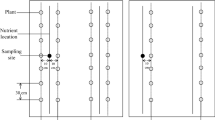
Abstract
Plants can respond to competition with a myriad of physiological or morphological changes. Competition has also been shown to affect the foraging decisions of plants belowground. However, a completely unexplored idea is that competition might also affect plants’ foraging for specific elements required to inhibit the growth of their competitors. In this study, we examined the effect of simulated competition on root foraging and accumulation of heavy metals in the metal hyperaccumulating perennial plant Arabidopsis halleri, whose metal accumulation has been shown to provide allelopathic ability. A. halleri plants originating from both metalliferous and non-metalliferous soils were grown in a “split-root” setup with one root in a high-metal pot and the other in a low-metal one. The plants were then assigned to either simulated light competition or no-competition (control) treatments, using vertical green or clear plastic filters, respectively. While simulated light competition did not induce greater root allocation into the high-metal pots, it did result in enhanced metal accumulation by A. halleri, particularly in the less metal-tolerant plants, originating from non-metalliferous soils. Interestingly, this accumulation response was particularly enhanced for zinc rather than cadmium. These results provide support to the idea that the accumulation of metals by hyperaccumulating plants can be facultative and change according to their demand following competition.



Similar content being viewed by others
Data accessibility statement
The source data for this manuscript will be deposited in the Dryad Digital Repository. The authors declare no competing financial interests. Correspondence and requests for materials should be addressed to M.G.
References
Aerts R, Boot RGA, van der Aart PJM (1991) The relation between above- and belowground biomass allocation patterns and competitive ability. Oecologia 87:551–559. https://doi.org/10.1007/BF00320419
Balafrej H, Bogusz D, El Abidine Triqui Z, Guedira A, Bendaou N, Smouni A, Fahr M (2020) Zinc hyperaccumulation in plants: a review. Plants 9:562. https://doi.org/10.3390/plants9050562
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Methodol 57:289–300
Bert V, Bonnin I, Saumitou-Laprade P, de Laguerie P, Petit D (2002) Do Arabidopsis halleri from nonmetallicolous populations accumulate zinc and cadmium more effectively than those from metallicolous populations? New Phytol 155:47–57. https://doi.org/10.1046/j.1469-8137.2002.00432.x
Bert V, Macnair MR, De Laguerie P, Saumitou-laprade P, Petit D (2008) Zinc tolerance and accumulation in metallicolous and nonmetallicolous populations of Arabidopsis halleri (Brassicaceae). New Phytol 146:225–233. https://doi.org/10.1046/j.1469-8137.2000.00634.x
Boucher U, Lamy I, Cambier P, Balabane M (2005) Decomposition of leaves of the metallophyte Arabidopsis halleri in soil microcosms: fate of Zn and Cd from plant residues. Environ Pollut 135:323–332. https://doi.org/10.1016/j.envpol.2004.07.031
Boyd RS, Jaffré T (2001) Phytoenrichment of soil Ni content by Sebertia acuminata in New Caledonia and the concept of elemental allelopathy. S Afr J Sci 97:535–538
Cahill JF, McNickle GG (2011) The behavioral ecology of nutrient foraging by plants. Annu Rev Ecol Evol Syst 42:289–311. https://doi.org/10.1146/annurev-ecolsys-102710-145006
Cahill JF, McNickle GG, Haag JJ, Lamb EG, Nyanumba SM, Clair CCS, St. Clair CC, (2010) Plants integrate information about nutrients and neighbors. Science 328:1657–1657. https://doi.org/10.1126/science.1189736
Campbell B, Grime JP, Mackey JM (1991) A trade/off between scale and precision in resourse foraging. Oecologia 87:532–538. https://doi.org/10.1007/BF00320417
Casper BB, Cahill JF, Hyatt LA (1998) Above-ground competition does not alter biomass allocated to roots in Abutilon theophrasti. New Phytol 140:231–238. https://doi.org/10.1046/j.1469-8137.1998.00271.x
Cowan JE, Reekie EG (2008) Effects of elevated CO2 on intra-specific competition in Sinapis alba: an examination of the role of growth responses to red:far-red ratio. Plant Biol 10:202–210. https://doi.org/10.1111/j.1438-8677.2007.00026.x
Dechamps C, Noret N, Mozek R, Draye X, Meerts P (2008) Root allocation in metal-rich patch by Thlaspi caerulescens from normal and metalliferous soil - new insights into the rhizobox approach. Plant Soil 310:211–224. https://doi.org/10.1007/s11104-008-9648-7
Einsmann JC, Jones RH, Pu M, Mitchell RJ (1999) Nutrient foraging traits in 10 co-occurring plant species of contrasting life forms. J Ecol 87:609–619. https://doi.org/10.1046/j.1365-2745.1999.00376.x
El Mehdawi AF, Quinn CF, Pilon-Smits EAH (2011) Effects of selenium hyperaccumulation on plant-plant interactions: evidence for elemental allelopathy? New Phytol 191:120–131. https://doi.org/10.1111/j.1469-8137.2011.03670.x
El Mehdawi AF, Cappa JJ, Fakra SC, Self J, Pilon-Smits EAH (2012) Interactions of selenium hyperaccumulators and nonaccumulators during cocultivation on seleniferous or nonseleniferous soil—the importance of having good neighbors. New Phytol 194:264–277. https://doi.org/10.1111/j.1469-8137.2011.04043.x
Farinati S, DalCorso G, Bona E, Corbella M, Lampis S, Cecconi D, Polati R, Berta G, Vallini G, Furini A (2009) Proteomic analysis of Arabidopsis halleri shoots in response to the heavy metals cadmium and zinc and rhizosphere microorganisms. Proteomics 9:4837–4850. https://doi.org/10.1002/pmic.200900036
Farinati S, Dalcorso G, Panigati M, Furini A (2011) Interaction between selected bacterial strains and Arabidopsis halleri modulates shoot proteome and cadmium and zinc accumulation. J Exp Bot 62:3433–3447. https://doi.org/10.1093/jxb/err015
Farley RA, Fitter HA, Fitter AH (1999) The responses of seven co-occurring woodland herbaceous perennials to localized nutrient-rich patches. J Ecol 87:849–859. https://doi.org/10.1046/j.1365-2745.1999.00396.x
Goldberg DE, Barton AM (1992) Patterns and consequences of interspecific competition in natural communities: a review of field experiments with plants. Am Nat 139:771–801
Goldberg DE, Fleetwood L (1987) Competitive effect and response in four annual plants. J Ecol 75:1131–1143. https://doi.org/10.2307/2260318
Gruntman M, Shirata C, Novoplansky, (2011) Plasticity in apical dominance and damage tolerance under variable resource availability in Medicago truncatula. Plant Ecol 212:1537–1548. https://doi.org/10.1007/s11258-011-9929-8
Gruntman M, Anders C, Mohiley A, Laaser T, Clemens S, Höreth S, Tielbörger K (2016) Clonal integration and heavy-metal stress: responses of plants with contrasting evolutionary backgrounds. Evol Ecol 31:305–316. https://doi.org/10.1007/s10682-016-9840-9
Gruntman M, Groß D, Májeková M, Tielbörger K (2017) Decision-making in plants under competition. Nat Commun 8:2235. https://doi.org/10.1038/s41467-017-02147-2
Haines BJ (2002) Zincophilic root foraging in Thlaspi caerulescens. New Phytol 155:363–372. https://doi.org/10.1046/j.1469-8137.2002.00484.x
Hodge A (2004) The plastic plant: root responses to heterogeneous supplies of nutrients. New Phytol 162:9–24. https://doi.org/10.1111/j.1469-8137.2004.01015.x
Houghland E (1993) Competition between an established grass sward and seedlings. Nor J Agric Sci 7:409–420
Hutchings MJ, de Kroon H (1994) Foraging in plants: the role of morphological plasticity in resource acquisition. Adv Ecol Res 25:159–238. https://doi.org/10.1016/S0065-2504(08)60215-9
Jaffe BD, Ketterer ME, Shuster SM (2018) Elemental allelopathy by an arsenic hyperaccumulating fern, Pteris vittata L. J Plant Ecol 11:553–559. https://doi.org/10.1093/jpe/rtx020
Kassaw TK, Frugoli JA (2012) Simple and efficient methods to generate split roots and grafted plants useful for long-distance signaling studies in Medicago truncatula and other small plants. Plant Methods 8:38. https://doi.org/10.1186/1746-4811-8-38
Kazemi-Dinan A, Thomaschky S, Stein RJ, Krämer U, Müller C (2014) Zinc and cadmium hyperaccumulation act as deterrents towards specialist herbivores and impede the performance of a generalist herbivore. New Phytol 202:628–639. https://doi.org/10.1111/nph.12663
Kembel SW, Cahill JF (2005) Plant phenotypic plasticity belowground: a phylogenetic perspective on root foraging trade-offs. Am Nat 166:216–230. https://doi.org/10.1086/431287
Krämer U (2010) Metal hyperaccumulation in plants. Annu Rev Plant Biol 61:517–534. https://doi.org/10.1146/annurev-arplant-042809-112156
Krebs JR, Davies NB (1987) An Introduction to Behavioural Ecology, 2nd edn. Black-Well Scientific Publications, Oxford
Küpper H, Lombi E, Zhao F-J, McGrath SP (2000) Cellular compartmentation of cadmium and zinc in relation to other elements in the hyperaccumulator Arabidopsis halleri. Planta 212:75–84. https://doi.org/10.1007/s004250000366
Liu F, Tang Y, Du R, Yang H, Wu Q, Qiu R (2010) Root foraging for zinc and cadmium requirement in the Zn/Cd hyperaccumulator plant Sedum alfredii. Plant Soil 327:365–375. https://doi.org/10.1007/s11104-009-0060-8
Lombi E, Zhao FJ, Mcgrath SP, Young SD, Sacchi GA (2001) Physiōlogical evidence for a high-affinity cadmium transporter highly expressed in a Thlaspi caerulescens ecotype. New Phytol 149:53–60. https://doi.org/10.1046/j.1469-8137.2001.00003.x
Maliakal S, McDonnell K, Dudley S, Schmitt J (1999) Effects of red to far-red ratio and plant density on biomass allocation and gas exchange in Impatiens capensis. Int J Plant Sci 160:723-733. https://doi.org/10.1086/314157
McNickle GG, St. Clair CC, Cahill JF, (2009) Focusing the metaphor: plant root foraging behaviour. Trends Ecol Evol 24:419–426. https://doi.org/10.1016/j.tree.2009.03.004
Mohiley A, Tielbörger K, Seifan M, Gruntman M (2020) The role of biotic interactions in determining metal hyperaccumulation in plants. Funct Ecol 34:658–668. https://doi.org/10.1111/1365-2435.13502
Mommer L, van Ruijven J, Jansen C, van de Steeg HM, de Kroon H (2012) Interactive effects of nutrient heterogeneity and competition: implications for root foraging theory? Funct Ecol 26:66–73. https://doi.org/10.1111/j.1365-2435.2011.01916.x
Morris C, Grossl PR, Call CA (2009) Elemental allelopathy: processes, progress, and pitfalls. Plant Ecol 202:1–11. https://doi.org/10.1007/s11258-008-9470-6
Muehe EM, Weigold P, Adaktylou IJ, Planer-Friedrich B, Kraemer U, Kappler A, Behrens S (2015) Rhizosphere microbial community composition affects cadmium and zinc uptake by the metal-hyperaccumulating plant Arabidopsis halleri. Appl Environ Microbiol 81:2173–2181. https://doi.org/10.1128/AEM.03359-14
Pattison RR, Goldstein G, Ares A (1998) Growth, biomass allocation and photosynthesis of invasive and native Hawaiian rainforest species. Oecologia 117:449–459. https://doi.org/10.1007/s004420050680
Pauwels M, Saumitou-Laprade P, Catherine AH, Petit D, Bonnin I (2005) Multiple origin of metallicolous populations of the pseudometallophyte Arabidopsis halleri (Brassicaceae) in central Europe: the cpDNA testimony. Mol Ecol 14:4403–4414. https://doi.org/10.1111/j.1365-294X.2005.02739.x
Peng JS, Guan YH, Lin XJ, Xu XJ, Xiao L, Wang HH, Meng S (2021) Comparative understanding of metal hyperaccumulation in plants: a mini-review. Environ Geochem Health 43:1599–1607. https://doi.org/10.1007/s10653-020-00533-2
Plaza S, Weber J, Pajonk S, Thomas J, Talke IN, Schellenberg M, Pradervānd S, Burla B, Geisler M, Martinoia E, Krämer U (2015) Wounding of Arabidopsis halleri leaves enhances cadmium accumulation that acts as a defense against herbivory. Biometals 28:521–528. https://doi.org/10.1007/s10534-015-9829-9
Pomilio AB, Leicach SR, Grass MY, Ghersa CM, Santoro M, Vitale AA (2000) Constituents of the root exudate of Avena fatua grown under far-infrared-enriched light. Phytochem Anal 11:304–308. https://doi.org/10.1002/1099-1565(200009/10)11:5%3c304::AID-PCA531%3e3.0.CO;2-G
Rajaniemi TK (2007) Root foraging traits and competitive ability in heterogeneous soils. Oecologia 153:145–152. https://doi.org/10.1007/s00442-007-0706-2
Rasher DB, Hay ME (2014) Competition induces allelopathy but suppresses growth and anti-herbivore defence in a chemically rich seaweed. Proc Royal Soc B 281:20132615. https://doi.org/10.1098/rspb.2013.2615
Robinson D (1996) Resource capture by localized root proliferation: why do plants bother? Ann Bot 77:179–185. https://doi.org/10.1006/anbo.1996.0020
Saiz-Fernández I, Černý M, Skalák J, Brzobohatý B (2021) Split-root systems: detailed methodology, alternative applications, and implications at leaf proteome level. Plant Methods 17:1–19. https://doi.org/10.1186/s13007-020-00706-1
Sarret G, Saumitou-Laprade P, Bert V, Proux O, Hazemann JL, Traverse A, Marcus MA, Manceau A (2002) Forms of zinc accumulated in the hyperaccumulator Arabidopsis halleri. Plant Physiol 130:1815–1826. https://doi.org/10.1104/pp.007799
Schenk HJ (2006) Root competition: beyond resource depletion. J Ecol 94:725–739. https://doi.org/10.1111/j.1365-2745.2006.01124.x
Schenk HJ, Callaway RM, Mahall BE (1999) Spatial root segregation: are plants territorial? Adv Ecol Res 28:145–168. https://doi.org/10.1016/S0065-2504(08)60032-X
Spielmann J, Ahmadi H, Scheepers M, Weber M, Nitsche S, Carnol M, Bosman B, Kroymann J, Motte P, Clemens S, Hanikenne M (2020) The two copies of the zinc and cadmium ZIP6 transporter of Arabidopsis halleri have distinct effects on cadmium tolerance. Plant Cell and Environ 43:2143–2157. https://doi.org/10.1111/pce.13806
Stein RJ, Horeth S, de Melo JRF, Syllwasschy L, Lee G, Garbin ML, Clemens S, Kramer U (2016) Relationships between soil and leaf mineral composition are element-specific, environment-dependent and geographically structured in the emerging model Arabidopsis halleri. New Phytol 213:1274–1286. https://doi.org/10.1111/nph.14219
Stephens DW (2008) Decision ecology: foraging and the ecology of animal decision making. Cogn Affect Behav Neurosci 8:475–484. https://doi.org/10.3758/CABN.8.4.475
Stuefer JF, Huber H (1998) Differential effects of light quantity and spectral light quality on growth, morphology and development of two stoloniferous Potentilla species. Oecologia 117:1–8. https://doi.org/10.1007/s004420050624
Tilman D (1994) Competition and biodiversity in spatially structured habitats. Ecology 75:2–16. https://doi.org/10.2307/1939377
Tognacchini A, Salinitro M, Puschenreiter M, van der Ent A (2020) Root foraging and avoidance in hyperaccumulator and excluder plants: a rhizotron experiment. Plant Soil 450:287–302. https://doi.org/10.1007/s11104-020-04488-2
Uber HEH, Schmitt J, Stinchcombe JR, Shane Heschel M, Huber H (2003) The adaptive evolution of plasticity: phytochrome-mediated shade avoidance responses. Integr Comp Biol 43:459–469. https://doi.org/10.1093/icb/43.3.459
Ueno D, Yamaji N, Kono I, Huang CF, Ando T, Yano M, Ma JF (2010) Gene limiting cadmium accumulation in rice. Proc Natl Acad Sci USA 107:16500–16505. https://doi.org/10.1073/pnas.1005396107
Valladares F, Niinemets Ü (2008) Shade tolerance, a key plant feature of complex nature and consequences. Annu Rev Ecol Evol Syst 39:237–257. https://doi.org/10.1146/annurev.ecolsys.39.110707.173506
Vandenbussche F, Pierik R, Millenaar FF, Voesenek LACJ, Van Der Straeten D (2005) Reaching out of the shade. Curr Opin Plant Biol 8:462–468. https://doi.org/10.1016/j.pbi.2005.07.007
Weijschedé J, Martínková J, De Kroon H, Huber H (2006) Shade avoidance in Trifolium repens, cost and benefits plasticity in petiole length and leaf size. New Phytol 172:655–666. https://doi.org/10.1111/j.1469-8137.2006.01885.x
Werner EE, Hall DJ, Ecology S, Summer NL (2016) Optimal foraging and the size selection of prey by the bluegill sunfish (Lepomis Macrochirus). Ecology 55:1042–1052. https://doi.org/10.2307/1940354
Wilson SD, Tilman D (1991) Components of plant competition along an experimental gradient of nitrogen availability. Ecology 72:1050–1065. https://doi.org/10.2307/1940605
Zhang Q, Li L, Li J, Wang H, Fang C, Yang X, He H (2018) Increasing rice allelopathy by induction of barnyard grass (Echinochloa crus-galli) root exudates. J Plant Growth Regul 37:745–754. https://doi.org/10.1007/s00344-017-9770-y
Zhao FJ, Jiang RF, Dunham SJ, Mcgrath SP (2004) Cadmium uptake, translocation and tolerance in the hyperaccumulator Arabidopsis halleri. New Phytol 172:646–654. https://doi.org/10.1111/j.1469-8137.2006.01867.x
Acknowledgements
We are grateful to Ute Krämer and Ricardo Stein for introducing us to the topic of metal hyperaccumulating plants, and to Mira Hoch, Bettina Springer and Anne Rysavy for the collection of A. halleri and soil in the field, the preparation of contaminated soil, and propagation of A. halleri in the greenhouse.
Funding
This study was funded by the SPP 1529 priority program ‘‘Adaptomics’’ grants of the German Research Foundation (DFG) to KT and MG (TI 338/ 10–1, and TI 338/10–2) and to SC (CL 152/9–2).
Author information
Authors and Affiliations
Contributions
AM, MG and KT conceived the project. AM, and MG designed the experiment. AM performed the experiments, data collection and statistical analyses. SC and MW performed the Cd analyses. AM wrote the manuscript draft with guidance from MG and all authors contributed to its revisions
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Edith B. Allen.
Rights and permissions
About this article
Cite this article
Mohiley, A., Tielbörger, K., Weber, M. et al. Competition for light induces metal accumulation in a metal hyperaccumulating plant. Oecologia 197, 157–165 (2021). https://doi.org/10.1007/s00442-021-05001-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-021-05001-x