul 45:215–224. https://doi.org/10.1007/s10725-005-4928-1
Farooq M, Wahid A, Kobayashi N, Fujita D, Basra S (2009) Plant drought stress: effects, mechanisms and management. Sustainable Agric 29:185–212. https://doi.org/10.1051/agro:2008021
Ghadakchiasl A, Mozafari A-a, Ghaderi N (2017) Mitigation by sodium nitroprusside of the effects of salinity on the morpho-physiological and biochemical characteristics of Rubus idaeus under in vitro conditions. Physiol Mol Biol Plants 23:73–83. https://doi.org/10.1007/s12298-016-0396-5
Gharibi S, Tabatabaei BES, Saeidi G, Talebi M, Matkowski A (2019) The effect of drought stress on polyphenolic compounds and expression of flavonoid biosynthesis related genes in Achillea pachycephala Rech. f. Phytochemistry 162:90–98. https://doi.org/10.1016/j.phytochem.2019.03.004
Ghassemi-Golezani K, Farhadi N, Nikpour-Rashidabad N (2018) Responses of in vitro-cultured Allium hirtifolium to exogenous sodium nitroprusside under PEG-imposed drought stress. Plant Cell Tiss Org Cult 133:237–248. https://doi.org/10.1007/s11240-017-1377-2
Guha A, Sengupta D, Rasineni GK, Reddy AR (2012) Non-enzymatic antioxidant defence in drought-stressed mulberry (Morus indica L.) genotypes. Trees 26:903–918. https://doi.org/10.1007/s00468-011-0665-4
Guo Y, Tian Z, Yan D, Zhang J, Qin P (2005) Effects of nitric oxide on salt stress tolerance in Kosteletzkya virginica. Life Sci 6:67–75. https://doi.org/10.15330/jpnu.2.1.122-131
Habib N, Ali Q, Ali S, Javed MT, Zulqurnain Haider M, Perveen R, Bin-Jumah M (2020) Use of nitric oxide and hydrogen peroxide for better yield of wheat (Triticum aestivum L.) under water deficit conditions: growth, osmoregulation, and antioxidative defense mechanism. Plants 9:285. https://doi.org/10.3390/plants9020285
Hameed A, Hameed A, Farooq T, Noreen R, Javed S, Batool S, Ahmad A, Gulzar T, Ahmad M (2019) Evaluation of structurally different benzimidazoles as priming agents, plant defence activators and growth enhancers in wheat. BMC Chemistry 13:29. https://doi.org/10.1186/s13065-019-0546-2
Hao G, Du X, Shi R (2007) Exogenous nitric oxide accelerates soluble sugar, proline and secondary metabolite synthesis in Ginkgo biloba under drought stress. J Plant Physiol Mol Biol 33:499–506
Hayat S, Yadav S, Wani AS, Irfan M, Ahmad A (2011) Nitric oxide effects on photosynthetic rate, growth, and antioxidant activity in tomato. J Veg Sci 17:333–348. https://doi.org/10.1080/19315260.2011.563275
Hemeda HM, Klein B (1990) Effects of naturally occurring antioxidants on peroxidase activity of vegetable extracts. J Food Sci 55:184–185. https://doi.org/10.1111/j.1365-2621.1990.tb06048.x
Hesami M, Daneshvar MH (2018) In vitro adventitious shoot regeneration through direct and indirect organogenesis from seedling-derived hypocotyl segments of Ficus religiosa L.: an important medicinal plant. HortScience 53:55–61. https://doi.org/10.21273/HORTSCI12637-17
Hesami M, Daneshvar MH, Yoosefzadeh-Najafabadi M (2018a) Establishment of a protocol for in vitro seed germination and callus formation of Ficus religiosa L., an important medicinal plant. Jundishapur J Nat Pharm Prod 13:e62682. https://doi.org/10.5812/jjnpp.62682
Hesami M, Daneshvar MH, Yoosefzadeh-Najafabadi M (2019) An efficient in vitro shoot regeneration through direct organogenesis from seedling-derived petiole and leaf segments and acclimatization of Ficus religiosa. J for Res 30:807–815. https://doi.org/10.1007/s11676-018-0647-0
Hesami M, Daneshvar MH, Yoosefzadeh-Najafabadi M, Alizadeh M (2018b) Effect of plant growth regulators on indirect shoot organogenesis of Ficus religiosa through seedling derived petiole segments. J Genet Eng Biotechnol 16:175–180. https://doi.org/10.1016/j.jgeb.2017.11.0
Hesami M, Tohidfar M, Alizadeh M, Daneshvar MH (2020) Effects of sodium nitroprusside on callus browning of Ficus religiosa: an important medicinal plant. J for Res 31:1–8. https://doi.org/10.1007/s11676-018-0860-x
Heuer B (2010) Role of proline in plant response to drought and salinity. Handbook of plant and crop stress. CRC Press, Boca Raton, pp 213-238
Hodges DM, Deiong JM, Forney CF, Prange R (1999) Improving the thiobarbituric acid-reactive–substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611. https://doi.org/10.1007/s004250050524
Hussain HA, Men S, Hussain S, Chen Y, Ali S, Zhang S, Wang L (2019) Interactive effects of drought and heat stresses on morpho-physiological attributes, yield, nutrient uptake and oxidative status in maize hybrids. Sci Rep 9:1–12. https://doi.org/10.1038/s41598-019-40362-7
Jinrong L, Xiaorong X, Jianxiong D, Jixiong S, Xiaomin B (2008) Effects of simultaneous drought and heat stress on Kentucky bluegrass. Sci Hortic 115:190–195. https://doi.org/10.1016/j.scienta.2007.08.003
Kalra C, Babbar SB (2010) Nitric oxide promotes in vitro organogenesis in Linum usitatissimum L. Plant Cell Tiss Org Cult 103:353–359. https://doi.org/10.1007/s11240-010-9788-3
Khan MN, Mobin M, Abbas ZK, Siddiqui MH (2017) Nitric oxide-induced synthesis of hydrogen sulfide alleviates osmotic stress in wheat seedlings through sustaining antioxidant enzymes, osmolyte accumulation and cysteine homeostasis. Nitric Oxide 68:91–102. https://doi.org/10.1016/j.niox.2017.01.001
Kielkowska A, Adamus A, Oleksyk A (2012) In vitro selection of Allium cepa for water stress. Acta Hortic 969:117–120
Lai J, Li R, Xu X, Jin W, Xu M, Zhao H, Xiang Z, Song W, Ying K, Zhang M (2010) Genome-wide patterns of genetic variation among elite maize inbred lines. Nat Genet 42:1027. https://doi.org/10.1038/ng.684
Lee B-R, Islam MT, Park S-H, Jung H-i, Bae D-W, Kim T-HJE (2019) Characterization of salicylic acid-mediated modulation of the drought stress responses: reactive oxygen species, proline, and redox state in Brassica napus. Environ Exp Bot 157:1–10. https://doi.org/10.1016/j.envexpbot.2018.09.013
Lei Y, Yin C, Li C (2007) Adaptive responses of Populus przewalskii to drought stress and SNP application. Acta Physiol Plant 29:519–526. https://doi.org/10.1007/s11738-007-0062-1
Liao W-B, Huang G-B, Yu J-H, Zhang M-L (2012) Nitric oxide and hydrogen peroxide alleviate drought stress in marigold explants and promote its adventitious root development. Plant Physiol Biochem 58:6–15. https://doi.org/10.1016/j.plaphy.2012.06.012
Lichtenthaler HK, Buschmann C (2001) Chlorophylls and carotenoids: measurement and characterization by UV-VIS spectroscopy. Curr Protoc Food Chem 1:F4–F3. https://doi.org/10.1002/0471142913.faf0403s01
Metwali EMR, Soliman HIA, Howladar SM, Fuller MP, Al-Zahrani HS (2016) Appraisal of in vitro drought stress among three different cultivars of fig (Ficus carica L.) using RAPD and ISSR markers. Plant Omics 9:1–11. https://doi.org/10.3316/887026726696681
Mibei EK, Ambuko J, Giovannoni JJ, Onyango AN, Owino WO (2017) Carotenoid profiling of the leaves of selected African eggplant accessions subjected to drought stress. Food Sci Nutr 5:113–122. https://doi.org/10.1002/fsn3.370
Mohammadi H, Esmailpour M, Gheranpaye A (2016) Effects of TiO2 nanoparticles and water-deficit stress on morpho-physiological characteristics of dragonhead (Dracocephalum moldavica L.) plants. Acta Agric Slov 107:385–396. https://doi.org/10.14720/aas.2016.107.2.11
Mozafari A, Havas F, Ghaderi N (2018) Application of iron nanoparticles and salicylic acid in in vitro culture of strawberries (Fragaria× ananassa Duch.) to cope with drought stress. Plant Cell Tiss Org Cult 132:511–523. https://doi.org/10.1007/s11240-017-1347-8
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Murata N, Takahashi S, Nishiyama Y, Allakhverdiev SI (2007) Photoinhibition of photosystem II under environmental stress. Biochim Biophys Acta Rev Bioenerg 1767:414–421. https://doi.org/10.1016/j.bbabio.2006.11.019
Murray MB, Cape JN, Fowler D (1989) Quantification of frost damage in plant tissues by rates of electrolyte leakage. New Phytol 113:307–311. https://doi.org/10.1111/j.14698137.1989.tb02408.x
Nazar R, Umar S, Khan N, Sareer O (2015) Salicylic acid supplementation improves photosynthesis and growth in mustard through changes in proline accumulation and ethylene formation under drought stress. S Afr J Bot 98:84–94. https://doi.org/10.1016/j.sajb.2015.02.005
Neill SJ, Desikan R, Hancock JT (2003) Nitric oxide signaling in plants. New Phytol 159:11–35. https://doi.org/10.1046/j.1469-8137.2003.00804.x
Ozden M, Demirel U, Kahraman A (2009) Effects of proline on antioxidant system in leaves of grapevine (Vitis vinifera L.) exposed to oxidative stress by H2O2. Sci Hortic 119:163–168. https://doi.org/10.1016/j.scienta.2008.07.031
Pedersen A, Feldner H, Rosendahl L (1996) Effect of proline on nitrogenase activity in symbiosomes from root nodules of soybean (Glycine max L.) subjected to drought stress. J Exp Bot 47:1533–1539. https://doi.org/10.1093/jxb/47.10.1533
Pradhan N, Singh P, Dwivedi P, Pandey DK (2020) Evaluation of sodium nitroprusside and putrescine on polyethylene glycol induced drought stress in Stevia rebaudiana Bertoni under in vitro condition. Ind Crops Prod 154:112754. https://doi.org/10.1016/j.indcrop.2020.112754
Rao S, Jabeen F (2013) In vitro selection and characterization of polyethylene glycol (PEG) tolerant callus lines and regeneration of plantlets from the selected callus lines in sugarcane (Saccharum officinarum L.). Physiol Mol Biol Plants 19:261–268. https://doi.org/10.1007/s12298-013-0162-x
Rezayian M, Ebrahimzadeh H, Niknam V (2020) Nitric oxide stimulates antioxidant system and osmotic adjustment in soybean under drought stress. J Soil Sci Plant Nutr 16:1–11. 10.1007%2Fs42729–020–00198-x
Ruan H, Shen W, Ye M, Xu L (2002) Protective effects of nitric oxide on salt stress-induced oxidative damage to wheat (Triticum aestivum L.) leaves. Chin Sci Bull 47:677–682. https://doi.org/10.1360/02tb9154
Saed-Moocheshi A, Shekoofa A, Sadeghi H, Pessarakli M (2014) Drought and salt stress mitigation by seed priming with KNO3 and urea in various maize hybrids: an experimental approach based on enhancing antioxidant responses. J Plant Nutr 37:674–689. https://doi.org/10.1080/01904167.2013.868477
Said EM, Mahmoud RA, Al-Akshar R, Safwat G (2015) Drought stress tolerance and enhancement of banana plantlets in vitro. Aus J Biotech Bioengin 2:1040–1046
Santisree P, Bhatnagar-Mathur P, Sharma KK (2015) NO to drought-multifunctional role of nitric oxide in plant drought: do we have all the answers? Plant Sci 239:44–55. https://doi.org/10.1016/j.plantsci.2015.07.012
Sarropoulou V, Dimassi-Theriou K, Therios I (2014) Ιn vitro plant regeneration from leaf explants of the cherry rootstocks CAB-6P, Gisela 6, and MxM 14 using sodium nitroprusside. In Vitro Cell Dev Biol - Plant 50:226-234. https://doi.org/10.1007/s11627-013-9565-1
Sarropoulou V, Maloupa E (2017) Effect of the NO donor “sodium nitroprusside”(SNP), the ethylene inhibitor “cobalt chloride”(CoCl2) and the antioxidant vitamin E “α-tocopherol” on in vitro shoot proliferation of Sideritis raeseri Boiss. & Heldr. subsp. raeseri. Plant Cell Tiss Org Cult 128:619–629. https://doi.org/10.1007/s11240-016-1139-6
SAS (2001). STAT User’s Guide for Personal Computers, Release 6.12. SAS Institute Inc. Cary, NC, USA.
Shallan MA, Hassan HM, Namich AA, Ibrahim AA (2012) Effect of sodium nitroprusside, putrescine and glycine betaine on alleviation of drought stress in cotton plant. Am Eurasian J Agric Environ Sci 12:1252–1265. https://doi.org/10.5829/idosi.aejaes.2012.12.09.1902
Sheokand S, Bhankar V, Sawhney V (2010) Ameliorative effect of exogenous nitric oxide on oxidative metabolism in NaCl treated chickpea plants. Braz J Plant Physiol 22:81–90. https://doi.org/10.1590/S1677-04202010000200002
Shi Q, Ding F, Wang X, Wei M (2007) Exogenous nitric oxide protect cucumber roots against oxidative stress induced by salt stress. Plant Physiol Biochem 45:542–550. https://doi.org/10.1016/j.plaphy.2007.05.005
Siddiqui MH, Alamri S, Alsubaie QD, Ali HM, Khan MN, Al-Ghamdi A, Alsadon A (2020) Exogenous nitric oxide alleviates sulfur deficiency-induced oxidative damage in tomato seedlings. Nitric Oxide 94:95–107. https://doi.org/10.1016/j.niox.2019.11.002
Silva K S, Tabaldi LA, Rossato LV, Cavichioli BM, Basilio VB, Machado SLO (2019) Contents of pigments and activity of antioxidant enzymes in rice plants pre-treated with sodium nitroprusside and exposed to clomazone. Planta Daninha 37. https://doi.org/10.1590/s0100-83582019370100032
Silveira NM, Marcos FC, Frungillo L, Moura BB, Seabra AB, Salgado I, Machado EC, Hancock JT, Ribeiro RV (2017) S-nitrosoglutathione spraying improves stomatal conductance, Rubisco activity and antioxidant defense in both leaves and roots of sugarcane plants under water deficit. Physiol Plant 160:383–395. https://doi.org/10.1111/ppl.12575
Silvestri C, Celletti S, Cristofori V, Astolfi S, Ruggiero B, Rugini E (2017) Olive (Olea europaea L.) plants transgenic for tobacco osmotin gene are less sensitive to in vitro-induced drought stress. Acta Physiol Plant 39:229–238. https://doi.org/10.1007/s11738-017-2535-1
Simsek O (2018) Effect of drought stress in in vitro and drought-related gene expression in Carrizo citrange. Fresenius Environ Bull 27:9167–9171
Sofo A, Scopa A, Nuzzaci M, Vitti A (2015) Ascorbate peroxidase and catalase activities and their genetic regulation in plants subjected to drought and salinity stresses. Int J Mol Sci 16:13561–13578. https://doi.org/10.3390/ijms160613561
Sohag A A M, Tahjib-Ul-Arif M, Brestic M, Afrin S, Sakil M A, Hossain M T, Hossain M A (2020) Exogenous salicylic acid and hydrogen peroxide attenuate drought stress in rice. Plant Soil Environ 66:7–13. https://doi.org/10.17221/472/2019-PSE
Soliman M, Alhaithloul HA, Hakeem KR, Alharbi BM, El-Esawi M, Elkelish A (2019) Exogenous nitric oxide mitigates nickel-induced oxidative damage in eggplant by upregulating antioxidants, osmolyte metabolism, and glyoxalase systems. Plants 8:562. https://doi.org/10.3390/plants8120562
Sun H, Jiao L, Song W, Tao J, Huang S, Si C (2015) Nitric oxide generated by nitrate reductase increases nitrogen uptake capacity by inducing lateral root formation and inorganic nitrogen uptake under partial nitrate nutrition in rice. J Exp Bot 66:2449–2459. https://doi.org/10.1093/jxb/erv030
Takahashi S, Badger MR (2011) Photoprotection in plants: a new light on photosystem II damage. Trends Plant Sci 16:53–60. https://doi.org/10.1016/j.tplants.2010.10.001
Tan J, Zhao H, Hong J, Han Y, Li H, Zhao W (2008) Effects of exogenous nitric oxide on photosynthesis, antioxidant capacity and proline accumulation in wheat seedlings subjected to osmotic stress. World J Agric Sci 4:307–313. https://doi.org/10.1007/s11627-013-9526-8
Tanou G, Molassiotis A, Diamantidis G (2009) Hydrogen peroxide-and nitric oxide-induced systemic antioxidant prime-like activity under NaCl-stress and stress-free conditions in citrus plants. J Plant Physiol 166:1904–1913. https://doi.org/10.1016/j.jplph.2009.06.012
Tun NN, Holk A, Scherer GF (2001) Rapid increase of NO release in plant cell cultures induced by cytokinin. FEBS Lett 509:174–176. https://doi.org/10.1016/S0014-5793(01)03164-7
Uchida A, Jagendorf AT, Hibino T, Takabe T, Takabe T (2002) Effects of hydrogen peroxide and nitric oxide on both salt and heat stress tolerance in rice. Plant Sci 163:515–523. https://doi.org/10.1016/S0168-9452(02)00159-0
Vand SH, Abdullah TL (2012) Identification and introduction of thornless lime (Citrus aurantifolia) in Hormozgan. Iran Indian J Sci Technol 5:3670–3673
Verma A, Malik C, Gupta V (2014) Sodium nitroprusside-mediated modulation of growth and antioxidant defense in the in vitro raised plantlets of peanut genotypes. Peanut Sci 41:25–31. https://doi.org/10.3146/PS12-13.1
Wu R, Wang T, Richardson AC, Allan AC, Macknight RC, Varkonyi-Gasic E (2019) Histone modification and activation by SOC1-like and drought stress-related transcription factors may regulate AcSVP2 expression during kiwifruit winter dormancy. Plant Sci 281:242–250. https://doi.org/10.1016/j.plantsci.2018.12.001
Wu S, Hu C, Tan Q, Lu L, Shi K, Yong Z (2015a) Drought stress tolerance mediated by zinc-induced antioxidative defense and osmotic adjustment in cotton (Gossypium hirsutum). Acta Physiol Plant 37:167. https://doi.org/10.1007/s11738-015-1919-3
Wu S, Hu C, Tan Q, Nie Z, Sun X (2014) Effects of molybdenum on water utilization, antioxidative defense system and osmotic-adjustment ability in winter wheat (Triticum aestivum) under drought stress. Plant Physiol Biochem 83:365–374. https://doi.org/10.1016/j.plaphy.2014.08.022
Wu X, Zhu W, Zhang H, Ding H, Zhang HJ (2011) Exogenous nitric oxide protects against salt-induced oxidative stress in the leaves from two genotypes of tomato (Lycopersicom esculentum Mill.). Acta Physiol Plant 33:1199–1209. https://doi.org/10.1007/s11738-010-0648-x
Wu Z, Zhao X, Sun X, Tan Q, Tang Y, Nie Z (2015b) Antioxidant enzyme systems and the ascorbate–glutathione cycle as contributing factors to cadmium accumulation and tolerance in two oilseed rape cultivars (Brassica napus L.) under moderate cadmium stress. Chemosphere 138:526–536. https://doi.org/10.1016/j.chemosphere.2015.06.080
Xu J, Yin H, Wang W, Mi Q, Liu X (2009) Effects of sodium nitroprusside on callus induction and shoot regeneration in micropropagated Dioscorea opposita. Plant Growth Regul 59:279. https://doi.org/10.1007/s10725-009-9410-z
Yan F, Liu Y, Sheng H, Wang Y, Kang H, Zeng J (2016) The salicylic acid and nitric oxide increase photosynthesis and antioxidant defense in wheat under UV-B stress. Biol Plant 60:686–694. https://doi.org/10.1007/s10535-016-0622-6
Zeng C-L, Liu L, Wang B-R, Wu X-M, Zhou Y (2011) Physiological effects of exogenous nitric oxide on Brassica juncea seedlings under NaCl stress. Biol Plant 55:345–348. https://doi.org/10.1007/s10535-011-0051-5
Zhang A, Zhang J, Zhang J, Ye N, Zhang H, Tan M (2011) Nitric oxide mediates brassinosteroid-induced ABA biosynthesis involved in oxidative stress tolerance in maize leaves. Plant Cell Physiol 52:181–192. https://doi.org/10.1093/pcp/pcq187
Zhang LP, Mehta SK, Liu ZP, Yang ZM (2008) Copper-induced proline synthesis is associated with nitric oxide generation in Chlamydomonas reinhardtii. Plant Cell Physiol 49:411–419. https://doi.org/10.1093/pcp/pcn017.PMid:182527
Acknowledgements
The authors would like to thank the College of Agriculture, Shiraz University for their support, cooperation, and assistance throughout this research.
Author information
Authors and Affiliations
Contributions
Marziyeh Jafari: analysis and interpretation of data, methodology, data curation, writing — original draft; Alireza Shahsavar: conceptualization, methodology, project administration, supervision, validation, writing — review and editing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Jafari, M., Shahsavar, A.R. Sodium nitroprusside: its beneficial role in drought stress tolerance of “Mexican lime” (Citrus aurantifolia (Christ.) Swingle) under in vitro conditions. In Vitro Cell.Dev.Biol.-Plant 58, 155–168 (2022). https://doi.org/10.1007/s11627-021-10218-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-021-10218-9