Abstract
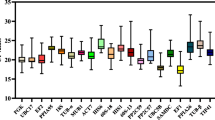
Medicago ruthenica (L.) Ledeb. has received considerable attention due to its value for forage production and ecological restoration in northern China. Gene expression profiles of interesting genes may provide accurate and reliable results for biological function research, in addition to mRNA, including microRNA (miRNA) as a key element that participates in various biological functions. Quantitative real-time polymerase chain reaction (RT-qPCR) is a useful method that has been widely used in gene expression analysis of mRNA and miRNA, and the selection of consistently stable reference genes is essential to normalize RT-qPCR data under different experimental conditions. In this study, the expression stability of nine candidate reference genes was evaluated with DeltaCt, Bestkeeper, NormFinder, geNorm and RefFinder methods. The expression stability of reference genes varied under different experimental conditions. Overall, EF-1α as much more stably reference gene in different subsets recommended for normalization. The relative expression levels of GN4 and miR156f were analyzed to test the reliability of the optimal reference genes. The results suggested that the best stable reference genes can be simultaneously utilized in the normalization of both mRNA and miRNA expression by RT-qPCR analysis, which may facilitate genetic studies of this leguminous plant.






Similar content being viewed by others
Data Availability
All the relevant data is provided in the manuscript and supplementary material.
Code availability
DeltaCt, Bestkeeper, NormFinder, geNorm and RefFinder.
References
Andersen CL, Jensen JL, Ørntoft TF (2004) Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64:5245–5250. https://doi.org/10.1158/0008-5472.CAN-04-0496
Bao WL, Qu YL, Shan XY, Wan YL (2016) Screening and validation of housekeeping genes of the root and cotyledon of Cunninghamia lanceolata under abiotic stresses by using quantitative Real-Time PCR. Int J Mol Sci 17:1198. https://doi.org/10.3390/ijms17081198
Barciszewska-Pacak M, Milanowska K, Knop K, Bielewicz D, Nuc P, Plewka P, Pacak AM, Vazquez F, Karlowski W, Jarmolowski A, Szweykowska-Kulinska Z (2015) Arabidopsis microRNA expression regulation in a wide range of abiotic stress responses. Front Plant Sci 6:410. https://doi.org/10.3389/fpls.2015.00410
Borges A, Tsai SM, Caldas DGG (2012) Validation of reference genes for RT-qPCR normalization in common bean during biotic and abiotic stresses. Plant Cell Rep 31:827–838. https://doi.org/10.1007/s00299-011-1204-x
Cai J, Li PF, Luo X, Chang TL, Li JX, Zhao YW, Xu Y (2018) Selection of appropriate reference genes for the detection of rhythmic gene expression via quantitative real-time PCR in Tibetan hulless barley. PLoS ONE 13:e0190559. https://doi.org/10.1371/journal.pone.0190559
Campbell TA, Xia BG, ZL, (1997) Agronomic evaluation of Medicago ruthenica collected in Inner Mongolia. Crop Sci 37:599–604. https://doi.org/10.2135/cropsci1997.0011183X003700020048x
Campbell TA, Bao G, Xia ZL (1999) Completion of the agronomic evaluations of Medicago ruthenica [(L.) Ledebour] germplasm collected in Inner Mongolia. Genet Resour Crop Ev 46:477–484. https://doi.org/10.1023/A:1008730322306
Chen CF, Ridzon DA, Broomer AJ, Zhou ZH, Lee DH, Nguyen JT, Barbisin M, Xu NL, Mahuvakar VR, Andersen MR, Lao KQ, Livak KJ, Guegler KJ (2005) Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Res 33:e179. https://doi.org/10.1093/nar/gni178
Exposito-Rodriguez M, Borges AA, Perez B-P, JA, (2008) Selection of internal control genes for quantitative real-time RT-PCR studies during tomato development process. BMC Plant Biol 8:131. https://doi.org/10.1186/1471-2229-8-131
Gao MM, Liu YP, Ma X, Shuai Q, Gai JY, Li Y (2017) Evaluation of reference genes for normalization of gene expression using quantitative RT-PCR under aluminum, cadmium, and heat stresses in soybean. PLoS ONE 12:e0168965. https://doi.org/10.1371/journal.pone.0168965
Guan B, Zhou D, Zhang H, Tian Y, Japhet W, Wang P (2009) Germination responses of Medicago ruthenica seeds to salinity, alkalinity, and temperature. J Arid Environ 73:135–138. https://doi.org/10.1016/j.jaridenv.2008.08.009
Jain M, Nijhawan A, Tyagi AK, Khurana JP (2006) Validation of housekeeping genes as internal control for studying gene expression in rice by quantitative real-time PCR. Biochem Biophys Res Commun 345:646–651. https://doi.org/10.1016/j.bbrc.2006.04.140
Karuppaiya P, Yan XX, Liao W, Wu J, Chen F, Tang L (2017) Identification and validation of superior reference gene for gene expression normalization via RT-qPCR in staminate and pistillate flowers of Jatropha curcas - A biodiesel plant. PLoS ONE 12:e0172460
Li J, Li HY, Chi EH, Huang F, Liu L, Ding ZY, Shi WG, Mi FG, Li ZY (2018) Development of simple sequence repeat (SSR) markers in Medicago ruthenica and their application for evaluating outcrossing fertility under open-pollination conditions. Mol Breeding 38:143. https://doi.org/10.1007/s11032-018-0905-z
Liu S, Wu L, Qi HR, Xu M (2019) LncRNA/circRNA–miRNA–mRNA networks regulate the development of root and shoot meristems of Populus. Ind Crops Prod 133:333–347. https://doi.org/10.1016/j.indcrop.2019.03.048
Luo XY, Shi T, Sun HL, Song J, Ni ZJ, Gao ZH (2014) Selection of suitable inner reference genes for normalisation of microRNA expression response to abiotic stresses by RT-qPCR in leaves, flowers and young stems of peach. Sci Hortic 165:281–287. https://doi.org/10.1016/j.scienta.2013.10.030
Luo M, Gao Z, Li H, Li Q, Zhang CX, Xu WP, Song S, Ma C, Wang SP (2018) Selection of reference genes for miRNA qRT-PCR under abiotic stress in grapevine. Sci Rep 8:4444. https://doi.org/10.1038/s41598-018-22743-6
Ma SH, Niu HW, Liu CJ, Zhang J, Hou CY, Wang DM (2013) Expression stabilities of candidate reference genes for RT-qPCR under different stress conditions in soybean. PLoS ONE 8:e75271. https://doi.org/10.1371/journal.pone.0075271
Martins PK, Mafra V, de Souza WR, Ribeiro AP, Vinecky F, Basso MF, da Cunha BA, Kobayashi AK, Molinari HB (2016) Selection of reliable reference genes for RT-qPCR analysis during developmental stages and abiotic stress in Setaria viridis. Sci Rep 6:28348. https://doi.org/10.1038/srep28348
Moraes GP, Benitez LC, Do Amaral MN, Vighi IL, Auler PA, Da Maia LC, Bianchi VJ, Braga EJ (2015) Evaluation of reference genes for RT-qPCR studies in the leaves of rice seedlings under salt stress. Genet Mol Res 14:2384–2398. https://doi.org/10.4238/2015.March.27.24
Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: bestkeeper – excel-based tool using pair-wise correlations. Biotechnol Lett 26:509–515. https://doi.org/10.1023/b:bile.0000019559.84305.47
Qiao YL, Zhang JJ, Zhang JW, Wang ZW, Ran A, Guo HX, Wang D, Zhang JL (2016) Integrated RNA-seq and sRNA-seq analysis reveals miRNA effects on secondary metabolism in Solanum tuberosum L. Mol Genet Genomics 292:37–52. https://doi.org/10.1007/s00438-016-1253-5
Radonić A, Thulke S, Mackay IM, Landt O, Siegert W, Nitsche A (2004) Guideline to reference gene selection for quantitative real-time PCR. Biochem Biophys Res Commun 313:856–862. https://doi.org/10.1016/j.bbrc.2003.11.177
Sang J, Han XJ, Liu MY, Qiao GR, Jiang J, Zhuo RY (2013) Selection and validation of reference genes for real-time quantitative PCR in hyperaccumulating ecotype of Sedum alfredii under different heavy metals stresses. PLoS ONE 8:e82927. https://doi.org/10.1371/journal.pone.0082927
Sheshadri SA, Nishanth MJ, Yamine V, Simon B (2018) Effect of Melatonin on the stability and expression of reference genes in Catharanthus roseus. Sci Rep 8:2222. https://doi.org/10.1038/s41598-018-20474-2
Shu YJ, Li W, Zhao JY, Liu Y, Guo CH (2018) Transcriptome sequencing and expression profiling of genes involved in the response to abiotic stress in Medicago ruthenica. Genet Mol Biol 41:638–648. https://doi.org/10.1590/1678-4685-gmb-2017-0284
Silver N, Best S, Jiang J, Thein SL (2006) Selection of housekeeping genes for gene expression studies in human reticulocytes using real-time PCR. BMC Mol Biol 7:33. https://doi.org/10.1186/1471-2199-7-33
Song XW, Li Y, Cao XF, Qi YJ (2019) MicroRNAs and their regulatory roles in plant-environment interactions. Annu Rev Plant Biol 70:489–525. https://doi.org/10.1146/annurev-arplant-050718-100334
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3(7):1–2. https://doi.org/10.1186/gb-2002-3-7-research0034
Wan Q, Chen SL, Shan ZH, Yang ZL, Chen LM, Zhang CJ, Yuan SL, Hao QN, Zhang XJ, Qiu DZ, Chen HF, Zhou XN (2017) Stability evaluation of reference genes for gene expression analysis by RT-qPCR in soybean under different conditions. PLoS ONE 12:e0189405. https://doi.org/10.1371/journal.pone.0189405
Wang DK, Li H, Luo XY (2008) Crossbreeding of Melilotoides ruthenicus and Medicago sativa. Acta Agrestia Sinica 16:458–465. https://doi.org/10.3901/CJME.2008.04.040
Wang T, Lu JX, Xu ZD, Yang W, Wang JG, Cheng T, Zhang QX (2014) Selection of suitable reference genes for miRNA expression normalization by qRT-PCR during flower development and different genotypes of Prunus mume. Sci Hortic 169:130–137. https://doi.org/10.1016/j.cell.2009.06.014
Wang JW, Czech B, Weigel D (2009) miR156-regulated SPL transcription factors define an endogenous flowering pathway in Arabidopsis thaliana. Cell 138(4):738–749. https://doi.org/10.1016/j.cell.2009.06.014
Wu ZJ, Tian C, Jiang Q, Li XH, Zhuang J (2016) Selection of suitable reference genes for qRT-PCR normalization during leaf development and hormonal stimuli in tea plant (Camellia sinensis). Sci Rep 6:19748. https://doi.org/10.1038/srep19748
Xie FL, Xiao P, Chen DL, Xu L, Zhang BH (2012) miRDeepFinder: a miRNA analysis tool for deep sequencing of plant small RNAs. Plant Mol Biol 80:75–84. https://doi.org/10.1007/s11103-012-9885-2
Yang YT, Zhang X, Chen Y, Guo JL, Ling H, Gao SW, Su YC, Que YX, Xu LP (2016) Selection of reference genes for normalization of MicroRNA expression by RT-qPCR in sugarcane buds under cold stress. Front Plant Sci 7:86. https://doi.org/10.3389/fpls.2016.00086
Yue BL, Li H, Liu M, Wu JY, Li MX, Lei CZ, Huang BZ, Chen H (2019) Characterization of lncRNA-miRNA-mRNA network to reveal potential functional ceRNAs in bovine skeletal muscle. Front Genet 10:91. https://doi.org/10.3389/fgene.2019.00091
Zhang YT, Peng XR, Liu Y, Li YL, Luo Y, Wang XR, Tang HR (2018) Evaluation of suitable reference genes for qRT-PCR normalization in strawberry (Fragaria x ananassa) under different experimental conditions. BMC Mol Biol 19:8. https://doi.org/10.1186/s12867-018-0109-4
Zhang JC, Xie WG, Yu XX, Zhang ZY, Zhao YQ, Wang N, Wang YR (2019) Selection of suitable reference genes for RT-qPCR gene expression analysis in Siberian wild rye (Elymus sibiricus) under different experimental conditions. Genes (basel) 10:451. https://doi.org/10.3390/genes10060451
Zheng C, Zhao L, Wang Y, Shen JZ, Zhang YF, Jia SS, Li YS, Ding ZT (2015) Integrated RNA- Seq and sRNA-Seq analysis identifies chilling and freezing responsive key molecular players and pathways in tea plant (Camellia sinensis). PLoS ONE 10:e0125031. https://doi.org/10.1371/journal.pone.0125031
Zheng QL, Liu JG, Goff BM, Dinkins RD, Zhu HY (2016) Genetic manipulation of miR156 for improvement of biomass production and forage quality in red clover. Crop Sci 56:1199–1205. https://doi.org/10.2135/cropsci2015.11.0726
Zhu H, Chen CJ, Zeng J, Yun Z, Liu YL, Qu HX, Jiang YM, Duan XW, Xia R (2020) MicroRNA528, a hub regulator modulating ROS homeostasis via targeting of a diverse set of genes encoding copper-containing proteins in monocots. New Phytol 225:385–399. https://doi.org/10.1111/nph.16130
Funding
This study has been supported by a grant from the National Natural Science Foundation of China (No. 31601998), the Innovation Talent Plan of National Forestry and Grassland Science and Technology (No. 2019132603), the Inner Mengolia Science & Technology Plan (No. 2020ZD14, No. 2018MS03001).
Author information
Authors and Affiliations
Contributions
Conceptualization, MWG and JL; software, LZ; validation, MWG and JL; formal analysis, HYL; investigation, MWG; Methodology, ZNW; resources, JL; data curation, ZYL; writing—original draft preparation, MWG; writing—review and editing, ZYL; visualization, JL; supervision, ZYL; project administration, ZYL; funding acquisition, JL. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
Authors declare that there are no conflicts of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Guo, MW., Zhu, L., Li, HY. et al. Evaluation of reference genes for normalization of mRNA and microRNA expression by RT-qPCR under different experimental conditions in Medicago ruthenica (L.) Ledeb.. Genet Resour Crop Evol 69, 587–600 (2022). https://doi.org/10.1007/s10722-021-01243-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-021-01243-z