Abstract
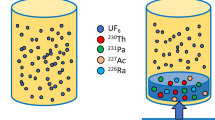
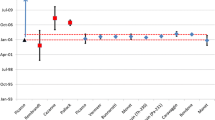
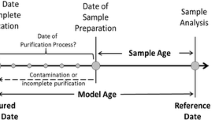
Radiochronometry analyses of three hydrolyzed UF6 gas samples were performed using the 230Th-234U and 231Pa-235U chronometers. All 230Th-234U model ages are younger than, and two of the three 231Pa-235U model ages overlap with, the known UF6 gas transfer dates. The 230Th-234U discordance is caused by 8–10% 230Th loss relative to the measurement reference date, likely during alkali hydrolysis. These results confirm that UF6 gas transfers effectively purify uranium from daughter progeny. Daughter progeny fractionation can occur if solutions are not kept within optimal conditions post-UF6 gas transfer and hydrolysis, and discordance between the 230Th-234U and 231Pa-235U chronometers may result from the laboratory procedures used to prepare samples.



Similar content being viewed by others
References
Williams RW, Gaffney AM (2011) 230Th-234U model ages of some uranium standard reference materials. Radiochim Acta 1:31–35
Gaffney AM, Hubert A, Kinman WS, Magara M, Okubo A, Pointurier F, Schorzman KC, Steiner RE, Williams RW (2016) Round-robin 230Th-234U age dating of bulk uranium for nuclear forensics. J Radioanal Nucl Chem 307:2055–2060
Rolison JM, Williams RW (2018) Application of the 226Ra-230Th-234U and 227Ac-231Pa-235U radiochronometers to UF6 cylinders. J Radioanal Nucl Chem 317:897–905
Varga Z, Mayer K, Bonamici CE, Hubert A, Hutcheon I, Kinman W, Kristo M, Pointurier F, Spencer K, Stanley F, Steiner R, Tandon L, Williams RW (2015) Validation of reference materials for uranium radiochronometry in the frame of nuclear forensic investigations. Appl Radiat Isot 102:81–86
Shimada S, Okumura I, Higashi K (1973) Experimental study on fluoride volatility process for thorium fuels. J Nucl Sci Tech 10(11):689–695
Treinen KC, Gaffney AM, Rolison JM, Samperton KM, McHugh KC, Miller ML, Williams RW (2018) Improved protactinium spike calibration method applied to 231Pa-235U age-dating of certified reference materials for nuclear forensics. J Radioanal Nucl Chem 318:209–219
Essex RM, Mann JL, Williams RW, Kinman WS, Hubert A, Bennett ME, Gurgiotis A (2018) A new thorium-229 reference material. Appl Radiat Isot 134:23–31
Essex RM, Williams RW, Treinen KC, Colléfitzgearld R, Galea R, Keightley J, LaRosa J, Laureano-Pérez L, Nour S, Pibida L (2019) Preparation and calibration of a 231Pa reference material. J Radioanal Nucl Chem 322:1593–1604
BIPM, IEC, IFCC, ISO, IUPAC, IUPAP, OIML (1995) Guide to the Expression of Uncertainty in Measurement. International Organization for Standardization, Geneva. ISBN 92-67-10188-9, First Edition 1993, corrected and reprinted 1995. (BSI Equivalent: BSI PD 6461: 1995, Vocabulary of Metrology, Part 3. Guide to the Expression of Uncertainty in Measurement. British Standards Institution, London)
Bateman, H (1910) Solution of a system of differential equations occurring in the theory of radioactive transformations. Proc Camb Philos Soc 15(V): 423–427
Jaffey AH, Flynn KF, Glendenin LE, Bentley WC, Essling AM (1971) Precision measurement of half-lives and specific activities of 235U and 238U. Phys Rev C 4:1889–1906
Cheng H, Edwards RL, Hoff J, Gallup CD, Richards DA, Asmerom Y (2000) The half-lives of uranium-234 and thorium-230. Chem Geol 169:17–33
IAEA (2004) Handbook of nuclear data for safeguards: Database extensions, August 2008. International Nuclear Data Committee INDC (NDS)-0534
Lau KH, Brittain RD, Hildenbrand DL (1989) High temperature thermodynamic studies of some gaseous thorium fluorides. J Chem Phys 90:1158–1164
Andrews L, Thanthiriwatte KS, Wang X, Dixon DA (2013) Thorium fluorides ThF, ThF2, ThF3, ThF4, ThF3(F2), and ThF5- characterized by infrared spectra in solid argon and electronic structure and vibrational frequency calculations. Inorg Chem 52:8228–8233
Hu S-W, Lin H, Wang X-Y, Chu T-W (2014) Effect of H2O on the hydrolysis of UF6 in the gas phase. J Mol Struct 1062:29–34
Baran V, Škvor F, Voseček V (1984) Formation of the ammonium-uranyl-carbonate complexes of the type (NH4)4[UO2(CO3)3], prepared by precipitative re-extraction. Inorgananica Chim Acta 81:83–89
Shimada-Fujiwara A, Hoshi A, Kameo Y, Nakashima M (2009) Influence of hydrofluoric acid on extraction of thorium using a commercially available extraction chromatographic resin. J Chromatogr A 1216:4125–4127
Stoll W (2000) In: Ley C, Elvers B, and 19 others (ed) Ullmann’s encyclopedia of industrial chemistry. Wiley‐VCH Verlag GmbH & Co
Kirby HW (1959) The radiochemistry of protactinium. National Academy of Sciences National Research Council Nuclear Science Series NAS-NS 3016
De Sio SM, Wilson RE (2014) Structural and spectroscopic studies of fluoroprotactinates. Inorg Chem 53:1750–1755
Shinnosuke H (1959) Determination of the solubility of thorium hydroxide. Bull Instit Chem Res Kyoto Uni 37(3):200–206
Neck V, Kim JI (1999) Solubility and hydrolysis of tetravalent actinides. Forschungszentrum Karlsruhe Technik und Umwelt, Wissenschaftliche Berichte FZKA 6350
Brown D, Easey JF (1970) Protactinium(V) fluorides. J Chem Soc A. https://doi.org/10.1039/j19700003378
Sakanoue M, Abe M (1967) Adsorption method for the separation of protactinium isotopes. Radioisotopes 16(12):645–651
Acknowledgements
We would like to thank the Department of Homeland Security for postdoctoral support to LNH, MCL, Inc., for helpful discussion and information on the samples used in this study, and one anonomous reviewer for comments that improved this work. This work was performed under the auspices of the U.S. Department of Energy by Lawrence Livermore National Laboratory under Contract DE-AC52-07NA27344 and supported by funding from the Department of Homeland Security. Releasable to external audiences, LLNL-JRNL-818999.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Harrison, L.N., Gaffney, A.M. 230Th-234U and 231Pa-235U radiochronometry of hydrolyzed uranium hexafluoride gas. J Radioanal Nucl Chem 329, 1513–1521 (2021). https://doi.org/10.1007/s10967-021-07903-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-021-07903-9