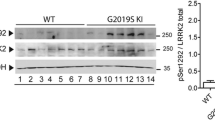
Abstract
Mutations in the leucine-rich repeat kinase 2 (LRRK2) gene are the most frequent cause of autosomal dominant Parkinson’s disease (PD), producing psychiatric and motor symptoms. We conducted this study to explore whether microglial dopaminergic (DAergic) fiber refinement and synaptic pruning are involved in the abnormal behavioral phenotypes of carriers of the LRRK2 G2019S mutation, by employing young and middle-aged PD model mice. The results revealed a characteristic late-onset hyperactivity and a progressive decline in the motor coordination of the LRRK2 G2019S mutation mice. LRRK2 G2019S mutation-induced aberrant microglial morphogenesis, with more branches and junctions per cell, resulted in excessive microglial refinement of dopaminergic (DAergic) fibers. Moreover, aberrant synaptic pruning distinctly impacted the prefrontal cortex (PFC) and dorsal striatum (DS), with significantly higher spine density in the PFC but the opposite effects in the DS region. Furthermore, LRRK2 G2019S mutation remodeled the inflammatory transcription landscape of microglia, rendering certain cerebral areas highly susceptible to microglial immune response. These findings indicate that LRRK2 G2019S mutation induces the production of inflammatory cytokines and mediates abnormal microglial morphogenesis and activity, resulting in abnormal phagocytosis, synaptic pruning and loss of DAergic fibers during aging, and, eventually, PD-related behavioral abnormalities.






Similar content being viewed by others
Availability of Data and Materials
The datasets used and/or analyzed in this study are available from the corresponding authors on reasonable request.
Abbreviations
- PD:
-
Parkinson’s disease
- LRRK2:
-
Leucine-rich repeat kinase 2
- DA:
-
Dopaminergic
- DAT:
-
Dopamine transporter
- TH:
-
Tyrosine hydroxylase
- CD68:
-
Cluster of differentiation 68
- DS:
-
Dorsal striatum
- GS:
-
LRRK2 G2019S mutation mice
- Iba1:
-
Ionized calcium binding adaptor molecule 1
- PFC:
-
Prefrontal cortex
- PSD95:
-
Postsynaptic density protein 95
- VGLUT1:
-
Vesicular glutamate transporter 1
References
Arganda-Carreras I, Fernandez-Gonzalez R, Munoz-Barrutia A, Ortiz-De-Solorzano C (2010) 3D reconstruction of histological sections: Application to mammary gland tissue. Microsc Res Tech 73(11):1019–1029. https://doi.org/10.1002/jemt.20829
Bonnard C, Navaratnam N, Ghosh K, Chan PW, Tan TT, Pomp O, Ng AYJ, Tohari S, Changede R, Carling D, Venkatesh B, Altunoglu U, Kayserili H, Reversade B (2020) A loss-of-function NUAK2 mutation in humans causes anencephaly due to impaired Hippo-YAP signaling. J Exp Med 217 (12). https://doi.org/10.1084/jem.20191561
Bras JP, Bravo J, Freitas J, Barbosa MA, Santos SG, Summavielle T, Almeida MI (2020) TNF-alpha-induced microglia activation requires miR-342: impact on NF-kB signaling and neurotoxicity. Cell Death Dis 11(6):415. https://doi.org/10.1038/s41419-020-2626-6
Choi C, Kim A, Byun JW, Baik JS, Yun H, Kim A, Jung CR, Song Q, Shin ES, Seo H, Suh YH, Jou I, Park BJ, Kang HC, Joe EH (2015) LRRK2 G2019S mutation attenuates microglial motility by inhibiting focal adhesion kinase. Nat Commun 6:8255. https://doi.org/10.1038/ncomms9255
Chou JS, Chen CY, Chen YL, Weng YH, Yeh TH, Lu CS, Chang YM, Wang HL (2014) (G2019S) LRRK2 causes early-phase dysfunction of SNpc dopaminergic neurons and impairment of corticostriatal long-term depression in the PD transgenic mouse. Neurobiol Dis 68:190–199. https://doi.org/10.1016/j.nbd.2014.04.021
Coley AA, Gao WJ (2018) PSD95: A synaptic protein implicated in schizophrenia or autism? Prog Neuropsychopharmacol Biol Psychiatry 82:187–194. https://doi.org/10.1016/j.pnpbp.2017.11.016
De Virgilio A, Greco A, Fabbrini G, Inghilleri M, Rizzo MI, Gallo A, Conte M, Rosato C, Ciniglio Appiani M, de Vincentiis M (2016) Parkinson’s disease: Autoimmunity and neuroinflammation. Autoimmun Rev 15(10):1005–1011. https://doi.org/10.1016/j.autrev.2016.07.022
Dwyer Z, Rudyk C, Thompson A, Farmer K, Fenner B, Fortin T, Derksen A, Sun H, Hayley S, Clint (2020) Leucine-rich repeat kinase-2 (LRRK2) modulates microglial phenotype and dopaminergic neurodegeneration. Neurobiol Aging 91:45–55. https://doi.org/10.1016/j.neurobiolaging.2020.02.017
Fernagut PO, Diguet E, Labattu B, Tison F (2002) A simple method to measure stride length as an index of nigrostriatal dysfunction in mice. J Neurosci Methods 113(2):123–130. https://doi.org/10.1016/s0165-0270(01)00485-x
Filipello F, Morini R, Corradini I, Zerbi V, Canzi A, Michalski B, Erreni M, Markicevic M, Starvaggi-Cucuzza C, Otero K, Piccio L, Cignarella F, Perrucci F, Tamborini M, Genua M, Rajendran L, Menna E, Vetrano S, Fahnestock M, Paolicelli RC, Matteoli M (2018) The microglial innate immune receptor TREM2 is required for synapse elimination and normal brain connectivity. Immunity 48 (5):979–991.e978. https://doi.org/10.1016/j.immuni.2018.04.016
Gardet A, Benita Y, Li C, Sands BE, Ballester I, Stevens C, Korzenik JR, Rioux JD, Daly MJ, Xavier RJ, Podolsky DK (2010) LRRK2 is involved in the IFN-gamma response and host response to pathogens. J Immunol 185(9):5577–5585. https://doi.org/10.4049/jimmunol.1000548
Giesert F, Hofmann A, Burger A, Zerle J, Kloos K, Hafen U, Ernst L, Zhang J, Vogt-Weisenhorn DM, Wurst W (2013) Expression analysis of Lrrk1, Lrrk2 and Lrrk2 splice variants in mice. PLoS One 8(5):e63778. https://doi.org/10.1371/journal.pone.0063778
Hakimi M, Selvanantham T, Swinton E, Padmore RF, Tong Y, Kabbach G, Venderova K, Girardin SE, Bulman DE, Scherzer CR, LaVoie MJ, Gris D, Park DS, Angel JB, Shen J, Philpott DJ, Schlossmacher MG (2011) Parkinson’s disease-linked LRRK2 is expressed in circulating and tissue immune cells and upregulated following recognition of microbial structures. J Neural Transm 118(5):795–808. https://doi.org/10.1007/s00702-011-0653-2
Harry GJ (2013) Microglia during development and aging. Pharmacol Ther 139(3):313–326. https://doi.org/10.1016/j.pharmthera.2013.04.013
Healy DG, Falchi M, O’Sullivan SS, Bonifati V, Durr A, Bressman S, Brice A, Aasly J, Zabetian CP, Goldwurm S, Ferreira JJ, Tolosa E, Kay DM, Klein C, Williams DR, Marras C, Lang AE, Wszolek ZK, Berciano J, Schapira AHV, Lynch T, Bhatia KP, Gasser T, Lees AJ, Wood NW (2008) Phenotype, genotype, and worldwide genetic penetrance of LRRK2-associated Parkinson’s disease: a case-control study. Lancet Neurol 7(7):583–590. https://doi.org/10.1016/s1474-4422(08)70117-0
Ho M (2019) Microglia in Parkinson’s disease. Adv Exp Med Biol 1175:335–353. https://doi.org/10.1007/978-981-13-9913-8_13
Huntley GW, Benson DL (2020) Origins of parkinson’s disease in brain development: insights from early and persistent effects of LRRK2-G2019S on striatal circuits. Front Neurosci 14:265. https://doi.org/10.3389/fnins.2020.00265
Ikegami A, Haruwaka K, Wake H (2019) Microglia: Lifelong modulator of neural circuits. Neuropathology : Official Journal of the Japanese Society of Neuropathology 39(3):173–180. https://doi.org/10.1111/neup.12560
Ip CW, Klaus LC, Karikari AA, Visanji NP, Brotchie JM, Lang AE, Volkmann J, Koprich JB (2017) AAV1/2-induced overexpression of A53T-alpha-synuclein in the substantia nigra results in degeneration of the nigrostriatal system with Lewy-like pathology and motor impairment: a new mouse model for Parkinson’s disease. Acta Neuropathol Commun 5(1):11. https://doi.org/10.1186/s40478-017-0416-x
Kim KS, Marcogliese PC, Yang J, Callaghan SM, Resende V, Abdel-Messih E, Marras C, Visanji NP, Huang J, Schlossmacher MG, L Trinkle-Mulcahy L, Slack RS, Lang AE, Canadian Lrrk2 in Inflammation T, Park DS (2018) Regulation of myeloid cell phagocytosis by LRRK2 via WAVE2 complex stabilization is altered in Parkinson's disease. Proc Natl Acad Sci USA 115 22 E5164 E5173. https://doi.org/10.1073/pnas.1718946115
Kundey SMA, Bajracharya A, Boettger-Tong H, Fountain SB, Rowan JD (2019) Sex differences in serial pattern learning in mice. Behavioural processes 168:103958. https://doi.org/10.1016/j.beproc.2019.103958
Lee J, Lee S, Ryu YJ, Lee D, Kim S, Seo JY, Oh E, Paek SH, Kim SU, Ha CM, Choi SY, Kim KT (2019) Vaccinia-related kinase 2 plays a critical role in microglia-mediated synapse elimination during neurodevelopment. Glia 67(9):1667–1679. https://doi.org/10.1002/glia.23638
Liu G, Sgobio C, Gu X, Sun L, Lin X, Yu J, Parisiadou L, Xie C, Sastry N, Ding J, Lohr KM, Miller GW, Mateo Y, Lovinger DM, Cai H (2015) Selective expression of Parkinson’s disease-related Leucine-rich repeat kinase 2 G2019S missense mutation in midbrain dopaminergic neurons impairs dopamine release and dopaminergic gene expression. Hum Mol Genet 24(18):5299–5312. https://doi.org/10.1093/hmg/ddv249
Mancini A, Mazzocchetti P, Sciaccaluga M, Megaro A, Bellingacci L, Beccano-Kelly DA, Di Filippo M, Tozzi A, Calabresi P (2020) From synaptic dysfunction to neuroprotective strategies in genetic parkinson’s disease: Lessons From LRRK2. Front Cell Neurosci 14:158. https://doi.org/10.3389/fncel.2020.00158
Mandemakers W, Snellinx A, O’Neill MJ, de Strooper B (2012) LRRK2 expression is enriched in the striosomal compartment of mouse striatum. Neurobiol Dis 48(3):582–593. https://doi.org/10.1016/j.nbd.2012.07.017
Matikainen-Ankney BA, Kezunovic N, Menard C, Flanigan ME, Zhong Y, Russo SJ, Benson DL, Huntley GW (2018) Parkinson’s disease-linked LRRK2-G2019S mutation alters synaptic lasticity and promotes resilience to chronic social stress in young adulthood. J Neurosci 38(45):9700–9711. https://doi.org/10.1523/JNEUROSCI.1457-18.2018
Nolan SO, Reynolds CD, Smith GD, Holley AJ, Escobar B, Chandler MA, Volquardsen M, Jefferson T, Pandian A, Smith T, Huebschman J, Lugo JN (2017) Deletion of Fmr1 results in sex-specific changes in behavior. Brain and Behav 7(10):e00800. https://doi.org/10.1002/brb3.800
Novello S, Arcuri L, Dovero S, Dutheil N, Shimshek DR, Bezard E, Morari M (2018) G2019S LRRK2 mutation facilitates alpha-synuclein neuropathology in aged mice. Neurobiol Dis 120:21–33. https://doi.org/10.1016/j.nbd.2018.08.018
Pan RY, Ma J, Kong XX, Wang XF, Li SS, Qi XL, Yan YH, Cheng J, Liu Q, Jin W, Tan CH, Yuan Z (2019) Sodium rutin ameliorates Alzheimer's disease-like pathology by enhancing microglial amyloid-beta clearance. Sci Adv 5 (2):eaau6328. https://doi.org/10.1126/sciadv.aau6328
Papageorgiou IE, Lewen A, Galow LV, Cesetti T, Scheffel J, Regen T, Hanisch UK, Kann O (2016) TLR4-activated microglia require IFN-gamma to induce severe neuronal dysfunction and death in situ. Proc Natl Acad Sci U S A 113(1):212–217. https://doi.org/10.1073/pnas.1513853113
Pimenova AA, Herbinet M, Gupta I, Machlovi SI, Bowles KR, Marcora E, Goate AM (2021) Alzheimer's-associated PU.1 expression levels regulate microglial inflammatory response. Neurobiol dis 148:105217. https://doi.org/10.1016/j.nbd.2020.105217
Russo I, Bubacco L, Greggio E (2014) LRRK2 and neuroinflammation: partners in crime in Parkinson’s disease? J Neuroinflammation 11(52):52. https://doi.org/10.1186/1742-2094-11-52
Rustenhoven J, Smith AM, Smyth LC, Jansson D, Scotter EL, Swanson MEV, Aalderink M, Coppieters N, Narayan P, Handley R, Overall C, Park TIH, Schweder P, Heppner P, Curtis MA, Faull RLM, Dragunow M (2018) PU.1 regulates Alzheimer's disease-associated genes in primary human microglia. Mol Neurodegener 13 (1):44. https://doi.org/10.1186/s13024-018-0277-1
Schapansky J, Nardozzi JD, LaVoie MJ (2015) The complex relationships between microglia, alpha-synuclein, and LRRK2 in Parkinson’s disease. Neuroscience 302:74–88. https://doi.org/10.1016/j.neuroscience.2014.09.049
Schildt A, Walker MD, Dinelle K, Miao Q, Schulzer M, O’Kusky J, Farrer MJ, Doudet DJ, Sossi V (2019) Single inflammatory trigger leads to neuroinflammation in LRRK2 rodent model without degeneration of dopaminergic neurons. J Parkinsons Dis 9(1):121–139. https://doi.org/10.3233/JPD-181446
Sellgren C, Gracias J, Watmuff B, Biag J, Thanos J, Whittredge P, Fu T, Worringer K, Brown H, Wang J, Kaykas A, Karmacharya R, Goold C, Sheridan S, Perlis R (2019) Increased synapse elimination by microglia in schizophrenia patient-derived models of synaptic pruning. Nat Neurosci 22(3):374–385. https://doi.org/10.1038/s41593-018-0334-7
Shapouri-Moghaddam A, Mohammadian S, Vazini H, Taghadosi M, Esmaeili SA, Mardani F, Seifi B, Mohammadi A, Afshari JT, Sahebkar A (2018) Macrophage plasticity, polarization, and function in health and disease. J Cell Physiol 233(9):6425–6440. https://doi.org/10.1002/jcp.26429
Sheerin UM, Houlden H, Wood NW (2014) Advances in the Genetics of Parkinson’s Disease: A Guide for the Clinician. Mov Disord Clin Pract 1(1):3–13. https://doi.org/10.1002/mdc3.12000
Wang Z, Hou L, Wang D (2019) Effects of exercise-induced fatigue on the morphology of asymmetric synapse and synaptic protein levels in rat striatum. Neurochem Int 129:104476. https://doi.org/10.1016/j.neuint.2019.104476
Wile DJ, Agarwal PA, Schulzer M, Mak E, Dinelle K, Shahinfard E, Vafai N, Hasegawa K, Zhang J, McKenzie J, Neilson N, Strongosky A, Uitti RJ, Guttman M, Zabetian CP, Ding Y-S, Adam M, Aasly J, Wszolek ZK, Farrer M, Sossi V, Stoessl AJ (2017) Serotonin and dopamine transporter PET changes in the premotor phase of LRRK2 parkinsonism: cross-sectional studies. Lancet Neurol 16(5):351–359. https://doi.org/10.1016/s1474-4422(17)30056-x
Wynn TA (2003) IL-13 effector functions. Annu Rev Immunol 21:425–456. https://doi.org/10.1146/annurev.immunol.21.120601.141142
Xiao Q, Yang S, Le W (2015) G2019S LRRK2 and aging confer susceptibility to proteasome inhibitor-induced neurotoxicity in nigrostriatal dopaminergic system. J Neural Transm 122(12):1645–1657. https://doi.org/10.1007/s00702-015-1438-9
Xu Q, Shenoy S, Li C (2012) Mouse models for LRRK2 Parkinson’s disease. Parkinsonism Relat Disord 18:S186–S189. https://doi.org/10.1016/s1353-8020(11)70058-x
Zhang Y, Wu Q, Zhang L, Wang Q, Yang Z, Liu J, Feng L (2019) Caffeic acid reduces A53T alpha-synuclein by activating JNK/Bcl-2-mediated autophagy in vitro and improves behaviour and protects dopaminergic neurons in a mouse model of Parkinson’s disease. Pharmacol Res 150:104538. https://doi.org/10.1016/j.phrs.2019.104538
Acknowledgements
We would like to thank Professor Hongzhi Huang from School of Arts and Sciences of Fujian Medical University for his kind proofreading and polishing of this manuscript.
Funding
This work was supported by grants from the National Natural Science Foundation of China (No. 81771179, No. 82071175); Natural Science Foundation (Key Project) of Fujian Province, China (No. 2020J02022); Fujian Provincial Health-Education Joint Research Project (No. WKJ2016-2–08); Startup Fund for scientific research from Fujian Medical University (No. 2017XQ2023, No. 2017XQ2035).
Author information
Authors and Affiliations
Contributions
Xiaodong Pan conceived the study design and Qiuyang Zhang conducted the animal study and quantitative analysis, and they were chief contributors in writing the manuscript. Xiaojuan Cheng, Wei Wu, Siyu Yang and Hanlin You performed the animal behavior study and histochemical experiments. Xiaochun Chen, Nan Liu and Zucheng Ye reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All animal experimental procedures conformed to the National Institute of Health Guidelines for the Care and Use of Laboratory Animals and were approved by the Institutional Animal Care and Use Committee of Fujian Medical University.
Consent for Publication
All authors read and approved the final manuscript.
Disclosures
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, Q., Cheng, X., Wu, W. et al. Age-related LRRK2 G2019S Mutation Impacts Microglial Dopaminergic Fiber Refinement and Synaptic Pruning Involved in Abnormal Behaviors. J Mol Neurosci 72, 527–543 (2022). https://doi.org/10.1007/s12031-021-01896-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-021-01896-6