Abstract
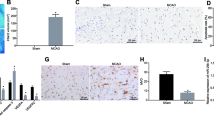
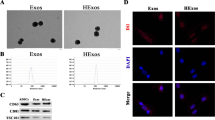
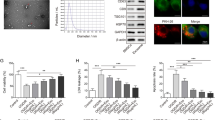
Exosomes harvested from bone marrow-derived mesenchymal stromal cells (BMSCs) have shown treatment potential in many diseases. In vitro, Zeb2/Axin2 stimulated endogenous neurogenesis, which induced functional recovery after stroke. Here, we investigated whether the Zeb2/Axin2-enriched exosomes harvested from BMSCs transfected with a Zeb2/Axin2 overexpression plasmid would enhance neurological recovery. Compared with the control, both exosome treatments significantly improved functional recovery, and Zeb2/Axin2-enriched exosomes had significantly more improved effects on neurological function, neurogenesis, and neurite remodeling/neuronal dendrite plasticity than the control BMSC exosome treatment in a middle cerebral artery occlusion MCAO rat model. After stimulation with Zeb2/Axin2-enriched BMSC exosomes, the spatial memory and nerve function of MCAO rats showed marked recovery. The number of neurons was increased in the subventricular zone (SVZ), hippocampus, and cortex area, while the expression of nerve growth factors (NGF, BDNF, etc.) was upregulated. In the ischemic boundary zone, Zeb2/Axin2-enriched exosomes promoted synaptic remodeling by increasing the number of synapses and reversed the axonal loss of phosphorylated neurofilament (SMI-31) and synaptophysin (SYN) caused by ischemic injury, thus alleviating axonal demise and promoting synaptic proliferation. In vitro, Zeb2/Axin2-enriched exosomes significantly increased neurite branching and elongation of cultured cortical embryonic rat neurons under oxygen- and glucose-deprived (OGD) conditions. Moreover, Ex-Zeb2/Axin2-enriched exosomes downregulated the protein level of SOX10, endothelin-3/EDNRB, and Wnt/β-catenin expression. In conclusion, exosomes harvested from Ex-Zeb2/Axin2 BMSC could improve post-stroke neuroplasticity and functional recovery in MCAO rats by promoting proliferation and differentiation of neural stem cells. The mechanism may be related to the SOX10, Wnt/β-catenin, and endothelin-3/EDNRB pathways.





Similar content being viewed by others
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aimone JB, Li Y, Lee SW, Clemenson GD, Deng W, Gage FH (2014) Regulation and function of adult neurogenesis: from genes to cognition. Physiol Rev 94(4):991–1026
Braccioli L, Velthoven C, Heijnen CJ (2014) Exosomes: A New Weapon to Treat the Central Nervous System. Mol Neurobiol 49(1):113–119
Brouns R, Espinoza AV, Goudman L, Moens M, Verlooy J (2019) Interventions to promote work participation after ischaemic stroke: A systematic review. Clin Neurol Neurosurg 105458
Chang D-J, Oh S-H, Lee N, Choi C, Jeon I, Kim HS et al (2013) Contralaterally transplanted human embryonic stem cell-derived neural precursor cells (ENStem-A) migrate and improve brain functions in stroke-damaged rats. Exp Mol Med 45(11):e53–e53
Chavez JC, Baranova O, Lin J, Pichiule P (2006) The transcriptional activator hypoxia inducible factor 2 (HIF-2/EPAS-1) regulates the oxygen-dependent expression of erythropoietin in cortical astrocytes. 26(37):9471–9481
Chen J, Magavi SS, Macklis JD (2004) Neurogenesis of corticospinal motor neurons extending spinal projections in adult mice. Proc Natl Acad Sci 101(46):16357–16362
Chen Z-Z, Jiang X-D, Zhang L-L, Shang J-H, Du M-X, Xu G et al (2008) Beneficial effect of autologous transplantation of bone marrow stromal cells and endothelial progenitor cells on cerebral ischemia in rabbits. Neurosci Lett 445(1):36–41
Chivet M, Hemming F, Pernet-Gallay K, Fraboulet S, Sadoul R (2012) Emerging Role of Neuronal Exosomes in the Central Nervous System. Front Physiol 3:145
Comijn J, Berx G, Vermassen P, Verschueren K, van Grunsven L, Bruyneel E et al (2001) The two-handed E box binding zinc finger protein SIP1 downregulates E-cadherin and induces invasion. Mol Cell 7(6):1267–1278
Dai Z-M, Sun S, Wang C, Huang H, Hu X, Zhang Z et al (2014) Stage-specific regulation of oligodendrocyte development by Wnt/β-catenin signaling. J Neurosci 34(25):8467–8473
Dao DY, Yang X, Di C, Zuscik M, O’Keefe, RJ (2007) Axin1 and Axin2 are regulated by TGF-β and mediate cross-talk between TGF-β and Wnt signaling pathways. Ann N Y Acad Sci 1116:82
Darsalia V, Kallur T, Kokaia Z (2007) Survival, migration and neuronal differentiation of human fetal striatal and cortical neural stem cells grafted in stroke-damaged rat striatum. Eur J Neurosci 26(3):605–614
Fancy SP, Harrington EP, Yuen TJ, Silbereis JC, Zhao C, Baranzini SE et al (2011) Axin2 as regulatory and therapeutic target in newborn brain injury and remyelination. Nat Neurosci 14(8):1009
Felling RJ, Levison SW (2003) Enhanced neurogenesis following stroke. J Neurosci Res 73(3):277–283
Ferro JM, Caeiro L, Figueira ML (2016) Neuropsychiatric sequelae of stroke. Nat Rev Neurol 12(5):269
Ji L-L, Long X-F, Tian H, Liu Y-F (2013) Effect of transplantation of bone marrow stem cells on myocardial infarction size in a rabbit model. World J Emerg Med 4(4):304
Jin C, Tian H, Li J et al (2018) Stem cell education for medical students at Tongji University: Primary cell culture and directional differentiation of rat bone marrow mesenchymal stem cells. Biochem Mol Biol Educ 46:151–154
Khodanovich M, Nemirovich-Danchenko NM (2019) New neurons in the post-ischemic and injured brain: migrating or resident? Front Neurosci 13:588
Kim YR, Kim HN, Ahn SM, Choi YH, Shin HK, Choi BT (2014) Electroacupuncture promotes post-stroke functional recovery via enhancing endogenous neurogenesis in mouse focal cerebral ischemia. PLoS One 9(2)
Lee E-G, Son H (2009) Adult hippocampal neurogenesis and related neurotrophic factors. BMB Rep 42(5):239–244
Ling X, Zhang G, Zhu Q, Zhang J, Li Q, Niu X et al (2018) Exosomes from Human Urine-Derived Stem Cells Promote Neurogenesis Via Histone Deacetylase6 Regulation in Ischemic Stroke. Available at SSRN 3288885
Longa EZ, Weinstein P, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91
Magavi SS, Leavitt BR, Macklis JD (2000) Induction of neurogenesis in the neocortex of adult mice. Nature 405(6789):951–955
Mahmood A, Lu D, Qu C, Goussev A, Chopp M (2006) Long-term recovery after bone marrow stromal cell treatment of traumatic brain injury in rats. J Neurosurg 104(2):272–277
Manuel GE, Johnson T, Liu D (2017) Therapeutic angiogenesis of exosomes for ischemic stroke. International Journal of Physiology, Pathophysiology and Pharmacology 9(6):188
Moreno-Jiménez EP, Flor-García M, Terreros-Roncal J, Rábano A, Cafini F, Pallas-Bazarra N et al (2019) Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat Med 25(4):554–560
Nagasawa H, Kogure K (1989) Correlation between cerebral blood flow and histologic changes in a new rat model of middle cerebral artery occlusion. Stroke 20(8):1037–1043
Nudo RJ (2001) Role of adaptive plasticity in recovery of function after damage to motor cortex. Muscle Nerve 24
Oki K, Tatarishvili J, Wood J, Koch P, Wattananit S, Mine Y et al (2012) Human-induced pluripotent stem cells form functional neurons and improve recovery after grafting in stroke-damaged brain. STEM CELLS 30(6):1120–1133
Qu R, Li Y, Gao Q, Shen L, Zhang J, Liu Z et al (2007) Neurotrophic and growth factor gene expression profiling of mouse bone marrow stromal cells induced by ischemic brain extracts. Neuropathology 27(4):355–363
Quesseveur G, David D, Gaillard M, Pla P, Wu M, Nguyen H et al (2013) BDNF overexpression in mouse hippocampal astrocytes promotes local neurogenesis and elicits anxiolytic-like activities. Transl Psychiatry 3(4):e253–e253
Rexach J, Swarup V, Chang T, Geschwind D (2019) Dementia risk genes engage gene networks poised to tune the immune response towards chronic inflammatory states. bioRxiv 597542
Scharfman H, Goodman J, Macleod A, Phani S, Antonelli C, Croll S (2005) Increased neurogenesis and the ectopic granule cells after intrahippocampal BDNF infusion in adult rats. Exp Neurol 192(2):348–356
Schmidt HD, Duman RS (2007) The role of neurotrophic factors in adult hippocampal neurogenesis, antidepressant treatments and animal models of depressive-like behavior. Behav Pharmacol 18(5–6):391–418
Seki T, Hori T, Miyata H, Maehara M, Namba T (2019) Analysis of proliferating neuronal progenitors and immature neurons in the human hippocampus surgically removed from control and epileptic patients. Sci Rep 9(1):1–14
Shin MK, Levorse J, Ingram RS, Tilghman SM (1999) The temporal requirement for endothelin receptor-B signalling during neural crest development. Nature 402:496–501
Sidebotham EL, Woodward MN, Kenny SE et al (2002) Localization and endothelin-3 dependence of stem cells of the enteric nervous system in the embryonic colon. J Pediatr Surg 37(2):145–150
Tang Y, Cui Y-C, Wang X-J, Wu A-L, Hu G-F, Luo F-L et al (2012) Neural progenitor cells derived from adult bone marrow mesenchymal stem cells promote neuronal regeneration. Life Sci 91(19–20):951–958
Théry C, Amigorena S, Raposo G (2006) Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol 3(3):22
Théry C, Zitvogel L, Amigorena S (2002) Exosomes: composition, biogenesis and function. Nat Rev Immunol 2(8):569–579
Tian T, Zhang H-X, He C-P, Fan S, Zhu Y-L, Qi C et al (2018a) Surface functionalized exosomes as targeted drug delivery vehicles for cerebral ischemia therapy. Biomaterials 150:137–149
Tian T, Zhang H, He C, Fan S, Zhu Y, Qi C et al (2018b) Surface Functionalized Exosomes as Targeted Drug Delivery Vehicles for Cerebral Ischemia Therapy 150:137–149
Vahidy FS, Rahbar MH, Zhu H, Rowan PJ, Bambhroliya AB, Savitz SI (2016) Systematic review and meta-analysis of bone marrow–derived mononuclear cells in animal models of ischemic stroke. Stroke 47(6):1632–1639
Valadi H, Ekström K, Bossios A, Sjöstrand M (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9(6):654–659
Watanabe Y, Stanchina L, Lecerf L, Gacem N, Conidi A, Baral V et al (2017) Differentiation of mouse enteric nervous system progenitor cells is controlled by endothelin 3 and requires regulation of Ednrb by SOX10 and ZEB2. Gastroenterology 152(5):1139–1150. e1134
Wu Z, Liang Y, Yu S (2020) Downregulation of microRNA-103a reduces microvascular endothelial cell injury in a rat model of cerebral ischemia by targeting AXIN2. J Cell Physiol 235(5):4720–4733
Xin H, Katakowski M, Wang F, Qian J-Y, Liu XS, Ali MM et al (2017a) MicroRNA-17–92 cluster in exosomes enhance neuroplasticity and functional recovery after stroke in rats. Stroke 48(3):747–753
Xin H, Li Y, Chopp M (2014) Exosomes/miRNAs as mediating cell-based therapy of stroke. Front Cell Neurosci 8:377
Xin H, Li Y, Cui Y, Yang JJ, Zhang ZG, Chopp M (2013a) Systemic administration of exosomes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats. J Cereb Blood Flow Metab 33(11):1711–1715
Xin H, Li Y, Liu Z, Wang X, Shang X, Cui Y et al (2013b) MiR-133b Promotes Neural Plasticity and Functional Recovery After Treatment of Stroke with Multipotent Mesenchymal Stromal Cells in Rats Via Transfer of Exosome-Enriched Extracellular Particles. STEM CELLS 31(12):2737–2746
Xin H, Wang F, Li Y, Lu Q-E, Cheung WL, Zhang Y et al (2017b) Secondary release of exosomes from astrocytes contributes to the increase in neural plasticity and improvement of functional recovery after stroke in rats treated with exosomes harvested from microRNA 133b-overexpressing multipotent mesenchymal stromal cells. Cell Transplant 26(2):243–257
Funding
This work was supported by National Natural Science Foundation of China (Grant 81760349, 81560319) and Applied Basic Research Foundation of Yunnan Province (Grant 2016FB130).
Author information
Authors and Affiliations
Contributions
RW and WZ contributed to the conception and design of the study. LZ, WH, XS and YH performed the experiments, and analyzed and interpreted data. RW and WZ drafted and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
The present study was approved by the Laboratory Animal Ethics Review Committee of Kunming Medical University (Kunming, China).
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wei, R., Zhang, L., Hu, W. et al. Zeb2/Axin2-Enriched BMSC-Derived Exosomes Promote Post-Stroke Functional Recovery by Enhancing Neurogenesis and Neural Plasticity. J Mol Neurosci 72, 69–81 (2022). https://doi.org/10.1007/s12031-021-01887-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-021-01887-7