Abstract
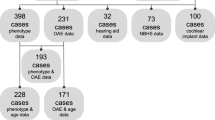
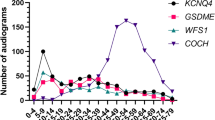
Pathogenic variations in the OTOF gene are a common cause of hearing loss. To refine the natural history and genotype–phenotype correlations of OTOF-related auditory neuropathy spectrum disorders (ANSD), audiograms and distortion product otoacoustic emissions (DPOAEs) were collected from a diverse cohort of individuals diagnosed with OTOF-related ANSD by comprehensive genetic testing and also reported in the literature. Comparative analysis was undertaken to define genotype–phenotype relationships using a Monte Carlo algorithm. 67 audiograms and 25 DPOAEs from 49 unique individuals positive for OTOF-related ANSD were collected. 51 unique OTOF pathogenic variants were identified of which 21 were missense and 30 were loss of function (LoF; nonsense, splice-site, copy number variants, and indels). There was a statistically significant difference in low, middle, and high frequency hearing thresholds between missense/missense and LoF/missense genotypes as compared to LoF/LoF genotypes (average hearing threshold for low, middle and high frequencies 70.9, 76.0, and 73.4 dB vs 88.5, 95.6, and 94.7 dB) via Tukey’s test with age as a co-variate (P = 0.0180, 0.0327, and 0.0347, respectively). Hearing declined during adolescence with missense/missense and LoF/missense genotypes, with an annual mid-frequency threshold deterioration of 0.87 dB/year and 1.87 dB/year, respectively. 8.5% of frequencies measured via DPOAE were lost per year in individuals with serial tests. Audioprofiling of OTOF-related ANSD suggests significantly worse hearing with LoF/LoF genotypes. The unique pattern of variably progressive OTOF-related autosomal recessive ANSD may be amenable to gene therapy in selected clinical scenarios.




Similar content being viewed by others
Data availability
Data supporting this study’s findings are available within the article and its supplementary materials.
References
Adato A et al (2000) Deafness heterogeneity in a Druze isolate from the Middle East: novel OTOF and PDS mutations, low prevalence of GJB2 35delG mutation and indication for a new DFNB locus. Eur J Hum Genet EJHG 8(6):437–442. https://doi.org/10.1038/sj.ejhg.5200489
Azaiez H et al (2004) GJB2: The spectrum of deafness-causing allele variants and their phenotype. Hum Mutat 24(4):305–311. https://doi.org/10.1002/humu.20084
Azaiez H et al (2018) Genomic landscape and mutational signatures of deafness-associated genes. Am J Hum Genet 103(4):484–497. https://doi.org/10.1016/j.ajhg.2018.08.006
Azaiez H, Thorpe RK, Smith RJ (2021) OTOF-related deafness. In: Adam MP et al (eds) GeneReviews®. University of Washington, Seattle, WA
Booth KT, Azaiez H, Smith RJH (2020) DFNA5 (GSDME) c.991-15_991-13delTTC: founder mutation or mutational hotspot? Int J Mol Sci 21(11):3951. https://doi.org/10.3390/ijms21113951
Butler BE, Lomber SG (2013) Functional and structural changes throughout the auditory system following congenital and early-onset deafness: implications for hearing restoration. Front Syst Neurosci. https://doi.org/10.3389/fnsys.2013.00092
Chiu Y-H et al (2010) Mutations in the OTOF gene in Taiwanese patients with auditory neuropathy. Audiol Neurotol 15(6):364–374. https://doi.org/10.1159/000293992
Choi BY et al (2009) Identities and frequencies of mutations of the otoferlin gene (OTOF) causing DFNB9 deafness in Pakistan. Clin Genet 75(3):237–243. https://doi.org/10.1111/j.1399-0004.2008.01128.x
Hams N et al (2017) Otoferlin is a multivalent calcium-sensitive scaffold linking SNAREs and calcium channels. Proc Natl Acad Sci 114(30):8023–8028. https://doi.org/10.1073/pnas.1703240114
Iwasa Y et al (2013) OTOF mutation screening in Japanese severe to profound recessive hearing loss patients. BMC Med Genet 14:95. https://doi.org/10.1186/1471-2350-14-95
Jin YJ et al (2014) Identification of a novel splice site variant of OTOF in the Korean nonsyndromic hearing loss population with low prevalence of the OTOF mutations. Int J Pediatr Otorhinolaryngol 78(7):1030–1035. https://doi.org/10.1016/j.ijporl.2014.03.033
Karczewski KJ et al (2020) The mutational constraint spectrum quantified from variation in 141,456 humans. Nature 581(7809):434–443. https://doi.org/10.1038/s41586-020-2308-7
Kitao K et al (2019) Deterioration in distortion product otoacoustic emissions in auditory neuropathy patients with distinct clinical and genetic backgrounds. Ear Hear 40(1):184–191. https://doi.org/10.1097/AUD.0000000000000586
Mahdieh N et al (2012) Screening of OTOF mutations in Iran: a novel mutation and review. Int J Pediatr Otorhinolaryngol 76(11):1610–1615. https://doi.org/10.1016/j.ijporl.2012.07.030
Matsunaga T et al (2012) A prevalent founder mutation and genotype-phenotype correlations of OTOF in Japanese patients with auditory neuropathy. Clin Genet 82(5):425–432. https://doi.org/10.1111/j.1399-0004.2012.01897.x
Michalski N et al (2017) Otoferlin acts as a Ca2+ sensor for vesicle fusion and vesicle pool replenishment at auditory hair cell ribbon synapses. eLife 6:e31013. https://doi.org/10.7554/eLife.31013
Migliosi V et al (2002) Q829X, a novel mutation in the gene encoding otoferlin (OTOF), is frequently found in Spanish patients with prelingual non-syndromic hearing loss. J Med Genet 39(7):502–506. https://doi.org/10.1136/jmg.39.7.502
Omichi R et al (2019) Gene therapy for hearing loss. Hum Mol Genet 28(R1):R65–R79. https://doi.org/10.1093/hmg/ddz129
Rodríguez-Ballesteros M et al (2003) Auditory neuropathy in patients carrying mutations in the otoferlin gene (OTOF). Hum Mutat 22(6):451–456. https://doi.org/10.1002/humu.10274
Rodríguez-Ballesteros M et al (2008) A multicenter study on the prevalence and spectrum of mutations in the otoferlin gene (OTOF) in subjects with nonsyndromic hearing impairment and auditory neuropathy. Hum Mutat 29(6):823–831. https://doi.org/10.1002/humu.20708
Romanos J et al (2009) Novel OTOF mutations in Brazilian patients with auditory neuropathy. J Hum Genet 54(7):382–385. https://doi.org/10.1038/jhg.2009.45
Rouillon I et al (2006) Results of cochlear implantation in two children with mutations in the OTOF gene. Int J Pediatr Otorhinolaryngol 70(4):689–696. https://doi.org/10.1016/j.ijporl.2005.09.006
Santarelli R et al (2009) Abnormal cochlear potentials from deaf patients with mutations in the otoferlin gene. J Assoc Res Otolaryngol JARO 10(4):545–556. https://doi.org/10.1007/s10162-009-0181-z
Schmuziger N, Probst R, Smurzynski J (2004) Test-retest reliability of pure-tone thresholds from 0.5 to 16 kHz using sennheiser HDA 200 and etymotic research ER-2 earphones. Ear Hear 25(2):127–132. https://doi.org/10.1097/01.AUD.0000120361.87401.C8
Shearer AE, Hansen MR (2019) Auditory synaptopathy, auditory neuropathy, and cochlear implantation. Laryngosc Investig Otolaryngol 4(4):429–440. https://doi.org/10.1002/lio2.288
Smith RJ, Bale JF, White KR (2005) Sensorineural hearing loss in children. Lancet 365(9462):879–890. https://doi.org/10.1016/S0140-6736(05)71047-3
Snoeckx RL et al (2005) GJB2 Mutations and degree of hearing loss: a multicenter study. Am J Hum Genet 77(6):945–957. https://doi.org/10.1086/497996
Strenzke N et al (2016) Hair cell synaptic dysfunction, auditory fatigue and thermal sensitivity in otoferlin Ile515Thr mutants. EMBO J 35(23):2519–2535. https://doi.org/10.15252/embj.201694564
Tang H-Y et al (2015) DNA sequence analysis and genotype-phenotype assessment in 71 patients with syndromic hearing loss or auditory neuropathy. BMJ Open 5(5):e007506. https://doi.org/10.1136/bmjopen-2014-007506
Taylor KR et al (2013) Audiogene: predicting hearing loss genotypes from phenotypes to guide genetic screening. Hum Mutat 34(4):539–545. https://doi.org/10.1002/humu.22268
Teagle HFB et al (2010) Cochlear implantation in children with auditory neuropathy spectrum disorder. Ear Hear 31(3):325–335. https://doi.org/10.1097/AUD.0b013e3181ce693b
Valente M, Valente M, Goebel J (1992) High-frequency thresholds: circumaural earphone versus insert earphone. J Am Acad Audiol 3(6):410–418
Varga R et al (2003) Non-syndromic recessive auditory neuropathy is the result of mutations in the otoferlin (OTOF) gene. J Med Genet 40(1):45–50. https://doi.org/10.1136/jmg.40.1.45
Varga R et al (2006) OTOF mutations revealed by genetic analysis of hearing loss families including a potential temperature sensitive auditory neuropathy allele. J Med Genet 43(7):576–581. https://doi.org/10.1136/jmg.2005.038612
Virtanen P et al (2020) SciPy 1.0: fundamental algorithms for scientific computing in Python. Nat Methods 17(3):261–272. https://doi.org/10.1038/s41592-019-0686-2
Walls WD et al (2020) A comparative analysis of genetic hearing loss phenotypes in European/American and Japanese populations. Hum Genet 139(10):1315–1323. https://doi.org/10.1007/s00439-020-02174-y
Webb B et al (2016) Identification of a novel pathogenic OTOF variant causative of nonsyndromic hearing loss with high frequency in the Ashkenazi Jewish population. Appl Clin Genet 9:141–146. https://doi.org/10.2147/TACG.S113828
Yasunaga S et al (1999) A mutation in OTOF, encoding otoferlin, a FER-1-like protein, causes DFNB9, a nonsyndromic form of deafness. Nat Genet 21(4):363–369. https://doi.org/10.1038/7693
Yasunaga S et al (2000) OTOF encodes multiple long and short isoforms: genetic evidence that the long ones underlie recessive deafness DFNB9. Am J Hum Genet 67(3):591–600. https://doi.org/10.1086/303049
Yildirim-Baylan M et al (2014) Evidence for genotype-phenotype correlation for OTOF mutations. Int J Pediatr Otorhinolaryngol 78(6):950–953. https://doi.org/10.1016/j.ijporl.2014.03.022
Zadro C et al (2010) Five new OTOF gene mutations and auditory neuropathy. Int J Pediatr Otorhinolaryngol 74(5):494–498. https://doi.org/10.1016/j.ijporl.2010.02.004
Zheng D, Liu X (2020) cochlear implantation outcomes in patients with OTOF mutations. Front Neurosci. https://doi.org/10.3389/fnins.2020.00447
Acknowledgements
This project was supported in part by NIH-NIDCD grant R01′s DC002842, DC012049, and DC017955 (RJHS) and NIH-NIDCD grant 5T32DC000040 (RKT).
Funding
This project was supported in part by NIH-NIDCD grant R01's DC002842, DC012049, and DC017955 (RJHS) and NIH-NIDCD grant 5T32DC000040 (RKT).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethics approval
Clinical data were collected as approved by the University of Iowa Institutional Review Board (IRB), project identification number 201602772.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Thorpe, R.K., Azaiez, H., Wu, P. et al. The natural history of OTOF-related auditory neuropathy spectrum disorders: a multicenter study. Hum Genet 141, 853–863 (2022). https://doi.org/10.1007/s00439-021-02340-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-021-02340-w