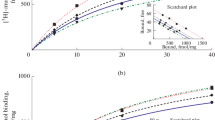
The effects of chronic intranasal administration of cycloprolylglycine (CPG, 1 and 2 mg/kg) on the behavior of BALB/c and C57BL/6 mice and the brain neuroreceptor characteristics in BALB/c mice were studied. Intranasal administration of CPG (1 and 2 mg/kg, 2 weeks) to BALB/c mice in the Porsolt test resulted in a significant antidepressant-like effect more potent than that from intraperitoneal administration of CPG. At the same time, intranasal administration of CPG to C57BL/6 mice in the tail-suspension test was the same as that upon intraperitoneal administration and led to a moderate antidepressant-like effect (1 mg/kg, 4 weeks). Intranasal CPG did not produce any effect on anxiety or exploratory activity in the closed cross-maze test. Radioligand analyses showed an increased density of GABAA receptors in the PFC (1 mg/kg) and reduced density of NMDAand 5HT2A receptors in the hippocampus and striatum, respectively, after intranasal administration of CPG (1 and 2 mg/kg) for two weeks while not affecting mGluII receptor binding in the hippocampus. The significant difference in hippocampal NMDA binding between the two routes may indicate that this receptor is involved in the mechanism of the antidepressant-like effect of CPG.



Similar content being viewed by others
References
T. A. Gudasheva, S. S. Boyko, V. Kh. Akparov, et al., FEBS Lett., 391, 149 – 152 (1996).
J. Guan, P. Harris, M. Brimble, et al., Expert Opin. Ther. Targets, 19(6), 785 – 793 (2015).
T. A. Gudasheva, M. A. Konstantinopol’skii, R. U. Ostrovskaya, et al., Byull. Eksp. Biol. Med., 131(5), 547 – 550 (2001).
T. A. Gudasheva, R. U. Ostrovskaya, S. S. Trofimov, et al., Byull. Eksp. Biol. Med., 128(10), 411 – 413 (1999).
K. N. Kolyasnikova, T. A. Gudasheva, G. A. Nazarova, et al., Eksp. Klin. Farmakol., 75(9), 3 – 6 (2012).
T. A. Gudasheva, V. V. Grigor’ev, K. N. Kolyasnikova, et al., Dokl. Akad. Nauk, 471(1), 106 – 108 (2016).
T. A. Gudasheva, K. N. Kolyasnikova, T. A. Antipova, et al., Dokl. Akad. Nauk, 469(4), 492 – 495 (2016).
E. V. Vasil’eva, A. A. Abdullina, E. A. Kondrakhin, et al., Eur. Neuropsychopharmacol., 29(S1), s511-s512 (2019).
E. V. Vasil’eva, R. M. Salmov, and G. I. Kovalev, Farmakokinet. Farmakodin., No. 2, 31 – 36 (2016).
G. I. Kovalev, E. V. Vasil’eva, and R. M. Salimov, Zh. Vyssh. Nervn. Deyat. im. I. P. Pavlova, 69(1), 123 – 130 (2019).
A. N. Mironov, Handbook for Preclinical Drug Trials [in Russian], Part 1, Grif i K, Moscow (2012).
R. M. Salimov, Zh. Vyssh. Nervn. Deyat. im. I. P. Pavlova, 38(3), 569 – 571 (1988).
N. G. Bowery, D. R. Hill, and A. L. Hudson, Neuropharmacology, 24(3), 207 – 210 (1985).
J. E. Hawkinson, M. Acosta-Burruel, C. L. Kimbrought, et al., Eur. J. Pharmacol., 304, 141 – 146 (1996).
Y. Ito, D. Koo Lim, Y. Hayase, et al., Neurochem. Res., 4, 307 – 313 (1992).
K. T. LaPage, J. E. Ishmael, C. W. Low, et al., Neuropharmacology, 49, 1 – 16 (2005).
J. E. Leysen and C. J. Niemegeers, Mol. Pharmacol., 21(2), 301 – 314 (1982).
H. Schaffhauser, J. G. Richards, J. Cartmell, et al., Mol. Pharmacol., 53, 228 – 233 (1998).
A. M. Szekely, M. L. Barbaccia, and E. Costa, J. Pharmacol. Exp. Ther., 243(1), 155 – 159 (1987).
L. M. Zhou, Z. Q. Gu, A. M. Costa, et al., J. Pharmacol. Exp. Ther., 280(1), 422 – 427 (1997).
I. G. Kovalev, E. V. Vasil’eva, E. A. Kondrakhin, et al., Neirokhimiya, 4, 1 – 10 (2017).
Yu. Yu. Firstova, E. V. Vasil’eva, and G. I. Kovalev, Farmakokinet. Farmakodin., No. 3, 42 – 47 (2018).
E. V. Vasil’eva, R. M. Salimov, and G. I. Kovalev, Eksp. Klin. Farmakol., 75(7), 32 – 37 (2012).
E. V. Vasil’eva, A. A. Abdullina, and G. I. Kovalev, in: Proceedings of the Russian Scientific Conference “Pharmacology of Regulatory Neuropeptides” [in Russian], St. Petersburg (2017), pp. 10 – 11.
H. Shoji and T. Miyakawa, Mol. Brain, 12, 70 (2019).
S. Farley, S. Dumas, S. El Mestikawy, et al., Neuropharmacology, 62(1), 503 – 517 (2012).
H. Ito, M. Nagano, H. Suzuki, et al., Neuropharmacology, 58, 746 – 757 (2010).
G. I. Kovalev, E. A. Kondrakhin, and R. M. Salimov, Neirokhimiya, 30(2), 128 – 134 (2013).
E. V. Vasil’eva, Author’s Abstract of a Candidate Dissertation in Biological Sciences, Moscow (2013).
C. A. Browne, G. Clarke, T. G. Dinan, et al., Neuropharmacology, 60(4), 683 – 691 (2011).
C. M. O’Mahony, F. F. Sweeney, E. Daly, et al., Behav. Brain Res., 213(2), 148 – 154 (2010).
A. M. Privalova, T. V. Bukreeva, and N. V. Gulyaeva, Neurochem. J., 6(2), 77 – 88 (2012).
S. S. Boiko, K. N. Kolyasnikova, and V. P. Zherdev, Farmakokinet. Farmakodin., No. 3, 34 – 38 (2019).
A. A. Abdullina, E. V. Vasileva, E. A. Kondrakhin, et al., Neurochem. J., 13, 249 – 255 (2019).
F. Radja, L. Descarries, K. M. Dewar, et al., Brain Res., 606(2), 273 – 285 (1993).
S. Numan, K. H. Lundgren, D. E. Wright, et al., Mol. Brain Res., 29, 391 – 396 (1995).
A. A. Abdullina, E. V. Vasil’eva, V. S. Kudrin, et al., Farmakokinet. Farmakodin., No. 1, 3 – 10 (2020).
G. Sanacora, C. A. Zarate, and J. H. Krystal, Nat. Rev. Drug Discovery, 7, 426 – 437 (2008).
G. Sanacora, G. Treccani, and M. Popoli, Neuropharmacology, 62, 63 – 77 (2011).
L. Musazzi, G. Racagni, and M. Popoli, Neurochem. Int., 59(2), 138 – 149 (2011).
F. Calabrese, G. Guidotti, and R. Molteni, PLoS One, 7(5), e37916 (2012).
P. Skolnick, P. Popik, and R. Trullas, Trends Pharmacol. Sci., 30, 563 – 569 (2009).
G. Nowak, R. Trullas, R. T. Layer, et al., J. Pharmacol. Exp. Ther., 265, 1380 – 1386 (1993).
B. Bobula, K. Tokarski, and G. Hess, Neuroscience, 120, 765 – 769 (2003).
S. Ghosal, B. Hare, and R. S. Duman, Curr. Opin. Behav. Sci., 14, 1 – 8 (2017).
Q. Shen, R. Lal, B. A. Luellen, et al., Biol. Psychiatry, 68(6), 512 – 520 (2010).
B. Luscher and T. Fuchs, Adv. Pharmacol., 73, 97 – 144 (2015).
C. Caldji, D. Francis, S. Sharma, et al., Neuropsychopharmacology, 22(3), 219 – 229 (2000).
C. Braestrup, M. Nielsen, E. B. Nielsen, et al., Psychopharmacology (Berlin, Ger.), 65(3), 273 – 277 (1979).
G. B. Acosta, M. E. Otero Losada, and M. C. Rubio, Neurosci. Lett., 154(1–2), 175 – 178 (1993).
K. Matsumoto, G. Puia, E. Dong, et al., Stress, 10(1), 3 – 12 (2007).
Z. Merali, L. Du, P. Hrdina, et al., J. Neurosci., 24, 1478 – 1485 (2004).
A. Sequeira, T. Klempan, L. Canetti, et al., Mol. Psychiatry, 12, 640 – 655 (2007).
T. A. Klempan, A. Sequeira, L. Canetti, et al., Mol. Psychiatry, 14, 175 – 189 (2009).
B. Luscher, Q. Shen, and N. Sahir, Mol. Psychiatry, 16, 383 – 406 (2011).
Acknowledgments
The work was performed in the framework of State Task No. 0521-2019-0009 “Analysis of receptor mechanisms and search for CNS pharmacological protection agents with disrupted brain blood flow and cognitive disorders.
Conflict of interests. The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 55, No. 5, pp. 25 – 31, May, 2021.
Rights and permissions
About this article
Cite this article
Abdullina, A.A., Vasil’eva, E.V., Kondrakhin, E.A. et al. Psychotropic and Neuroreceptor Effects of Cyclopropylglycine Upon Intranasal Administration. Pharm Chem J 55, 483–489 (2021). https://doi.org/10.1007/s11094-021-02445-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-021-02445-6