Abstract
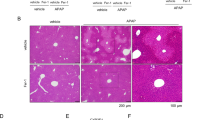
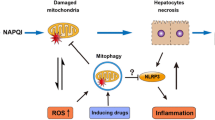
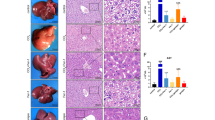
Acetaminophen (APAP) overdose is a common cause of drug-induced liver injury (DILI). Ferroptosis has been recently implicated in APAP-induced liver injury (AILI). However, the functional role and underlying mechanisms of mitochondria in APAP-induced ferroptosis are unclear. In this study, the voltage-dependent anion channel (VDAC) oligomerization inhibitor VBIT-12 and ferroptosis inhibitors were injected via tail vein in APAP-injured mice. Targeted metabolomics and untargeted lipidomic analyses were utilized to explore underlying mechanisms of APAP-induced mitochondrial dysfunction and subsequent ferroptosis. As a result, APAP overdose led to characteristic changes generally observed in ferroptosis. The use of ferroptosis inhibitor ferrostatin-1 (or UAMC3203) and iron chelator deferoxamine further confirmed that ferroptosis was responsible for AILI. Mitochondrial dysfunction, which is associated with the tricarboxylic acid cycle and fatty acid β-oxidation suppression, may drive APAP-induced ferroptosis in hepatocytes. APAP overdose induced VDAC1 oligomerization in hepatocytes, and protecting mitochondria via VBIT-12 alleviated APAP-induced ferroptosis. Ceramide and cardiolipin levels were increased via UAMC3203 or VBIT-12 in APAP-induced ferroptosis in hepatocytes. Knockdown of Smpd1 and Taz expression responsible for ceramide and cardiolipin synthesis, respectively, aggravated APAP-induced mitochondrial dysfunction and ferroptosis in hepatocytes, whereas Taz overexpression protected against these processes. By immunohistochemical staining, we found that levels of 4-hydroxynonenal (4-HNE) protein adducts were increased in the liver biopsy samples of patients with DILI compared to that in those of patients with autoimmune liver disease, chronic viral hepatitis B, and non-alcoholic fatty liver disease (NAFLD). In summary, protecting mitochondria via inhibiting VDAC1 oligomerization attenuated hepatocyte ferroptosis by restoring ceramide and cardiolipin content in AILI.
Graphical abstract









Similar content being viewed by others
Abbreviations
- APAP :
-
Acetaminophen
- DILI :
-
Drug-induced liver injury
- 4-HNE :
-
4-Hydroxynonenal
- VDAC1 :
-
Voltage-dependent anion channel 1
- AILI :
-
APAP-induced liver injury
- NAPQI :
-
N-Acetyl-p-benzoquinone imine
- GSH :
-
Glutathione
- ROS :
-
Reactive oxygen species
- AIH :
-
Autoimmune liver disease
- CHB :
-
Chronic viral hepatitis B
- NAFLD :
-
Non-alcoholic fatty liver disease steatosis
- ALD :
-
Alcoholic liver disease
- DFO :
-
Deferoxamine
- siRNA :
-
Small interfering RNA
- ALT :
-
Alanine aminotransferase
- AST :
-
Aspartate aminotransferase
- H&E :
-
Hematoxylin and eosin
- MMP :
-
Mitochondrial membrane potential
- MDA :
-
Malondialdehyde
- OCR :
-
Oxygen consumption rate
- MOM :
-
Mitochondrial outer membrane
- BSA :
-
Bovine serum albumin
- PA :
-
Palmitic acid
- LC-MS :
-
Liquid chromatography-mass spectrometry
- H-SCORE :
-
Histological scores
- ALP :
-
Alkaline phosphatase
- RNA-seq :
-
RNA sequencing
- GSEA :
-
Gene set enrichment analysis
- Fer-1 :
-
Ferrostatin-1
- TUNEL :
-
Terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling
- PMHs :
-
Primary mouse hepatocytes
- DCFH-DA :
-
Dichloro-dihydro-fluorescein diacetate
- TCA :
-
Tricarboxylic acid
- ETC :
-
Electron transport chain
- CL :
-
Cardiolipin
- CER :
-
Ceramide
- Taz :
-
Tafazzin
References
Andrade RJ, Chalasani N, Bjornsson ES, Suzuki A, Kullak-Ublick GA, Watkins PB, et al. Drug-induced liver injury. Nat Rev Dis Primers. 2019;5(1):58. https://doi.org/10.1038/s41572-019-0105-0.
Baba Y, Higa JK, Shimada BK, Horiuchi KM, Suhara T, Kobayashi M, Woo JD, Aoyagi H, Marh KS, Kitaoka H, Matsui T. Protective effects of the mechanistic target of rapamycin against excess iron and ferroptosis in cardiomyocytes. Am J Physiol Heart Circ Physiol. 2018;314(3):H659–H68. https://doi.org/10.1152/ajpheart.00452.2017.
Baulies A, Ribas V, Nunez S, Torres S, Alarcon-Vila C, Martinez L, et al. Lysosomal cholesterol accumulation sensitizes to acetaminophen hepatotoxicity by impairing mitophagy. Sci Rep. 2015;5:18017. https://doi.org/10.1038/srep18017.
Ben-Hail D, Begas-Shvartz R, Shalev M, Shteinfer-Kuzmine A, Gruzman A, Reina S, de Pinto V, Shoshan-Barmatz V. Novel compounds targeting the mitochondrial protein vdac1 inhibit apoptosis and protect against mitochondrial dysfunction. J Biol Chem. 2016;291(48):24986–5003. https://doi.org/10.1074/jbc.M116.744284.
Bjornsson ES, Bergmann OM, Bjornsson HK, Kvaran RB, Olafsson S. Incidence, presentation, and outcomes in patients with drug-induced liver injury in the general population of iceland. Gastroenterology. 2013;144(7):1419–25, 25 e1-3; quiz e19-20. https://doi.org/10.1053/j.gastro.2013.02.006.
Bock FJ, Tait SWG. Mitochondria as multifaceted regulators of cell death. Nat Rev Mol Cell Biol. 2020;21(2):85–100. https://doi.org/10.1038/s41580-019-0173-8.
Chen C, Hennig GE, Whiteley HE, Corton JC, Manautou JE. Peroxisome proliferator-activated receptor alpha-null mice lack resistance to acetaminophen hepatotoxicity following clofibrate exposure. Toxicol Sci. 2000;57(2):338–44. https://doi.org/10.1093/toxsci/57.2.338.
Chen C, Krausz KW, Shah YM, Idle JR, Gonzalez FJ. Serum metabolomics reveals irreversible inhibition of fatty acid beta-oxidation through the suppression of pparalpha activation as a contributing mechanism of acetaminophen-induced hepatotoxicity. Chem Res Toxicol. 2009;22(4):699–707. https://doi.org/10.1021/tx800464q.
Church RJ, Kullak-Ublick GA, Aubrecht J, Bonkovsky HL, Chalasani N, Fontana RJ, Goepfert JC, Hackman F, King NMP, Kirby S, Kirby P, Marcinak J, Ormarsdottir S, Schomaker SJ, Schuppe-Koistinen I, Wolenski F, Arber N, Merz M, Sauer JM, et al. Candidate biomarkers for the diagnosis and prognosis of drug-induced liver injury: An international collaborative effort. Hepatology. 2019;69(2):760–73. https://doi.org/10.1002/hep.29802.
Devisscher L, Van Coillie S, Hofmans S, Van Rompaey D, Goossens K, Meul E, et al. Discovery of novel, drug-like ferroptosis inhibitors with in vivo efficacy. J Med Chem. 2018;61(22):10126–40. https://doi.org/10.1021/acs.jmedchem.8b01299.
Du K, Farhood A, Jaeschke H. Mitochondria-targeted antioxidant mito-tempo protects against acetaminophen hepatotoxicity. Arch Toxicol. 2017;91(2):761–73. https://doi.org/10.1007/s00204-016-1692-0.
Du K, Ramachandran A, Weemhoff JL, Woolbright BL, Jaeschke AH, Chao X, et al. Mito-tempo protects against acute liver injury but induces limited secondary apoptosis during the late phase of acetaminophen hepatotoxicity. Arch Toxicol. 2019;93(1):163–78. https://doi.org/10.1007/s00204-018-2331-8.
Ettel M, Gonzalez GA, Gera S, Eze O, Sigal S, Park JS, Xu R. Frequency and pathological characteristics of drug-induced liver injury in a tertiary medical center. Hum Pathol. 2017;68:92–8. https://doi.org/10.1016/j.humpath.2017.08.029.
European Association for the Study of the Liver. Electronic address eee, Clinical Practice Guideline Panel C, Panel m, representative EGB. Easl clinical practice guidelines: drug-induced liver injury. J Hepatol. 2019;70(6):1222–61. https://doi.org/10.1016/j.jhep.2019.02.014.
Flatmark T, Romslo I. Energy-dependent accumulation of iron by isolated rat liver mitochondria. Requirement of reducing equivalents and evidence for a unidirectional flux of fe(ii) across the inner membrane. J Biol Chem. 1975;250(16):6433–8.
Hassan A, Fontana RJ. The diagnosis and management of idiosyncratic drug-induced liver injury. Liver Int. 2019;39(1):31–41. https://doi.org/10.1111/liv.13931.
Hayashi N, George J, Takeuchi M, Fukumura A, Toshikuni N, Arisawa T, Tsutsumi M. Acetaldehyde-derived advanced glycation end-products promote alcoholic liver disease. PLoS One. 2013;8(7):e70034. https://doi.org/10.1371/journal.pone.0070034.
Hernandez-Corbacho MJ, Salama MF, Canals D, Senkal CE, Obeid LM. Sphingolipids in mitochondria. Biochim Biophys Acta Mol Cell Biol Lipids. 2017;1862(1):56–68. https://doi.org/10.1016/j.bbalip.2016.09.019.
Hong W, Guo F, Yang M, Xu D, Zhuang Z, Niu B, Bai Q, Li X. Hydroxysteroid sulfotransferase 2b1 affects gastric epithelial function and carcinogenesis induced by a carcinogenic agent. Lipids Health Dis. 2019;18(1):203. https://doi.org/10.1186/s12944-019-1149-6.
Hu J, Lemasters JJ. Suppression of iron mobilization from lysosomes to mitochondria attenuates liver injury after acetaminophen overdose in vivo in mice: protection by minocycline. Toxicol Appl Pharmacol. 2020;392:114930. https://doi.org/10.1016/j.taap.2020.114930.
Jaeschke H. Mitochondrial dysfunction as a mechanism of drug-induced hepatotoxicity: current understanding and future perspectives. Journal of Clinical and Translational Research. 2018;4(1):75–100. https://doi.org/10.18053/jctres.04.201801.005.
Jaeschke H, Xie Y, McGill MR. Acetaminophen-induced liver injury: from animal models to humans. J Clin Transl Hepatol. 2014;2(3):153–61. https://doi.org/10.14218/JCTH.2014.00014.
Jaeschke H, Adelusi OB, Ramachandran A. Ferroptosis and acetaminophen hepatotoxicity - are we going down another rabbit hole? Gene Expr. 2021;20:169–78. https://doi.org/10.3727/105221621x16104581979144.
Jenkins RW, Canals D, Idkowiak-Baldys J, Simbari F, Roddy P, Perry DM, Kitatani K, Luberto C, Hannun YA. Regulated secretion of acid sphingomyelinase: implications for selectivity of ceramide formation. J Biol Chem. 2010;285(46):35706–18. https://doi.org/10.1074/jbc.M110.125609.
Jollow DJ, Mitchell JR, Potter WZ, Davis DC, Gillette JR, Brodie BB. Acetaminophen-induced hepatic necrosis .2. Role of covalent binding in-vivo. J Pharmacol Exp Ther. 1973;187(1):195–202.
Kim J, Gupta R, Blanco LP, Yang S, Shteinfer-Kuzmine A, Wang K, Zhu J, Yoon HE, Wang X, Kerkhofs M, Kang H, Brown AL, Park SJ, Xu X, Zandee van Rilland E, Kim MK, Cohen JI, Kaplan MJ, Shoshan-Barmatz V, Chung JH. Vdac oligomers form mitochondrial pores to release mtdna fragments and promote lupus-like disease. Science. 2019;366(6472):1531–6. https://doi.org/10.1126/science.aav4011.
Kullak-Ublick GA, Andrade RJ, Merz M, End P, Benesic A, Gerbes AL, Aithal GP. Drug-induced liver injury: recent advances in diagnosis and risk assessment. Gut. 2017;66(6):1154–64. https://doi.org/10.1136/gutjnl-2016-313369.
Lange H, Kispal G, Lill R. Mechanism of iron transport to the site of heme synthesis inside yeast mitochondria. J Biol Chem. 1999;274(27):18989–96. https://doi.org/10.1074/jbc.274.27.18989.
Lewis JH. Diagnosis: liver biopsy differentiates dili from autoimmune hepatitis. Nat Rev Gastroenterol Hepatol. 2011;8(10):540–2. https://doi.org/10.1038/nrgastro.2011.140.
Li C, Ming Y, Hong W, Tang Y, Lei X, Li X, Mao Y. Comparison of hepatic transcriptome profiling between acute liver injury and acute liver failure induced by acetaminophen in mice. Toxicol Lett. 2018;283:69–76. https://doi.org/10.1016/j.toxlet.2017.11.020.
Lipper CH, Stofleth JT, Bai F, Sohn YS, Roy S, Mittler R, Nechushtai R, Onuchic JN, Jennings PA. Redox-dependent gating of vdac by mitoneet. Proc Natl Acad Sci U S A. 2019;116(40):19924–9. https://doi.org/10.1073/pnas.1908271116.
Lorincz T, Jemnitz K, Kardon T, Mandl J, Szarka A. Ferroptosis is involved in acetaminophen induced cell death. Pathol Oncol Res. 2015;21(4):1115–21. https://doi.org/10.1007/s12253-015-9946-3.
Nagakannan P, Islam MI, Karimi-Abdolrezaee S, Eftekharpour E. Inhibition of vdac1 protects against glutamate-induced oxytosis and mitochondrial fragmentation in hippocampal ht22 cells. Cell Mol Neurobiol. 2019;39(1):73–85. https://doi.org/10.1007/s10571-018-0634-1.
Nowaczyk MJ, Whelan D, Hill RE, Clarke JT, Pollitt RJ. Long-chain hydroxydicarboxylic aciduria, carnitine depletion and acetaminophen exposure. J Inherit Metab Dis. 2000;23(2):188–9. https://doi.org/10.1023/a:1005630218986.
Patterson AD, Shah YM, Matsubara T, Krausz KW, Gonzalez FJ. Peroxisome proliferator-activated receptor alpha induction of uncoupling protein 2 protects against acetaminophen-induced liver toxicity. Hepatology. 2012;56(1):281–90. https://doi.org/10.1002/hep.25645.
Paul BT, Manz DH, Torti FM, Torti SV. Mitochondria and iron: Current questions. Expert Rev Hematol. 2017;10(1):65–79. https://doi.org/10.1080/17474086.2016.1268047.
Podszun MC, Chung JY, Ylaya K, Kleiner DE, Hewitt SM, Rotman Y. 4-hne immunohistochemistry and image analysis for detection of lipid peroxidation in human liver samples using vitamin e treatment in nafld as a proof of concept. J Histochem Cytochem. 2020;68(9):635–43. https://doi.org/10.1369/0022155420946402.
Ramachandran A, Jaeschke H. Acetaminophen hepatotoxicity: a mitochondrial perspective. Adv Pharmacol. 2019;85:195–219. https://doi.org/10.1016/bs.apha.2019.01.007.
Ramachandran A, Lebofsky M, Weinman SA, Jaeschke H. The impact of partial manganese superoxide dismutase (sod2)-deficiency on mitochondrial oxidant stress, DNA fragmentation and liver injury during acetaminophen hepatotoxicity. Toxicol Appl Pharmacol. 2011;251(3):226–33. https://doi.org/10.1016/j.taap.2011.01.004.
Ren M, Phoon CKL, Schlame M. Metabolism and function of mitochondrial cardiolipin. Prog Lipid Res. 2014;55:1–16. https://doi.org/10.1016/j.plipres.2014.04.001.
Sakaida I, Kayano K, Wasaki S, Nagatomi A, Matsumura Y, Okita K. Protection against acetaminophen-induced liver injury in vivo by an iron chelator, deferoxamine. Scand J Gastroenterol. 1995;30(1):61–7. https://doi.org/10.3109/00365529509093237.
Schnellmann JG, Pumford NR, Kusewitt DF, Bucci TJ, Hinson JA. Deferoxamine delays the development of the hepatotoxicity of acetaminophen in mice. Toxicol Lett. 1999;106(1):79–88. https://doi.org/10.1016/s0378-4274(99)00021-1.
Sgro C, Clinard F, Ouazir K, Chanay H, Allard C, Guilleminet C, Lenoir C, Lemoine A, Hillon P. Incidence of drug-induced hepatic injuries: a French population-based study. Hepatology. 2002;36(2):451–5. https://doi.org/10.1053/jhep.2002.34857.
Shen T, Liu Y, Shang J, Xie Q, Li J, Yan M, Xu J, Niu J, Liu J, Watkins PB, Aithal GP, Andrade RJ, Dou X, Yao L, Lv F, Wang Q, Li Y, Zhou X, Zhang Y, et al. Incidence and etiology of drug-induced liver injury in mainland china. Gastroenterology. 2019;156(8):2230–41 e11. https://doi.org/10.1053/j.gastro.2019.02.002.
Shoshan-Barmatz V, Nahon-Crystal E, Shteinfer-Kuzmine A, Gupta R. Vdac1, mitochondrial dysfunction, and alzheimer's disease. Pharmacol Res. 2018;131:87–101. https://doi.org/10.1016/j.phrs.2018.03.010.
Stockwell BR, Friedmann Angeli JP, Bayir H, Bush AI, Conrad M, Dixon SJ, Fulda S, Gascón S, Hatzios SK, Kagan VE, Noel K, Jiang X, Linkermann A, Murphy ME, Overholtzer M, Oyagi A, Pagnussat GC, Park J, Ran Q, et al. Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell. 2017;171(2):273–85. https://doi.org/10.1016/j.cell.2017.09.021.
Szczepanek K, Allegood J, Aluri H, Hu Y, Chen Q, Lesnefsky EJ. Acquired deficiency of tafazzin in the adult heart: Impact on mitochondrial function and response to cardiac injury. Biochim Biophys Acta. 2016;1861(4):294–300. https://doi.org/10.1016/j.bbalip.2015.12.004.
Tujios S, Fontana RJ. Mechanisms of drug-induced liver injury: from bedside to bench. Nat Rev Gastroenterol Hepatol. 2011;8(4):202–11. https://doi.org/10.1038/nrgastro.2011.22.
Vergeade A, Bertram CC, Bikineyeva AT, Zackert WE, Zinkel SS, May JM, Dikalov SI, Roberts LJ II, Boutaud O. Cardiolipin fatty acid remodeling regulates mitochondrial function by modifying the electron entry point in the respiratory chain. Mitochondrion. 2016;28:88–95. https://doi.org/10.1016/j.mito.2016.04.002.
Wang Z, Yang X, Chen L, Zhi X, Lu H, Ning Y, Yeong J, Chen S, Yin L, Wang X, Li X. Upregulation of hydroxysteroid sulfotransferase 2b1b promotes hepatic oval cell proliferation by modulating oxysterol-induced lxr activation in a mouse model of liver injury. Arch Toxicol. 2017;91(1):271–87. https://doi.org/10.1007/s00204-016-1693-z.
Wang H, Liu C, Zhao Y, Gao G. Mitochondria regulation in ferroptosis. Eur J Cell Biol. 2020;99(1):151058. https://doi.org/10.1016/j.ejcb.2019.151058.
Wu H, Wang F, Ta N, Zhang T, Gao W. The multifaceted regulation of mitochondria in ferroptosis. Life (Basel, Switzerland). 2021;11(3):222. https://doi.org/10.3390/life11030222.
Yagoda N, von Rechenberg M, Zaganjor E, Bauer AJ, Yang WS, Fridman DJ, Wolpaw AJ, Smukste I, Peltier JM, Boniface JJ, Smith R, Lessnick SL, Sahasrabudhe S, Stockwell BR. Ras-raf-mek-dependent oxidative cell death involving voltage-dependent anion channels. Nature. 2007;447(7146):864–8. https://doi.org/10.1038/nature05859.
Yamada N, Karasawa T, Kimura H, Watanabe S, Komada T, Kamata R, Sampilvanjil A, Ito J, Nakagawa K, Kuwata H, Hara S, Mizuta K, Sakuma Y, Sata N, Takahashi M. Ferroptosis driven by radical oxidation of n-6 polyunsaturated fatty acids mediates acetaminophen-induced acute liver failure. Cell Death Dis. 2020;11(2):144. https://doi.org/10.1038/s41419-020-2334-2.
Yapar K, Kart A, Karapehlivan M, Atakisi O, Tunca R, Erginsoy S, Citil M. Hepatoprotective effect of l-carnitine against acute acetaminophen toxicity in mice. Exp Toxicol Pathol. 2007;59(2):121–8. https://doi.org/10.1016/j.etp.2007.02.009.
Younes M, Siegers CP. The role of iron in the paracetamol- and ccl4-induced lipid peroxidation and hepatotoxicity. Chem Biol Interact. 1985;55(3):327–34. https://doi.org/10.1016/s0009-2797(85)80139-3.
Yu YC, Mao YM, Chen CW, Chen JJ, Chen J, Cong WM, et al. Csh guidelines for the diagnosis and treatment of drug-induced liver injury. Hepatol Int. 2017;11(3):221–41. https://doi.org/10.1007/s12072-017-9793-2.
Availability of data and material
Data and materials used and/or analyzed during the current study are available from the corresponding authors on reasonable request.
Code availability
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (NSFC 31771308, NSFC 81970513, and NSFC 81670524), the Shanghai Municipal Natural Science Foundation (17ZR1401800), the Innovative Research Team of High-level Local Universities in Shanghai, Shanghai Key Laboratory of Bioactive Small Molecules (ZDSYS14005), the Major Project of National Thirteenth Five Plan (2017ZX09304016), the Project of Shanghai Shenkang Hospital Development Center (16CR2009A), and the Project of Clinical Research Center, Shanghai Jiao Tong University School of Medicine (DLY201607).
Author information
Authors and Affiliations
Contributions
B.N performed mouse and cell experiments and implemented the data analysis. Q.X and XH.L performed clinical evaluation and collected clinical samples. XH. L, Y.J, D.X, L.M, and J.L helped collecting samples andperformed western blot analysis and quantitative real-time PCR analysis. X.Z assisted with pathology and scoring. Y.Z. helped with bioinformatics analysis. XB.L, Y. Mao, and X.Z conceived and supervised the project and analyzed the data. XB.L. and B.N. wrote the manuscript. Prof. Sun Ning helped with manuscript reviewing and editing. All authors discussed the results and revised the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
All animal experiments were approved by the Animal Ethics Committee of Fudan University School of Basic Medical Sciences. Animal research was conducted in accordance with international guidelines. The clinical research was approved by the Ethics Committee of Shanghai Jiaotong University Renji Hospital in accordance with the ethical guidelines of the 1975 Declaration of Helsinki.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Highlights
1. Ferroptosis is responsible for APAP-induced hepatocyte cell death.
2. Protecting mitochondria via inhibiting VDAC1 oligomerization attenuates APAP-induced ferroptosis.
3. Taz and Smpd1, responsible for CL and CER synthesis, affect APAP-induced mitochondrial dysfunction and subsequent ferroptosis in hepatocytes.
4. Levels of 4-HNE protein adducts are increased in the liver samples of patients with DILI.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 2880 kb)
Rights and permissions
About this article
Cite this article
Niu, B., Lei, X., Xu, Q. et al. Protecting mitochondria via inhibiting VDAC1 oligomerization alleviates ferroptosis in acetaminophen-induced acute liver injury. Cell Biol Toxicol 38, 505–530 (2022). https://doi.org/10.1007/s10565-021-09624-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-021-09624-x