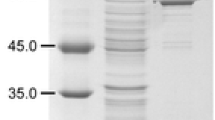
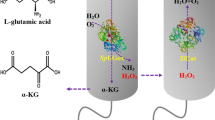
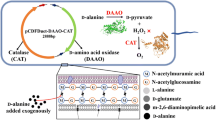
Abstract
Whole-cell systems offer many benefits for biochemical production, such as relatively easy enzyme control and higher tolerance toward harsh environments, than purified enzymes. These systems can be applied to many bioconversion reactions, but they sometimes require cofactor regeneration units to support reactions at high substrate concentrations. Here, we examined l-glutamate oxidase (GOX) from Streptomyces sp. X119-6, which produces α-ketoglutarate (α-KG) from l-glutamate, and catalase (KatE) from Escherichia coli, which removes hydrogen peroxide generated by GOX. After optimizing the expression vector, pH, strains, culture conditions, and isopropyl β-d-1-thiogalactopyranoside concentration, we compared their efficiency to that of a previously reported GOX from Streptomyces mobaraensis. Our results indicated that GOX from Streptomyces sp. X119-6 and KatE increased α-KG production by 2.76-fold. This GOX required high levels of α-KG as an amino donor to convert 5-aminovaleric acid to glutaric acid. Performing the reaction at pH 8 enabled us to avoid the exogenous addition of catalase, but severe substrate inhibition was observed, resulting in the production of 287 mM glutaric acid. This α-KG regeneration system has potential for improving production in various aminotransferase systems.
Similar content being viewed by others
References
J. Arima, C. Sasaki, C. Sakaguchi, H. Mizuno, T. Tamura, A. Kashima, H. Kusakabe, S. Sugio and K. Inagaki, FEBS J., 276, 3894 (2009).
P. Niu, X. Dong, Y. Wang and L. Liu, J. Biotechnol., 179, 56 (2014).
X. Zhang, N. Xu, J. Li, Z. Ma, L. Wei, Q. Liu and J. Liu, Enzyme Microbial Technol., 136, 109530 (2020).
O. Soldatkina, O. Soldatkin, B. O. Kasap, D. Y. Kucherenko, I. Kuche-renko, B. A. Kurc and S. Dzyadevych, Nanoscale Res. Lett., 12, 1 (2017).
A. J. Wagenmakers, Skeletal Muscle Metabolism in Exercise and Diabetes, 441, 307 (1998).
A. Kumari, Sweet biochemistry: Remembering structures, cycles, and pathways by mnemonics, Academic Press, Rohtak, Haryana, India (2017).
H. S. Srivastava and R. P. Singh, Phytochemistry, 26, 597 (1987).
Q. Liu, X. Ma, H. Cheng, N. Xu, J. Liu and Y. Ma, Biotechnol. Lett., 39, 913 (2017).
J. Wu, X. Fan, J. Liu, Q. Luo, J. Xu and X. Chen, Appl. Microbiol. Biotechnol., 102, 4755 (2018).
S.-Y. Yang, T.-R. Choi, H.-R. Jung, Y.-L. Park, Y.-H. Han, H.-S. Song, R. Gurav, S. K. Bhatia, K. Park and J.-O. Ahn, Enzyme Microbial Technol., 133, 109446 (2020).
A. Böhmer, A. Müller, M. Passarge, P. Liebs, H. Honeck and H. G. Müller, Eur. J. Biochem., 182, 327 (1989).
L. Wang, R. Peng, Y. Tian, M. Liu and Q. Yao, Biotechnol. Lett., 39, 523 (2017).
S. Wachiratianchai, A. Bhumiratana and S. Udomsopagit, Electronic J. Biotechnol., 7, 09 (2004).
Y. G. Hong, Y. M. Moon, T. R. Choi, H. R. Jung, S. Y. Yang, J. O. Ahn, J. C. Joo, K. Park, Y. G. Kim and S. K. Bhatia, Biotechnol. Bioengineering, 116, 333 (2019).
S.-Y. Yang, T.-R. Choi, H.-R. Jung, Y.-L. Park, Y.-H. Han, H.-S. Song, S. K. Bhatia, K. Park, J.-O. Ahn and W.-Y. Jeon, Enzyme Microbial Technol., 128, 72 (2019).
S. K. Bhatia, R. K. Bhatia and Y.-H. Yang, Rev. Environ. Sci. Bio/Technol., 15, 639 (2016).
Y.-G. Hong, Y.-M. Moon, J.-W. Hong, S.-Y. No, T.-R. Choi, H.-R. Jung, S.-Y. Yang, S. K. Bhatia, J.-O. Ahn and K.-M. Park, Enzyme Microbial Technol., 118, 57 (2018).
Y.-M. Moon, R. Gurav, J. Kim, Y.-G. Hong, S. K. Bhatia, H.-R. Jung, J.-W. Hong, T. R. Choi, S. Y. Yang and H. Y. Park, Biotechnol. Bioprocess Eng., 23, 442 (2018).
T.-R. Choi, J.-M. Jeon, S. K. Bhatia, R. Gurav, Y. H. Han, Y. L. Park, J.-Y. Park, H.-S. Song, H. Y. Park and J.-J. Yoon, Biotechnol. Bioprocess Eng., 25, 279 (2020).
J. Y. Park, Y.-L. Park, T.-R. Choi, H. J. Kim, H.-S. Song, Y.-H. Han, S. M. Lee, S. L. Park, H. S. Lee and S. K. Bhatia, Korean J. Chem. Eng., 37, 2225 (2020).
J. Kim, H.-M. Seo, S. K. Bhatia, H.-S. Song, J.-H. Kim, J.-M. Jeon, K.-Y. Choi, W. Kim, J.-J. Yoon and Y.-G. Kim, Sci. Rep., 7, 1 (2017).
Y.-M. Moon, S. Y. Yang, T. R. Choi, H.-R. Jung, H.-S. Song, Y.-H. Han, H. Y. Park, S. K. Bhatia, R. Gurav and K. Park, Enzyme Microbial Technol., 127, 58 (2019).
J.-H. Kim, J. Kim, H.-J. Kim, G. Sathiyanarayanan, S. K. Bhatia, H.-S. Song, Y.-K. Choi, Y.-G. Kim, K. Park and Y.-H. Yang, Enzyme Microbial Technol., 104, 9 (2017).
Acknowledgements
The authors would like to acknowledge the KU Research Professor Program of Konkuk University, Seoul, South Korea. This study was supported by the National Research Foundation of Korea (NRF) (NRF-2019R1F1A1058805 and NRF-2019M3E6A1103979) and the Research Program to solve the social issues of the NRF funded by the Ministry of Science and ICT (2017M3A9E4077234). This work was also supported by the R&D Program of MOTIE/KEIT (20009508 and 20014350).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Electronic supplementary material
11814_2021_855_MOESM1_ESM.pdf
Application of l-glutamate oxidase from Streptomyces sp. X119-6 with catalase (KatE) to whole-cell systems for glutaric acid production in Escherichia coli
Rights and permissions
About this article
Cite this article
Ham, S., Han, YH., Kim, S.H. et al. Application of l-glutamate oxidase from Streptomyces sp. X119-6 with catalase (KatE) to whole-cell systems for glutaric acid production in Escherichia coli. Korean J. Chem. Eng. 38, 2106–2112 (2021). https://doi.org/10.1007/s11814-021-0855-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-021-0855-8