Abstract
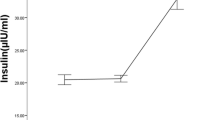
It is well established that acute food restriction (FR) has a suppressive effect on the reproductive system that may be mediated through attenuating the kisspeptin/GPR54 signaling in the hypothalamus. Kisspeptin neurons have been shown to express the dopamine D2 receptor (D2R). The aim of the present study was to investigate the effect of chronic food restriction and/or antagonism of D2R by sulpiride on testosterone release and kisspeptin/GPR54 signaling in male rats. Sixty-six male Wistar rats, weighing-200 ± 20 g, were divided into seven groups including control, sham, 25%FR, 50%FR, 75%FR, sulpiride (50 µg/rat i.c.v., once at the end of treatment), 75%FR + sulpiride. FR was applied for 21 days. At the end, the mean serum testosterone level and the hypothalamic mRNAs expression of KiSS1, GPR54, D2R, and dopamine concentration were evaluated. The results demonstrated that the testosterone level significantly decreased in 50% and 75% food-restricted animals, and in sulpiride group, compared to control rats (P < 0.05). Injection of sulpiride to 75% food-restricted animals reduced the testosterone levels compared to all other groups (P < 0.05). Also, chronic 75%-FR significantly increased kisspeptin mRNA, dopamine concentration, but decreased D2R-mRNA (P < 0.05), and non-significantly decreased the GPR54mRNA (P > 0.05) compared to the control. Sulpiride injection to 75% food-restricted animals significantly decreased kisspeptin mRNA and increased dopamine concentration (P < 0.05), but had no significant effect on GPR54 or D2R-mRNAs expression, compared to 75% food-restricted group. In conclusion, we suggest that chronic food restriction decreases the testosterone release and then increases the kisspeptin/GPR54 signaling, and that the involvement of dopamine D2 receptor in these effects.





Similar content being viewed by others
References
Abizaid A (2009) Ghrelin and dopamine: new insights on the peripheral regulation of appetite. J Neuroendocrinol 21:787–793. https://doi.org/10.1111/j.1365-2826.2009.01896.x
AbouHeif HM, Deif MM, Abdel Aziz HK (2010) Effect of food restriction on ghrelin in adult male rats and its relation to male reproductive hormones. Andrologia 42:97–105. https://doi.org/10.1111/j.1439-0272.2009.00962.x
Aliparasti MR, Alipour MR, Almasi S et al (2015) Ghrelin administration increases the Bax/Bcl-2 gene expression ratio in the heart of chronic hypoxic rats. Adv Pharm Bull 5:195–199. https://doi.org/10.15171/apb.2015.027
Anton S, Leeuwenburgh C (2013) Fasting or caloric restriction for healthy aging. Exp Gerontol 48:1003–1005. https://doi.org/10.1016/j.exger.2013.04.011
Appendix A of the European convention for the protection of vertebrate animals used for experimental and other scientific purpose (ETS NO. 123). Strasbourg, 15 June 2006. https://rm.coe.int/CoERMPublicCommonSearchServices/DisplayDCTMContent?documentId=090000168007a445
Baik JH (2013) Dopamine signaling in food addiction: role of dopamine D2 receptors. BMB Rep 46:519–526. https://doi.org/10.5483/BMBRep.2013.46
Brown MJ, Harland D (1986) B-HT 958 lowers blood pressure and heart rate in the rat through stimulation of dopamine receptors. Br J Pharmacol 87:361–370. https://doi.org/10.1111/j.1476-5381.1986.tb10825.x
Bruce-Keller AJ, Umberger G, McFall R et al (1999) Food restriction reduces brain damage and improves behavioral outcome following excitotoxic and metabolic insults. Ann Neurol 45:8–15. https://doi.org/10.1002/1531-8249(199901)45:1%3C8::AID-ART4%3E3.0.CO;2-V
Cangemi R, Friedmann AJ, Holloszy JO et al (2010) Long-term effects of calorie restriction on serum sex-hormone concentrations in men. Aging Cell 9:236–242. https://doi.org/10.1111/j.1474-9726.2010.00553.x
Carr KD, Tsimberg Y, Berman Y et al (2003) Evidence of increased dopamine receptor signaling in food-restricted rats. Neuroscience 119:1157–1167. https://doi.org/10.1016/S0306-4522(03)00227-6
Castellano JM, Navarro VM, Fernández-Fernández R et al (2005) Changes in hypothalamic KiSS-1 system and restoration of pubertal activation of the reproductive axis by kisspeptin in undernutrition. Endocrinology 146:3917–3925. https://doi.org/10.1210/en.2005-0337
Clarke H, Dhillo WS, Jayasena CN (2015) Comprehensive review on kisspeptin and its role in reproductive disorders. Endocrinol Metab (seoul) 30:124–141. https://doi.org/10.3803/EnM.2015.30.2.124
Forbes S, Li XF, Kinsey-Jones J et al (2009) Effects of ghrelin on kisspeptin mRNA expression in the hypothalamic medial preoptic area and pulsatile luteinising hormone secretion in the female rat. Neurosci Lett 460:143–147. https://doi.org/10.1016/j.neulet.2009.05.060
Ford CP (2014) The role of D2-autoreceptors in regulating dopamine neuron activity and transmission. Neurosci 282:13–22. https://doi.org/10.1016/j.neuroscience.2014.01.025
Frazao R, Dungan Lemko HM, da Silva RP et al (2014) Estradiol modulates Kiss1 neuronal response to ghrelin. Am J Physiol Endocrinol Metab 306:E606–E614. https://doi.org/10.1152/ajpendo.00211.2013
Go EH, Lee SH (2014) Effect of long term reverse feeding on the reproductive and non-reproductive tissues in male mice. Dev Reprod 18:161–166. https://doi.org/10.12717/DR.2014.18.3.161
Goodman RL, Maltby MJ, Millar RP et al (2012) Evidence that dopamine acts via kisspeptin to hold GnRH pulse frequency in check in anestrous ewes. Endocrinology 153:5918–5927. https://doi.org/10.1210/en.2012-1611
Kalamatianos T, Grimshaw SE, Poorun R et al (2008) Fasting reduces KiSS-1 expression in the anteroventral periventricular nucleus (AVPV): effects of fasting on the expression of KiSS-1 and neuropeptide Y in the AVPV or arcuate nucleus of female rats. J Neuroendocrinol 20:1089–1097. https://doi.org/10.1111/j.1365-2826.2008.01757.x
Kalm LM, Semba RD (2005) They starved so that others be better fed: remembering ancel keys and the minnesota experiment. J Nutr 135:1347–1352. https://doi.org/10.1093/jn/135.6.1347
Kawahara Y, Kawahara H, Kaneko F et al (2009) Peripherally administered ghrelin induces bimodal effects on the mesolimbic dopamine system depending on food-consumptive states. Neuroscience 161:855–864. https://doi.org/10.1016/j.neuroscience.2009.03.086
Kern A, Albarran-Zeckler R, Walsh HE et al (2012) Apo-ghrelin receptor forms heteromers with DRD2 in hypothalamic neurons and is essential for anorexigenic effects of DRD2 agonism. Neuron 73:317–332. https://doi.org/10.1016/j.neuron.2011.10.038
Khosravi F, Khazali H, Mahmoudi F et al (2018) Dopamine decreases the stimulatory effects of kisspeptin on serum LH hormone concentrations in male rats. Iran J Physiol Pharmacol 1:17–23
Ladyman SR, Woodside B (2014) Food restriction during lactation suppresses Kiss1 mRNA expression and kisspeptin-stimulated LH release in rats. Reproduction 147:743–751. https://doi.org/10.1530/REP-13-0426
Larionov A, Krause A, Miller W (2005) A standard curve based method for relative real time PCR data processing. BMC Bioinform 6:62. https://doi.org/10.1186/1471-2105-6-62
Lindblom J, Johansson A, Holmgren A et al (2006) Increased mRNA levels of tyrosine hydroxylase and dopamine transporter in the VTA of male rats after chronic food restriction. Eur J Neurosci 23:180–186. https://doi.org/10.1111/j.1460-9568.2005.04531.x
Lippincott MF, Chan YM, Delaney A et al (2016) Kisspeptin responsiveness signals emergence of reproductive endocrine activity: implications for human puberty. J Clin Endocrinol Metab 101:3061–3069. https://doi.org/10.1210/jc.2016-1545
Liu X, Herbison AE (2013) Dopamine regulation of gonadotropin-releasing hormone neuron excitability in male and female mice. Endocrinology 154:340–350. https://doi.org/10.1210/en.2012-1602
Liu X, Herbison AE (2016) Kisspeptin regulation of neuronal activity throughout the central nervous system. Endocrinol Metab (seoul) 31:193–205. https://doi.org/10.3803/EnM.2016.31.2.193
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta DeltaC(T)) Method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Matsuyama S, Ohkura S, Mogi K et al (2011) Morphological evidence for direct interaction between kisspeptin and gonadotropin-releasing hormone neurons at the median eminence of the male goat: an immunoelectron microscopic study. Neuroendocrinol 94:323–332. https://doi.org/10.1159/000331576
Meczekalski B, KatulskiPodfigurna-Stopa KA et al (2016) Spontaneous endogenous pulsatile release of kisspeptin is temporally coupled with luteinizing hormone in healthy women. Fertil Steril 105:1345–1350. https://doi.org/10.1016/j.fertnstert.2016.01.029
Moatt JP, Nakagawa S, Lagisz M et al (2016) The effect of dietary restriction on reproduction: a meta-analytic perspective. BMC Evol Biol 16:199. https://doi.org/10.5061/dryad.3fc02
Paxinos G, Watson C (2013) The rat brain in stereotaxic soordinates, 7th edn. Academic Press, Elsevier
Redman LM, Ravussin E (2009) Endocrine alterations in response to calorie restriction in humans. Mol Cell Endocrinol 299:129–136. https://doi.org/10.1016/j.mce.2008.10.014
Salehi MS, Khazali H, Mahmoudi F et al (2019) The effects of supraphysiological levels of testosterone on neural networks upstream of gonadotropin-releasing hormone neurons. Iran J Basic Med Sci 22:1065–1072. https://doi.org/10.22038/ijbms.2019.36127.8605
Shibata M, Friedman R, Ramaswamy S et al (2007) Evidence that down regulation of hypothalamic KiSS-1 expression is involved in the negative feedback action of testosterone to regulate luteinising hormone secretion in the adult male rhesus monkey (Macacamulatta). J Neuroendocrinol 19:432–438. https://doi.org/10.1111/j.1365-2826.2007.01549.x
Sirotkin AV, Chrenková M, Nitrayová S et al (2008) Effects of chronic food restriction and treatments with leptin or ghrelin on different reproductive parameters of male rats. Peptides 29:1362–1368. https://doi.org/10.1016/j.peptides.2008.03.011
Smith JT, Dungan HM, Stoll EA et al (2005) Differential regulation of KiSS-1 mRNA expression by sex steroids in the brain of the male mouse. Endocrinology 146:2976–2984. https://doi.org/10.1210/en.2005-0323
Unger EL, Wiesinger JA, Hao L et al (2008) Dopamine D2 receptor expression is altered by changes in cellular iron levels in PC12 cells and rat brain tissue. J Nutr 138:2487–2494. https://doi.org/10.3945/jn.108.095224
Wahab F, Atika B, Ullah F et al (2018) Metabolicimpact on the hypothalamic kisspeptin-Kiss1r signaling pathway. Front Endocrinol (lausanne) 9:123. https://doi.org/10.3389/fendo.2018.00123
Wilson CA, Herdon HJ, Bailey LC, Clayton RN (1986) Effect of testosterone on gonadotrophin-releasing hormone receptors in the castrated rat: preliminary evidence for a stimulatory effect of testosterone on gonadotrophin function in the male rat. J Endocrinol 108:441–449. https://doi.org/10.1677/joe.0.1080441
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Khatereh Noormohammadi and Farrin Babaei-Balderlou. The first draft of the manuscript was written by Farrin Babaei-Balderlou, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Human and Animal Rights
All experiments executed according to the Guide for the Care and Use of Laboratory Animals and approved by the institutional animal ethics committee.
Informed Consent
This manuscript does not contain any studies with human subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Nourmohammadi, K., Babaei-Balderlou, F. & Abtahi-Foroushani, S.M. Dopamine D2 Receptor Antagonist Alters the Testosterone Release and Kisspeptin/GPR54 Signaling in Food-Restricted Rats. Iran J Sci Technol Trans Sci 45, 1879–1886 (2021). https://doi.org/10.1007/s40995-021-01196-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-021-01196-z