Abstract
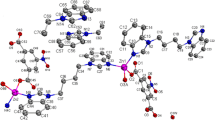
Bithiazole is considered as a domain of bleomycin which is countered as a chemotherapeutic drug to treat many types of cancer. Hence, the trend towards the design of model compounds containing the bithiazole moiety is increased recently. In this regard, the 2,2′-diamino-5,5′-dimethyl-4,4′-bithiazole organic ligand (L) is synthesized via a condensation reaction of 3,4-diaminobenzophenone with 2,3-butanedione monoxime. Moreover, its zinc(II) complex [Zn(L)3](NO3)2CH3OH (ZnL3) is synthesized and subsequently characterized by various analytical techniques, including Infrared spectroscopy, proton nuclear magnetic resonance, carbon nuclear magnetic resonance, thermal gravimetric-differential thermal analysis, and single crystal X-ray diffraction. Based on single crystal X-ray data, the ZnL3 complex belongs to the triclinic system with the space group P-1. In ZnL3, one of L ligands acts as a monodentate ligand to form the five-coordinated Zn(II) complex, giving a distorted trigonal bipyramidal geometry around the metal atom. Finally, UV-Vis and fluorescence spectroscopy is used to investigate the photophysical properties of the material explained via density functional theory.









Similar content being viewed by others
REFERENCES
A. Allawzi, H. Elajaili, E. F. Redente, and E. Nozik-Grayck. Curr. Opin. Toxicol., 2019, 13, 68.
J. P. Hénichart, J. L. Bernier, N. Helbecque, and R. Houssin. Nucleic Acids Res., 1985, 13, 6703.
S. A. Kane, A. Natrajan, and S. M. Hecht. J. Biol. Chem., 1994, 269, 10899.
J. B. Shipley and S. M. Hecht. Chem. Res. Toxicol., 1988, 1, 25.
P. E. Nielsen. Bioconjugate Chem., 1991, 2, 1.
F. Arjmand, Z. Afsan, S. Sharma, S. Parveen, I. Yousuf, S. Sartaj, H. R. Siddique, and S. Tabassum. Coord. Chem. Rev., 2019, 387, 47-59.
G. Schneider. Nat. Rev. Drug Discovery, 2018, 17, 97.
W. Lin, W. Sun, J. Yang, and Z. Shen. Mater. Chem. Phys., 2008, 112, 617.
T. Matsubara and K. Hirao. J. Mol. Struct.: THEOCHEM, 2002, 581, 203.
E. Ho. J. Nutr. Biochem., 2004, 15, 572.
M. Stefanidou, C. Maravelias, A. Dona, and C. Spiliopoulou. Arch. Toxicol., 2006, 80, 1.
V. Milacic, D. Chen, L. Giovagnini, A. Diez, D. Fregona, and Q. P. Dou. Toxicol. Appl. Pharm., 2008, 231, 24.
R. Lopez-Garzon, P. Arranz-Mascarós, M. Godino-Salido, M. Gutiérrez-Valero, A. Pérez-Cadenas, J. Cobo-Domingo, and J. Moreno. Inorg. Chim. Acta, 2000, 308, 59.
J. F. Da Silva and R. J. P. Williams. The Biological Chemistry of the Elements: The Inorganic Chemistry of Life. Oxford University Press, 2001.
K. D. Karlin. Science, 1993, 261, 701.
A. S. Lipton, R. W. Heck, and P. D. Ellis. J. Am. Chem. Soc., 2004, 126, 4735.
B. L. Vallee and A. Galdes. Adv. Enzymol. Relat. Areas Mol. Biol., 1984, 56, 283.
I. Bertini, C. Luchinat, W. Maret, and M. Zeppezauer. Progress in Inorganic Biochemistry and Biophysics. Birkhäuser: Boston, 1986, Vol. 1: Zinc Enzymes.
A. Mahjoub, A. Morsali, Z. Talaei, and A. Hosseinian. Z. Kristallogr. - New Cryst. Struct., 2005, 220, 47.
S. Sen, S. Mitra, P. Kundu, M. K. Saha, C. Krüger, and J. Bruckmann. Polyhedron, 1997, 16, 2475.
E. Caron, C. M. Brown, D. Hean, and M. O. Wolf. Dalton Trans., 2019, 48, 1263.
J. Weng, L. M. Jiang, W. L. Sun, Z. Q. Shen, and S. Q. Liang. Polymer, 2001, 42, 5491.
L. Jiang, W. Sun, J. Weng, and Z. Shen. Polymer, 2002, 43, 1563.
L. M. Engelhardt, D. L. Kepert, J. M. Patrick, and A. White. Aust. J. Chem., 1989, 42, 329.
Y. Tian, P. Yang, Q. Li, and S. Liu. J. Coord. Chem. 1997, 41, 223.
W.-Y. Wong, K.-H. Choi, G.-L. Lu, and Z. Lin. Organometallics, 2002, 21, 4475.
W. Y. Wong, G. J. Zhou, Z. He, K. Y. Cheung, A. M. C. Ng, A. B. Djurišić, and W. K. Chan. Macromol. Chem. Phys., 2008, 209, 1319.
W. Y. Wong, S. M. Chan, K. H. Choi, K. W. Cheah, and W. K. Chan. Macromol. Rapid Commun., 2000, 21, 453.
W. Sun, X. Gao, and F. Lu. J. Appl. Polym. Sci., 1997, 64, 2309.
L. Jiang and W. Sun. Polym. Adv. Technol., 2005, 16, 646.
L. Liu, Y. W. Lam, and W. Y. Wong. J. Organomet. Chem., 2006, 691, 1092.
A. Hosseinian, A. Mahjoub, and A. Morsali. Z. Kristallogr. - New Cryst. Struct., 2005, 220, 439.
G. Sheldrick. SHELXL-97, Program for the Refinement of Crystal Structures. University of Göttingen: Göttingen, Germany, 1997.
G. Sheldrick. SHELXTL V. 5.10, Structure determination software suite. Bruker AXS: Madison, Wisconsin, USA, 1998.
M. B. Ferrari, G. G. Fava, G. Pelosi, and P. Tarasconi. Polyhedron, 2000, 19, 1895.
Y. Zhang and H. Liu. Inorg. Chim. Acta, 2016, 450, 112.
A. Abolhosseini Sh., A. Mahjoub, M. Eslami-Moghadam, and H. Fakhri. J. Mol. Struct., 2014, 1076, 568.
A. Hosseinian, S. Jabbari, H. R. Rahimipour, and A. R. Mahjoub. J. Mol. Struct., 2012, 1028, 215.
Y.-Z. Zheng, D.-F. Chen, Y. Zhou, R. Guo, Q. Liang, and Z.-M. Fu. J. Mol. Struct., 2018, 1164 , 70.
T. S. Al-Harthy, M. S. Shongwe, J. Husband, R. Stoll, K. Merz, and R. J. Abdel-Jalil. J. Mol. Struct., 2019, 1176, 614.
A. Hosseinian, H. R. Rahimipour, H. Haddadi, A. A. Ashkarran, and A. R. Mahjoub. J. Mol. Struct., 2014, 1074, 673.
V. Amani, R. Alizadeh, H. S. Alavije, S. F. Heydari, and M. Abafat. J. Mol. Struct., 2017, 1142, 92.
J.-C. G. Bünzli and D. Wessner. Coord. Chem. Rev., 1984, 60, 191.
W. Carnall, S. Siegel, J. Ferraro, B. Tani, and E. Gebert. Inorg. Chem., 1973, 12, 560.
M. Eslami Moghadam, A. Divsalar, A. Abolhosseini Shahrnoy, and A. A. Saboury. J. Biomol. Struct. Dyn., 2016, 34, 1751.
D. Sellmann, K. Nakamoto. Infrared Spectra of Inorganic and Coordination Compounds. John Wiley & Sons: New York, London, Sydney, Toronto, 1970.
A. Hosseinian and A. R. Mahjoub. J. Mol. Struct., 2011, 985, 270.
A. Hosseinian, S. Jabbari, A. R. Mahjoub, and M. Movahedi. J. Coord. Chem., 2012, 65, 2623.
A. Hosseinian and A. R. Mahjoub. J. Coord. Chem., 2010, 63, 4245.
A. P. Lever, E. Mantovani, and B. Ramaswamy. Can. J. Chem., 1971, 49, 1957.
Funding
Support of this investigation by Iran National Science Foundation (grant No. 90001707) is gratefully acknowledged. We also thank the Institute of Organoelement Compounds of the Russian Academy of Science for determining the crystal structure by X-ray crystallography.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interests.
Additional information
Text © The Author(s), 2021, published in Zhurnal Strukturnoi Khimii, 2021, Vol. 62, No. 7, pp. 1121-1133.https://doi.org/10.26902/JSC_id74434
Rights and permissions
About this article
Cite this article
Chamack, M., Hosseinian, A. & Khazaee, Z. SYNTHESIS, CRYSTAL STRUCTURE, AND DFT INSIGHT OF A NEW TRIGONAL BIPYRAMIDAL ZINC(II) COMPLEX. J Struct Chem 62, 1043–1055 (2021). https://doi.org/10.1134/S0022476621070088
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476621070088