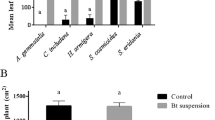
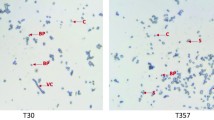
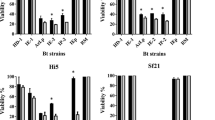
Abstract
Bacillus thuringiensis (Bt)-based bioinsecticides and transgenic plants expressing proteins with insecticidal activity (Cry and Vip) have been successfully used in several integrated pest management programs worldwide. Lepidoptera comprise some of the most economically important insect pests of the major agricultural crops. In this study, the toxicity of 150 Bt strains was evaluated against Helicoverpa armigera (Hübner) larvae. Eight strains (426, 520B, 1636, 1641, 1644, 1648, 1657 and 1658) showed high insecticide activity against H. armigera and were therefore tested against Anticarsia gemmatalis (Hübner), Spodoptera cosmioides (Walker), Chrysodeixis includens (Walker), and Diatraea saccharalis (Fabricius) larvae. Our results showed that most of the Bt strains were also toxic to these lepidopteran species. The biochemical and molecular analyses of these strains revealed that they had a similar protein profile; however, their cry and vip gene contents were variable. In addition, the median lethal concentration (LC50) of the selected strains indicated that the strains 1636, 1641, and 1658 were the most effective against H. armigera, showing LC50 values of 185.02, 159.44, and 192.98 ng/cm2, respectively. Our results suggest that the selected Bt strains have great potential to control the lepidopteran pests H. armigera, A. gemmatalis, D. saccharalis, S. cosmioides, and C. includes.

Similar content being viewed by others
References
Apaydin Ö, Çınar Ç, Turanli F, Harsa Ş, Güneş H (2008) Identification and bioactivity of native strains of Bacillus thuringiensis from grain-related habitats in Turkey. Biol Control 45:21–28. https://doi.org/10.1016/J.BIOCONTROL.2008.01.011
Arnemann JA, James WJ, Walsh TK, Guedes JVC, Smagghe G, Castiglioni E, Tay WT (2016) Mitochondrial DNA COI characterization of Helicoverpa armigera (Lepidoptera: Noctuidae) from Paraguay and Uruguay. Genet Mol Res 15. https://doi.org/10.4238/gmr.15028292
Arneodo JD, Balbi EI, Flores FM, Sciocco-Cap A (2015) Molecular identification of Helicoverpa armigera (Lepidoptera: Noctuidae: Heliothinae) in Argentina and development of a novel PCR-RFLP method for its rapid differentiation from H. zea and H. gelotopoeon. J Econ Entomol 108:2505–2510. https://doi.org/10.1093/jee/tov254
Azzouz H, Kebaili-Ghribi J, Daoud F, Abdelmalak N, Ennouri K, Belguith-Ben Hassan N, Tounsi S, Rouis S (2015) Selection and characterization of Bacillus thuringiensis strains toxic against pyralid stored-product pests. J Appl Entomol 139:690–700. https://doi.org/10.1111/jen.12205
Bacalhau FB, Dourado PM, Horikoshi RJ, Carvalho RA, Semeão A, Martinelli S, Berger GU, Head GP, Salvadori JR, Bernardi O (2020) Performance of genetically modified soybean expressing the Cry1A.105, Cry2Ab2, and Cry1Ac proteins against key lepidopteran pests in Brazil. J Econ Entomol 113:2883–2889. https://doi.org/10.1093/jee/toaa236
Bel Y, Zack M, Narva K, Escriche B (2019) Specific binding of Bacillus thuringiensis Cry1Ea toxin, and Cry1Ac and Cry1Fa competition analyses in Anticarsia gemmatalis and Chrysodeixis includens. Sci Rep 9:1–7. https://doi.org/10.1038/s41598-019-54850-3
Ben-dov E, Zaritsky A, Dahan E et al (1997) Extended screening by PCR for seven cry-group genes from field-collected strains of Bacillus thuringiensis. Appl Environ Microbiol 63:4883–4890
Blanco CA, Chiaravalle W, Dalla-Rizza M, Farias JR, García-Degano MF, Gastaminza G, Mota-Sánchez D, Murúa MG, Omoto C, Pieralisi BK, Rodríguez J, Rodríguez-Maciel JC, Terán-Santofimio H, Terán-Vargas AP, Valencia SJ, Willink E (2016) Current situation of pests targeted by Bt crops in Latin America. Curr Opin Insect Sci 15:131–138. https://doi.org/10.1016/j.cois.2016.04.012
Bortolotto OC, Pomari-Fernandes A, De RCO et al (2015) The use of soybean integrated pest management in Brazil: a review. Agron Sci Biotechnol 1:25–32
Bravo A, Likitvivatanavong S, Gill SS, Soberón M (2011) Bacillus thuringiensis: a story of a successful bioinsecticide. Insect Biochem Mol Biol 41:423–431. https://doi.org/10.1016/j.ibmb.2011.02.006
Brühl CA, Zaller JG (2019) Biodiversity decline as a consequence of an inappropriate environmental risk assessment of pesticides. Front Environ Sci 7:177. https://doi.org/10.3389/fenvs.2019.00177
Cerón J, Covarrubias L, Quintero R et al (1994) PCR analysis of the cryI insecticidal crystal family genes from Bacillus thuringiensis. Appl Envir Microbiol 60:353–356
Chakroun M, Banyuls N, Bel Y, Escriche B, Ferré J (2016) Bacterial vegetative insecticidal proteins (Vip) from entomopathogenic bacteria. Microbiol Mol Biol Rev 80:329–350. https://doi.org/10.1128/MMBR.00060-15
Chen M-L, Chen P-H, Pang J-C, Lin CW, Hwang CF, Tsen HY (2014) The correlation of the presence and expression levels of cry genes with the insecticidal activities against Plutella xylostella for Bacillus thuringiensis strains. Toxins (Basel) 6:2453–2470. https://doi.org/10.3390/toxins6082453
Crickmore N, Berry C, Panneerselvam S, Mishra R, Connor TR, Bonning BC (2020) A structure-based nomenclature for Bacillus thuringiensis and other bacteria-derived pesticidal proteins. J Invertebr Pathol:107438. https://doi.org/10.1016/j.jip.2020.107438
Czepak C, Cordeiro Albernaz K, Vivan LM et al (2013) First reported occurrence of Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae) in Brazil. Pesq Agropec Trop 43:110–113
Dively GP, Dilip Venugopal P, Bean D et al (2018) Regional pest suppression associated with widespread Bt maize adoption benefits vegetable growers. Proc Natl Acad Sci 115:3320–3325. https://doi.org/10.1073/pnas.1720692115
Estruch JJ, Warren GW, Mullins MA, Nye GJ, Craig JA, Koziel MG (1996) Vip3A, a novel Bacillus thuringiensis vegetative insecticidal protein with a wide spectrum of activities against lepidopteran insects. Proc Natl Acad Sci U S A 93:5389–5394. https://doi.org/10.1073/pnas.93.11.5389
Falcon T, Pinheiro DG, Ferreira-Caliman MJ, Turatti ICC, Abreu FCP, Galaschi-Teixeira JS, Martins JR, Elias-Neto M, Soares MPM, Laure MB, Figueiredo VLC, Lopes NP, Simões ZLP, Garófalo CA, Bitondi MMG (2019) Exploring integument transcriptomes, cuticle ultrastructure, and cuticular hydrocarbons profiles in eusocial and solitary bee species displaying heterochronic adult cuticle maturation. PLoS One 14:e0213796. https://doi.org/10.1371/journal.pone.0213796
Finney DJ (1971) Probit analysis. Third ed Cambridge Univ Press Cambridge 60:1432. https://doi.org/10.1002/jps.2600600940
Fortier M, Vachon V, Frutos R et al (2007) Effect of insect larval midgut proteases on the activity of Bacillus thuringiensis Cry toxins. Appl Environ Microbiol 73:6208–6213. https://doi.org/10.1128/AEM.01188-07
Girón-Pérez K, Oliveira AL, Teixeira AF, Guedes RNC, Pereira EJG (2014) Susceptibility of Brazilian populations of Diatraea saccharalis to Cry1Ab and response to selection for resistance. Crop Prot 62:124–128. https://doi.org/10.1016/j.cropro.2014.04.004
Hernández-Rodríguez CS, Boets A, Van Rie J, Ferré J (2009) Screening and identification of vip genes in Bacillus thuringiensis strains. J Appl Microbiol 107:219–225. https://doi.org/10.1111/j.1365-2672.2009.04199.x
ISAAA GM Approval Database. http://www.isaaa.org/gmapprovaldatabase (2018) Global status of commercialized biotech/GM crops: 2018. In: - ISAAA Br. 54-2018 | ISAAA.org. https://www.isaaa.org/resources/publications/briefs/54/default.asp. Accessed 3 Sep 2020
Jurat-Fuentes JL, Crickmore N (2017) Specificity determinants for Cry insecticidal proteins: insights from their mode of action. J Invertebr Pathol 142:5–10. https://doi.org/10.1016/J.JIP.2016.07.018
Jurat-Fuentes JL, Heckel DG, Heckel DG (2021) Mechanisms of resistance to insecticidal proteins from Bacillus thuringiensis. Annu Rev Entomol 66:121–140. https://doi.org/10.1146/annurev-ento-052620
Koch MS, Ward JM, Levine S et al (2015) The food and environmental safety of Bt crops. Front Plant Sci 6:283. https://doi.org/10.3389/fpls.2015.00283
Kriticos DJ, Ota N, Hutchison WD, Beddow J, Walsh T, Tay WT, Borchert DM, Paula-Moreas SV, Czepak C, Zalucki MP (2015) The potential distribution of invading Helicoverpa armigera in North America: is it just a matter of time? PLoS One 10:e0119618. https://doi.org/10.1371/journal.pone.0119618
Kunthic T, Watanabe H, Kawano R, Tanaka Y, Promdonkoy B, Yao M, Boonserm P (2017) pH regulates pore formation of a protease activated Vip3Aa from Bacillus thuringiensis. Biochim Biophys Acta Biomembr 1859:2234–2241. https://doi.org/10.1016/J.BBAMEM.2017.08.018
Lacey LA, Grzywacz D, Shapiro-Ilan DI, Frutos R, Brownbridge M, Goettel MS (2015) Insect pathogens as biological control agents: back to the future. J Invertebr Pathol 132:1–41. https://doi.org/10.1016/j.jip.2015.07.009
Lecadet MM, Chaufaux J, Ribier J, Lereclus D (1992) Construction of novel Bacillus thuringiensis strains with different insecticidal activities by transduction and transformation. Appl Environ Microbiol 58:840–849
Lemes ARN, Davolos CC, Legori PCBC, Fernandes OA, Ferré J, Lemos MVF, Desiderio JA (2014) Synergism and antagonism between Bacillus thuringiensis Vip3A and Cry1 proteins in Heliothis virescens, Diatraea saccharalis and Spodoptera frugiperda. PLoS One 9:e107196. https://doi.org/10.1371/journal.pone.0107196
Lone SA, Malik A, Padaria JC (2017) Selection and characterization of Bacillus thuringiensis strains from northwestern Himalayas toxic against Helicoverpa armigera. Microbiologyopen 6:484. https://doi.org/10.1002/mbo3.484
Mascarin GM, Lopes RB, Delalibera Í et al (2018) Current status and perspectives of fungal entomopathogens used for microbial control of arthropod pests in Brazil. J Invertebr Pathol 165:46–53. https://doi.org/10.1016/j.jip.2018.01.001
Mota-Sanchez, D., & Wise, J. C. (2021) Duynslager LA Arthropod Pesticide Resistance Database | Michigan State University. https://www.pesticideresistance.org/. Accessed 28 Apr 2021
Murúa AMG, Scalora FS, Navarro FR et al (2014) First record of Helicoverpa armigera (Lepidoptera: Noctuidae) in Argentina. Florida Entomol 97:854–856
Mushtaq R, Behle R, Liu R, Niu L, Song P, Shakoori AR, Jurat-Fuentes JL (2017) Activity of Bacillus thuringiensis Cry1Ie2, Cry2Ac7, Vip3Aa11 and Cry7Ab3 proteins against Anticarsia gemmatalis, Chrysodeixis includens and Ceratoma trifurcata. J Invertebr Pathol 150:70–72. https://doi.org/10.1016/j.jip.2017.09.009
Palma L, Ruiz De Escudero I, Maeztu M et al (2013) Screening of vip genes from a Spanish Bacillus thuringiensis collection and characterization of two Vip3 proteins highly toxic to five lepidopteran crop pests. Biol Control 66:141–149. https://doi.org/10.1016/j.biocontrol.2013.05.003
Pardo-López L, Soberón M, Bravo A (2013) Bacillus thuringiensis insecticidal three-domain Cry toxins: mode of action, insect resistance and consequences for crop protection. FEMS Microbiol Rev 37:3–22. https://doi.org/10.1111/j.1574-6976.2012.00341.x
R Core Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.r-project.org/. Accessed 29 Apr 2021
Romeis J, Naranjo SE, Meissle M, Shelton AM (2019) Genetically engineered crops help support conservation biological control. Biol Control 130:136–154. https://doi.org/10.1016/j.biocontrol.2018.10.001
Ruiz de Escudero I, Banyuls N, Bel Y, Maeztu M, Escriche B, Muñoz D, Caballero P, Ferré J (2014) A screening of five Bacillus thuringiensis Vip3A proteins for their activity against lepidopteran pests. J Invertebr Pathol 117:51–55. https://doi.org/10.1016/j.jip.2014.01.006
Santos BK, Neves P, Maria Meneguim A et al (2009) Selection and characterization of the Bacillus thuringiensis strains toxic to Spodoptera eridania (Cramer), Spodoptera cosmioides (Walker) and Spodoptera frugiperda (Smith) (Lepidoptera: Noctuidae). Biol Control 50:157–163. https://doi.org/10.1016/j.biocontrol.2009.03.014
Schnepf E, Crickmore N, Van Rie J et al (1998) Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol Mol Biol Rev 62:775–806
Sebastião I, Rita Nunes Lemes A, Soares Figueiredo C et al (2015) Toxicidade e capacidade de ligação de proteínas Cry1 a receptores intestinais de Helicoverpa armigera (Lepidoptera: Noctuidae). Pesqui Agropecuária Bras 50:999–1005. https://doi.org/10.1590/S0100-204X2015001100002
Shuhaimi M, Ali AM, Saleh NM, Yazid AM (2001) Utilisation of enterobacterial repetitive intergenic consensus (ERIC) sequence-based PCR to fingerprint the genomes of Bifidobacterium isolates and other probiotic bacteria. Biotechnol Lett 23:731–736. https://doi.org/10.1023/A:1010355003674
Silva MC, Siqueira HA, Marques EJ et al (2012) Bacillus thuringiensis isolates from northeastern Brazil and their activities against Plutella xylostella (Lepidoptera: Plutellidae) and Spodoptera frugiperda (Lepidoptera: Noctuidae). Biocontrol Sci Technol ISSN 22:583–599. https://doi.org/10.1080/09583157.2012.670802
Siviter H, Muth F (2020) Do novel insecticides pose a threat to beneficial insects? Proc R Soc B Biol Sci 287:20201265. https://doi.org/10.1098/rspb.2020.1265
Talaei-Hassanloui R, Bakhshaei R, Hosseininaveh V, Khorramnezhad A (2014) Effect of midgut proteolytic activity on susceptibility of lepidopteran larvae to Bacillus thuringiensis subsp. Kurstaki. Front Physiol 4:406. https://doi.org/10.3389/fphys.2013.00406
Tay WT, Soria MF, Walsh T, Thomazoni D, Silvie P, Behere GT, Anderson C, Downes S (2013) A brave new world for an old world pest: Helicoverpa armigera (Lepidoptera: Noctuidae) in Brazil. PLoS One 8:e80134. https://doi.org/10.1371/journal.pone.0080134
Valicente FH, Barreto MR (2003) Bacillus thuringiensis survey in Brazil: Geographical distribution and insecticidal activity against Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae). Neotrop Entomol 32:639–644
Valicente FH, da Silva RB (2014) fAFLP analysis of Brazilian Bacillus thuringiensis isolates. Springerplus 3:256. https://doi.org/10.1186/2193-1801-3-256
Vieira ECS, Ávila CJ, Vivan LM, Silva IF, Vieira MCS, Silva PG (2019) Control of Anticarsia gemmatalis (Hübner: 1818) (Lepidoptera: Erebidae) and Chrysodeixis includens (Walker: 1858) (Lepidoptera: Noctuidae) through insecticides applied to soybean seeds. J Agric Sci 11:88. https://doi.org/10.5539/jas.v11n18p88
Warren GW (1997) Vegetative insecticidal proteins: novel proteins for control of corn pests. In: Koziel M, Carozzi N (eds) Advances in insect control, the role of transgenic plants. CRC Press, pp 109–121
Acknowledgements
We are grateful to the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for supporting the first author through a scholarship. We thank Karine Carvalho and Luis Palhares for their assistance with the statistical analyses.
Author information
Authors and Affiliations
Contributions
Conceptualization: D.H.P. and F.H.V.; Methodology: D.H.P. Formal analysis and investigation: D.H.P.; Writing — original draft preparation: D.H.P.; Writing — review and editing: D.H.P. and F.H.V.; Funding acquisition: F.H.V.; Resources: F.H.V.; Supervision: F.H.V.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Edited by Sengottayan Senthil-Nathan
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 454 kb)
Rights and permissions
About this article
Cite this article
Pinheiro, D.H., Valicente, F.H. Identification of Bacillus thuringiensis Strains for the Management of Lepidopteran Pests. Neotrop Entomol 50, 804–811 (2021). https://doi.org/10.1007/s13744-021-00896-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-021-00896-w