Abstract
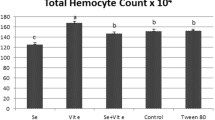
The objective of the present study was to investigate the impact of zinc and copper on some biomarkers in a model organism Galleria mellonella L. We investigated the effects of Cu and Zn (10, 50, and 100 mg/100 g diets) on different biomarkers such as oxidative stress parameters (SOD and CAT activities and MDA levels), energy resources (protein, lipid and glycogen levels), electrolyte contents (Ca, Na, and K levels), total hemocyte count (THC), and growth and development of G. mellonella. Additionally, the accumulation levels of the used metals were also studied. Cu caused a significant decrease in protein, lipid and glycogen levels. SOD and CAT activities significantly increased at all concentrations of Cu, while they significantly increased at only high concentrations of Zn (50 and 100 mg). Lipid peroxidation levels (MDA) significantly elevated at high concentrations of both metals. It was determined that the Cu and Zn accumulation increased depending on the increase of the concentration. Zn caused an alteration in Ca level at the concentrations of 50 and 100 mg, and K and Na levels at all concentrations. While, THC significantly reduced at all Cu concentrations, this reduction was observed only at higher Zn concentrations (50 and 100 mg). Larval and pupal development time significantly extended at the highest concentration (100 mg) of Cu, and females’ lifespan significantly shortened at all concentrations of Cu. Zinc caused an extension in larval development time at the highest concentration (100 mg), and caused a shortening in females and males’ lifetime at all concentrations. The observed changes in biomarkers can be used as the illustration of potential toxic effects of high levels of Cu and Zn in organisms.
Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Akman Gündüz N, Mercan S, Özcan Ö (2020) Effect of cadmium and lead on total hemocyte count of Achroia grisella Fabr. (Lepidoptera: Pyralidae). GUSTIJ 10 (1):190–194. https://doi.org/10.17714/gumusfenbil.531102
Al-Momani FA, Massadeh AM (2005) Effect of different heavy-metal concentrations on Drosophila melanogaster larval growth and development. Biol Trace Elem Res 108 (1–3):271–277. https://doi.org/10.1385/BTER:108:1-3:271
Balistrieri LS, Mebane CA, Schmidt TS (2020) Time-dependent accumulation of Cd, Co, Cu, Ni, and Zn in natural communities of mayfly and caddisfly larvae: metal sensitivity, uptake pathways, and mixture toxicity. Sci Total Environ 732:139011. https://doi.org/10.1016/j.scitotenv.2020.139011
Ballan-Dufrançais C (2002) Localization of metal in cells of pterygote insects. Microsc Res Tech 56 (6):403–420. https://doi.org/10.1002/jemt.10041
Bar-Or D et al (2001) An analog of the human albumin N-Terminus (Asp-Ala-His- Lys) prevents formation of copper induced reactive oxygen species. Biochem Biophys Res Commun 284 (3):856–862. https://doi.org/10.1006/bbrc.2001.5042
Bautista-Covarrubias JC, Velarde-Montes GJ, Voltolina D, García-de la Parra LM, Soto-Jiménez MF, Frías-Espericueta MG (2014) Humoral and haemocytic responses of emopenLitopenaeus vannameiemclose to Cd exposure. Sci World J. https://doi.org/10.1155/2014/903452
Beyenbach KW, Skaer H, Dow JA (2010) The developmental, molecular, and transport biology of malpighian tubules. Annu Rev Entomol 55:351–374. https://doi.org/10.1146/annurev-ento-112408-085512
Bhagat J, Ingole BS, Singh N (2016) Glutathione S-transferase, catalase, superoxide dismutase, glutathione peroxidase, and lipid peroxidation as biomarkers of oxidative stress in snails: a review. Invertebr Surviv J 13 (1):336–349. https://doi.org/10.25431/1824-307X/isj.v13i1.336-349
Borowska J, Sulima B, Niklinska M, Pyza E (2004) Heavy metal accumulation and its effects on development, survival and immuno-competent cells of the house fly Musca domestica from closed laboratory populations as model organism. Fresenius Environ Bull 13 (12):1403–1409
Bronskill JK (1961) A cage to simpllify the rearing of greater wax moth, Galleria mellonella (Pyralidae). J Lepid Soc 15 (2):102–104
Buyukguzel K (2006) Malathion-induced oxidative stress in a parasitoid wasp: effect on adult emergence, longevity and oxidative and antioxidative response of Pimpla turionellae (Hymenoptera:Ichneumonidae). J Econ Entomol 99 (4):1225–1234. https://doi.org/10.1093/jee/99.4.1225
Buyukguzel E (2009) Evidence of oxidative and antioxidative responses by Galleria mellonella larvae to malathion. J Econ Entomol 120 (1):152–159. https://doi.org/10.1603/029.102.0122
Canlı M, Atlı G (2003) The relationships between heavy metal (Cd, Cr, Cu, Fe, Pb, Zn) levels and the size of six Mediterranean fish species. Environ Pollut 121 (1):129–136. https://doi.org/10.1016/S0269-7491(02)00194-X
Catae AF, Roat TC, De Oliveira RA, Ferreira Nocelli RC, Malaspina O (2014) Cytotoxic effects of thiamethoxam in the midgut and malpighian tubules of Africanized Apis mellifera (Hymenoptera: Apidae). Microsc Res Tech 77:274–281. https://doi.org/10.1002/jemt.22339
Chahine S, O’Donnell MJ (2011) Interactions between detoxification mechanisms and excretion in malpighian tubules of Drosophila melanogaster. J Exp Biol 214 (3):462–468. https://doi.org/10.1242/jeb.048884
Chen J, Wang JW, Shu YH (2020) Review on the effects of heavy metal pollution on herbivorous insects. J Appl Ecol 31 (5):1773–1782. https://doi.org/10.13287/j.1001-9332.202005.035
Choi J, Roche H, Caquet T (2001) Hypoxia, hyperoxia and exposure to potassium dichromate or fenitrothion alter the energy metabolism in Chironomus riparius Mg. (Diptera: Chironomidae) larvae. Comp Biochem Physiol C Toxicol 130 (1):11–17. https://doi.org/10.1016/S1532-0456(01)00206-X
Coskun M, Kayis T, Gulsu E, Alp E (2020) Effects of selenium and vitamin E on enzymatic, biochemical, and immunological biomarkers in Galleria mellonella L. Sci Rep 10:9953. https://doi.org/10.1038/s41598-020-67072-9
Desai AV, Siddhapara MR, Patel PK, Prajapati AP (2019) Biology of greater wax moth, Galleria mellonella L. on artificial diet. J Exp Zool India 22 (2):1267–1272
Emre I, Kayis T, Coskun M, Dursun O, Cogun HY (2013) Changes in antioxidative enzyme activity, glycogen, lipid, protein, and malondialdehyde content in cadmium-treated Galleria mellonella larvae. Ann Entomol Soc Am 106 (3):371–377. https://doi.org/10.1603/AN12137
Eskin A, Öztürk Ş, Körükçü M (2019) Determination of the acute toxic effects of zinc oxide nanoparticles (ZnO NPs) in total hemocytes counts of Galleria mellonella (Lepidoptera: Pyralidae) with two different methods. Ecotoxicology 28 (7):801–808. https://doi.org/10.1007/s10646-019-02078-2
Falco JRP, Hashimoto JH, Fermino F, Toledo VAA (2010) Toxicity of thiamethoxam, behavioral effects and alterations in chromatin of Apis mellifera L., 1758 (Hymenoptera; Apidae). Res J Agri Biol Sci 6 (6):823–828
Farkas A, Salanki J, Varanka I (2000) Heavy metal concentrations in fish of Lake Balaton. Lake Reserv Manag 5 (4):271–279. https://doi.org/10.1046/j.1440-1770.2000.00127.x
Finke MD (2008) Nutrient content of insects. In: Capineira JL (ed) Encyclopedia of entomology, 2nd edn. Springer, Netherlands, pp 2623–2646
Fornazier RF, Ferreira RR, Pereira GJG, Molina SMG, Smith RJ, Lea PJ, Azevedo RA (2002) Cadmium stress in sugar zane callus cultures: effect on antioxidant enzymes. Plant Cell Tissue Organ Cult 71 (2):125–131. https://doi.org/10.1023/A:1019917705111
Gao HH, Zhao HY, Du C, Deng MM, Du EX, Hu ZQ, Hu XS (2012) Life table evaluation of survival and reproduction of the aphid, Sitobion avenae, exposed to cadmium. Int J Insect Sci 12:44. https://doi.org/10.1673/031.012.4401
Giglio A, Brandmayr P (2017) Structural and functional alterations in malpighian tubules as biomarkers of environmental pollution: synopsis and prospective. J Appl Toxicol 37 (8):889–894. https://doi.org/10.1002/jat.3454
Gramigni E et al (2013) Ants as bioaccumulators of metals from soils: Body content and tissue-specific distribution of metals in the ant Crematogaster scutellaris. Eur J Soil Biol 58:24–31. https://doi.org/10.1016/j.ejsobi.2013.05.006
Hayford BL, Ferrington LC (2005) Biological assessment of Cannon Creek, Missouri by use of emerging Chironomidae (Insecta: Diptera). J Kansas Entomol Soc 78 (2):89–99. https://doi.org/10.2317/0405.20.1
Hogervorst PA, Wäckers FL, Romeis (2007) Effects of honeydew sugar composition on the longevity of Aphidius ervi. Entomol Exp Appl 122 (3):223–232. https://doi.org/10.1111/j.1570-7458.2006.00505.x
Ilahi I, Yousafzai AM, Ali H (2020) Effect of Pb, Cd and Cu on survival and development of Culex quinquefasciatus (Diptera: Culicidae). Chem Ecol 36 (3):205–219. https://doi.org/10.1080/02757540.2020.1723558
Javed M, Usmani N (2015) Stress response of biomolecules (carbohydrate, protein and lipid profiles) in fish Channa punctatus inhabiting river polluted by Thermal Power Plant effluent. Saudi J Biol Sci 22 (2):237–242. https://doi.org/10.1016/j.sjbs.2014.09.021
Jensen P, Trumble JT (2003) Ecological consequences of bioavailability of metals and metalloids in insects. Recent Res Devel Entomol 4:1–17
Jeuniaux C (1971) Hemolymph-Arthropoda. In: Florkin M, Scheer BT (eds) Chemical Zoology, vol 6. Part B. Academic press, New York, pp 64–112
Jones JC (1962) Current concepts concerning insect hemocytes. Am Zool 2:209–246
Kabata-Pendias A (2010) Trace Elements in Soils and Plants, 4th edn. CRC Press. https://doi.org/10.1201/b10158
Kayis T, Coskun M, Dursun O, Emre I (2015) Alterations in antioxidant enzyme activity, lipid peroxidation and ion balance induced by dichlorvos in Galleria mellonella L. Ann Entomol Soc Am 108 (4):570–574. https://doi.org/10.1093/aesa/sav038
Khalil M, Gad N, Ahmed NA, Mostafa SS (2017) Antioxidant defense system alternations in fish as a bio-indicator of environmental pollution. Egypt J Aquat Biol Fish 21 (3):11–28. https://doi.org/10.21608/EJABF.2017.3536
Kim H, Lim B, Kim BD, Lee YM (2016) Effects of heavy metals on transcription and enzyme activity of Na+/K+-ATPase in the monogonont rotifer, Brachionus koreanus. J Toxicol Environ Health 8:128–134. https://doi.org/10.1007/s13530-016-0270-4
Kurihara Y, Shimazu T, Wago H (1992) Classification of hemocytes in the common cutworm, Spodoptera litura (Lepidoptera: Noctuidae) II. Possible roles of granular plasmatocytes and enocytoids in the cellular defense reactions. Appl Entomol Zool 27 (2):237–242. https://doi.org/10.1303/aez.27.237
Kurt D, Kayis T (2015) Effects of the pyrethroid insecticide deltamethrin on the hemocytes of Galleria mellonella. Turk J Zool 39 (3):452–457. https://doi.org/10.3906/zoo-1405-66
Lagadic L, Caquet T, Ramade F (1994) The role of biomarkers in environmental assessment. Invertebrate populations communities Ecotoxicology 3:193–208. https://doi.org/10.1007/BF00117084
Li Z et al (2011) Antioxidant responses and plasma biochemical characteristics in the freshwater rainbow trout, Oncorhynchus mykiss, after acute exposure to the fungicide propiconazole. Czech J Anim Sci 56:61–69. https://doi.org/10.17221/35/2010-CJAS
Lorenz S, Francese M, Smith VJ, Ferrero EA (2001) Heavy metals affect the circulating haemocyte number in the shrimp Palaemon elegans. Fish Shellfish Immunol 11 (6):459–472. https://doi.org/10.1006/fsim.2000.0321
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Maryanski M, Kramarz P, Laskowski R, Niklinska M (2002) Decreased energetic reserves, Morphological changes and accumulation of metals in Carabid Beetles (Poecilus cupreus L.) exposed to Zinc or Cadmium contaminated food. Ecotoxicology 11:127–139. https://doi.org/10.1023/A:1014425113481
Masindi V, Muedi KL (2018) Environmental contamination by heavy metals. Heavy Metals InTech. https://doi.org/10.5772/intechopen.76082
Migula P, Łaszczyca P, Augustyniak M, Wilczek G, Rozpędek K, Kafel A, Wołoszyn M (2004) Antioxidative defence enzymes in beetles from metal pollution gradient. Biologia 59 (5):645–654
Mnkandla SM, Basopo N, Siwela A (2019) The Effect of Persistent Heavy Metal Exposure on Some Antioxidant Enzyme Activities and Lipid Peroxidation of the Freshwater snail, Lymnaea natalensis. Bull Environ Contam Toxicol 103 (4):551–558. https://doi.org/10.1007/s00128-019-02693-z
Mozdzer TJ, Kramarz P, Piśkiewicz A, Niklińska M (2003) Effects of cadmium and zinc on larval growth and survival in the ground beetle, Pterostichus oblongopunctatus. Environ Int 28 (8):737–742. https://doi.org/10.1016/S0160-4120(02)00107-1
Muramoto S (1983) Elimination of copper from Cu-contaminated fish by long-term exposure to EDTA and freshwater. J Environ Sci Health A 18 (3):455–461. https://doi.org/10.1080/10934528309375113
Nadgorska-Socha A, Kafel A, Kandziora-Ciupa M, Gospodarek J, Zawisza-Raszka A (2013) Accumulation of heavy metals and antioxidant responses in Vicia faba plants grown on monometallic contaminated soil. Environ Sci Pollut Res 20 (2):1124–1134. https://doi.org/10.1007/s11356-012-1191-7
Olson D, Fadamiro H, Lundgren J, Heimpel GE (2000) Effects of sugar feeding on carbohydrate and lipid metabolism in a parasitoid wasp. Physiol Entomol 25:17–26. https://doi.org/10.1046/j.1365-3032.2000.00155
Osuala FI, Otitoloju AA, Igwo-Ezikpe MN (2013) Sublethal effects of cadmium, manganese, lead, zinc and iron on the plasma electrolytes regulation of mice. Mus Musculus Afr J Environ Sci Technol 7 (9):925–931. https://doi.org/10.5897/AJEST2013.1555
Patil HZ, Mahale PN, Kulkarni AB (2011) Alterations in the Biochemical Content in the Foot of a Freshwater Snail, Indoplanorbis exustus Exposed to Heavy Metals. J Exp Sci 2 (3):12–14
Piek T, Njio KD (1979) Morphology and electrochemistry of insect muscle fibre membrane. Adv Insect Phys 14:185–250. https://doi.org/10.1016/S0065-2806(08)60053-3
Rajak P, Dutta M, Roy S (2015) Altered differential hemocyte count in 3rd instar larvae of Drosophila melanogaster as a response to chronic exposure of Acephate. Interdiscip Toxicol 8 (2):84–88. https://doi.org/10.1515/intox-2015-0013
Safaee S, Fereidoni M, Shahri NM, Haddad F, Mirshamsi O (2014) Effect of lead on the development of Drosophila melanogaster. Period Biol 116 (3):259–265
Salema LH, Alwan MJ, Afaf A (2014) Antioxidative and antigenotoxic effects against cytotoxicity of thiamethoxam on mice. Int J Adv Res 2 (10):507–511
Sanchez W, Palluel O, Meunier L, Coquery M, Porcher JM, Selim A (2005) Copper induced oxidative stress in three-spined stickleback: relationship with hepatic metal levels. Environ Toxicol Pharmacol 19 (1):177–183. https://doi.org/10.1016/j.etap.2004.07.003
Sang W, Xu J, Bashir MH, Ali S (2018) Developmental responses of Cryptolaemus montrouzieri to heavy metals transferred across multi-trophic food chain. Chemosphere 205:690–697. https://doi.org/10.1016/j.chemosphere.2018.02.073
Sowa G, Skalski T (2019) Effects of chronic metal exposure on the morphology of beetles species representing different ecological niches. Bull Environ Contam Toxicol 102:191–197. https://doi.org/10.1007/s00128-018-02532-7
Stone D, Jepson P, Kramarz P, Laskowski R (2001) Time to death response in carabid beetles exposed to multiple stressors along a gradient of heavy metal pollution. Environ Pollut 113 (2):239–244. https://doi.org/10.1016/S0269-7491(00)00134-2
Sun HJ, Rathinasabapathi B, Wu B, Luo J, Pu LP, Ma LQ (2014) Arsenic and selenium toxicity and their interactive effects in humans. Environ Inter 69:148–158. https://doi.org/10.1016/j.envint.2014.04.019
Sun Y, Oberley LW, Li YA (1988) Simple method for clinical assay of superoxide dismutase. Clin Chem 34 (3):497–500
Talarico F et al (2014) Effects of metal pollution on survival and physiological responses in Carabus (Chaetocarabus) lefebvrei (Coleoptera, Carabidae). Eur J Soil Biol 61:80–89. https://doi.org/10.1016/j.ejsobi.2014.02.003
Tewari S, Bajpai S, Tripathi M (2019) Effect of cadmium on glycogen content in muscle, liver, gill and kidney tissues of freshwater fish Channa punctatus (Bloch). J Nat Appl Sci 11 (2):575–580. https://doi.org/10.31018/jans.v11i2.2115
Tkachenko H, Kurhaluk N, Grudniewska J, Andriichuk A (2014) Tissue-specific responses of oxidative stress biomarkers and antioxidant defenses in rainbow trout Oncorhynchus mykiss during a vaccination against furunculosis. Fish Physiol Biochem 40 (4):1289–1300. https://doi.org/10.1007/s10695-014-9924-9
Topkara EF, Yanar O (2019) Synergistic effects of some secondary compounds combined with some heavy metals on Hyphantria cunea Drury (Lepidoptera: Arctiidae) larvae. J Entomol Res Soc 21 (2):213–223
Tsai CJ, Loh JM, Proft T (2016) Galleria mellonella infection models for the study of bacterial diseases and for antimicrobial drug testing. Virulence 7 (3):214–229. https://doi.org/10.1080/21505594.2015.1135289
Tüzen M (2003) Determination of heavy metals in fish samples of the middle black sea (Turkey) by graphite furnace atomic absorption spectrometry. Food Chem 80 (1):119–123. https://doi.org/10.1016/S0308-8146(02)00264-9
Van Handel E (1985) Rapid determination of total lipids in mosquitoes. J Am Mosq Control Assoc 1 (3):302–304
Van Handel E (1985) Rapid determination of glycogen and sugars in mosquitoes. J Am Mosq Control Assoc 1 (3):299–301
Walker CH, Hopkin SP, Sibly RM, Peakall DB (2001) Principles of ecotoxicology, 4th edn. Taylor & Francis, London
Wu G, Yi Y (2015) Effects of dietary heavy metals on the immune and antioxidant systems of Galleria mellonella larvae. Comp Biochem Physiol Part C 167:131–139. https://doi.org/10.1016/j.cbpc.2014.10.004
Wu GX, Gao X, Ye GY, Li K, Hu C, Cheng JA (2009) Ultrastructural alterations in midgut and Malpighian tubules of Boettcherisca peregrina exposure to cadmium and copper. Ecotoxicol Environ Safe 72 (4):1137–1147. https://doi.org/10.1016/j.ecoenv.2008.02.017
Wu YY, Zhou T, Wang Q, Dai PL, Xu SF, Jia HR, Wang X (2015) Programmed cell death in the honey bee (Apis mellifera) (Hymenoptera: Apidae) worker brain induced by imidacloprid. J Econ Entomol 108 (4):1486–1494. https://doi.org/10.1093/jee/tov146
Yang X, Feng Y, He Z, Stoffella PJ (2005) Molecular mechanisms of heavy metal hyperaccumulation and phytoremediation. J Trace Elem Med Biol 18 (4):339–353. https://doi.org/10.1016/j.jtemb.2005.02.007
Yucel MS, Kayis T (2019) Imidacloprid induced alterations in oxidative stress, biochemical, genotoxic, and immunotoxic biomarkers in non-mammalian model organism Galleria mellonella L. (Lepidoptera: Pyralidae). J Environ Sci Health B 54 (1):27–34. https://doi.org/10.1080/03601234.2018.1530545
Zhu Q, He Y, Yao J, Liu Y, Tao L, Huang Q (2012) Effects of sublethal concentrations of the chitin synthesis inhibitor, hexaflumuron, on the development and hemolymph physiology of the cutworm, Spodoptera litura. J Insect Sci 12:27. https://doi.org/10.1673/031.012.2701
Funding
This research was supported by the Research Fund of Adiyaman University, Project No.: FEFBAP2009-6.
Author information
Authors and Affiliations
Contributions
MC conceived and supervised the study. MC, TK, MY, OD and IE performed the experimental work. MC, TK and IE analyzed the data. MC and TK wrote and revised the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflict of interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Coskun, M., Kayis, T., Yilmaz, M. et al. Copper and zinc impact on stress biomarkers and growth parameters in a model organism, Galleria mellonella larvae. Biometals 34, 1263–1273 (2021). https://doi.org/10.1007/s10534-021-00341-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-021-00341-w