Abstract
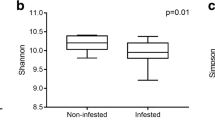
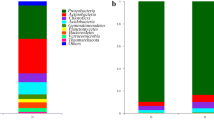
Roots can recruit beneficial microorganisms to suppress plant pathogens. However, conventional and organic practices differently shape the soil microbiome and consequently the root protection. Here, we investigated the suppressive activity of soil microbiome against the root-knot nematode (RKN) Meloidogyne incognita in horticultural areas under organic or conventional practices and the microbiome profiles in non-inoculated (RKN-absent) and inoculated (RKN-present) rhizospheres. Soils were collected from neighboring areas under long-term conventional or organic practices, but physicochemically similar. After a set of bio-tests in autoclaved and non-autoclaved soils, we concluded that the soil suppressiveness was of biological origin. However, plant growth, RKN suppression and defense induction were higher in organic soils. Also, RKN was highly suppressed when the organic soil microbiome was transferred to infested substrates. We used Illumina MiSeq platform to determine bacterial and fungal profiles in organic and conventional tomato rhizospheres, inoculated or not with M. incognita. Our data suggest that despite the higher bacterial abundance in the conventional RKN-absent rhizosphere, the organic RKN-present rhizosphere recruited more efficiently antagonistic bacteria and fungi. Microbiome α-diversity increased in the organic RKN-present rhizosphere. Antagonistic bacterial genera Pseudomonas, Serratia, Bradyrhizobium, Burkholderia and Azospirillum and fungal genera Beauveria, Clonostachys, Metarhizium, Purpureocillium and Arthrobotrys were highly abundant only in the organic RKN-present rhizosphere. This work suggests the organic and conventional practices interfere in the rhizosphere microbiome composition and consequently in the suppression of RKN, where organic practices intensified the assembly of bacterial and fungal antagonists in the presence of M. incognita.






Similar content being viewed by others
References
Abad P, Favery B, Rosso MN, Castagnone-Sereno P (2003) Root-knot nematode parasitism and host response: molecular basis of a sophisticated interaction. Mol Plant Pathol 4:217–224. https://doi.org/10.1046/j.1364-3703.2003.00170.x
Adam M, Westphal A, Hallmann J, Heuer H (2014) Specific microbial attachment to root knot nematodes in suppressive soil. Appl Environ Microbiol 80:2679–2686. https://doi.org/10.1128/AEM.03905-13
Babin D et al. (2021) Distinct rhizomicrobiota assemblages and plant performance in lettuce grown in soils with different agricultural management histories. Microbiol Ecol 97:fiab027. https://doi.org/10.1093/femsec/fiab027.
Bakker MG, Manter DK, Sheflin AM, Weir TL, Vivanco JM (2012) Harnessing the rhizosphere microbiome through plant breeding and agricultural management. Plant Soil 360:1–13. https://doi.org/10.1007/s11104-012-1361-x
Bakker PA, Pieterse CM, de Jonge R, Berendsen RL (2018) The soil-borne legacy. Cell 172:1178–1180. https://doi.org/10.1016/j.cell.2018.02.024
Caporaso JG et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336. https://doi.org/10.1038/nmeth.f.303
Castillo JD, Vivanco JM, Manter DK (2017) Bacterial microbiome and nematode occurrence in different potato agricultural soils. Microb Ecol 74:888–900. https://doi.org/10.1007/s00248-017-0990-2
Chairin T, Petcharat V (2017) Induction of defense responses in longkong fruit (Aglaia dookkoo Griff.) against fruit rot fungi by Metarhizium guizhouense. Biol Control 111:40–44. https://doi.org/10.1016/j.biocontrol.2017.05.012
Chong J, Liu P, Zhou G, Xia J (2020) Using MicrobiomeAnalyst for comprehensive statistical, functional, and meta-analysis of microbiome data. Nat Protoc 15:799–821
Crowder DW, Northfield TD, Strand MR, Snyder WE (2010) Organic agriculture promotes evenness and natural pest control. Nature 466:109–112. https://doi.org/10.1038/s41596-019-0264-1
De Carvalho TS, Jesus EdC, Barlow J, Gardner TA, Soares IC, Tiedje JM, Moreira FMdS (2016) Land use intensification in the humid tropics increased both alpha and beta diversity of soil bacteria. Ecology 97:2760–2771. https://doi.org/10.1002/ecy.1513
De Medeiros HA, de Araújo Filho JV, De Freitas LG, Castillo P, Rubio MB, Hermosa R, Monte E (2017). Tomato progeny inherit resistance to the nematode Meloidogyne javanica linked to plant growth induced by the biocontrol fungus Trichoderma atroviride. Sci Rep 7:1–13. srep40216.
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998. https://doi.org/10.1038/nmeth.2604
Elhady A, Adss S, Hallmann J, Heuer H (2018) Rhizosphere microbiomes modulated by pre-crops assisted plants in defense against plant-parasitic nematodes. Front Microbiol 9:1133. https://doi.org/10.3389/fmicb.2018.01133
Elhady A, Topalović O, Heuer H (2021) Plants specifically modulate the microbiome of root-lesion nematodes in the rhizosphere, affecting their fitness. Microorganisms 9:679. https://doi.org/10.3390/microorganisms9040679
Elhady A, Giné A, Topalovic O, Jacquiod S, Sørensen SJ, Sorribas FJ, Heuer H (2017) Microbiomes associated with infective stages of root-knot and lesion nematodes in soil. PloS one 12. https://doi.org/10.1371/journal.pone.0177145
Forghani F, Hajihassani A (2020) Recent advances in the development of environmentally benign treatments to control root-knot nematodes. Front Plant Sci 11. https://doi.org/10.3389/fpls.2020.01125
Giné A et al (2013) Natural occurrence of fungal egg parasites of root-knot nematodes, Meloidogyne spp. in organic and integrated vegetable production systems in Spain. Biocontrol 58:407–416. https://doi.org/10.1007/s10526-012-9495-6
Giné A, Carrasquilla M, Martínez-Alonso M, Gaju N, Sorribas FJ (2016) Characterization of soil suppressiveness to root-knot nematodes in organic horticulture in plastic greenhouse. Front Plant Sci 7:164. https://doi.org/10.3389/fpls.2016.00164
Gomes VA, Campos VP, da Silva JC, de Jesus SF, de Freitas SM, Pedroso MP (2020) Activity of papaya seeds (Carica papaya) against Meloidogyne incognita as a soil biofumigant. J Pest Sci 93:783–792. https://doi.org/10.1007/s10340-020-01192-z
Goss-Souza D, Mendes LW, Rodrigues JLM, Tsai SM (2020) Ecological processes shaping bulk soil and rhizosphere microbiome assembly in a long-term amazon forest-to-agriculture conversion. Microb Ecol 79:110–122. https://doi.org/10.1007/s00248-019-01401-y
Hannula SE, Hk Ma, Pérez-Jaramillo JE, Pineda A, Bezemer TM (2019) Structure and ecological function of the soil microbiome affecting plant-soil feedbacks in the presence of a soil-borne pathogen. Environ Microbiol 22:660–676. https://doi.org/10.1111/1462-2920.14882
Harkes P, Van Steenbrugge JJM, Van Den Elsen SJJ, Suleiman AKA, De Haan JJ, Holterman MHM, Helder J (2020) Shifts in the active rhizobiome paralleling low Meloidogyne chitwoodi densities in fields under prolonged organic soil management. Front Plant Sci 10:1697. https://doi.org/10.3389/fpls.2019.01697
Hussain M et al (2018). Bacterial community assemblages in the rhizosphere soil, root endosphere and cyst of soybean cyst nematode-suppressive soil challenged with nematodes. FEMS Microbiol Ecol 94:fiy142. https://doi.org/10.1093/femsec/fiy142.
Hussey RS, Barker KR (1973) A comparison of methods for collecting inocula of Meloidogyne spp. including a new technique. Plant Dis Reports 57:1025–1028
Jenkins WR (1964) A rapid centrifugal floatation technique for separating nematodes from soil. Plant Dis Reports 48:692–711
Jones JT et al (2013) Top 10 plant-parasitic nematodes in molecular plant pathology. Mol Plant Pathol 14:946–961. https://doi.org/10.1111/mpp.12057
Khan MR, Mohidin FA, Khan U, Ahamad F (2016) Native Pseudomonas spp. suppressed the root-knot nematode in in vitro and in vivo, and promoted the nodulation and grain yield in the field grown mungbean. Biol Control 101:159–168. https://doi.org/10.1016/j.biocontrol.2016.06.012
Kiriga AW, Haukeland S, Kariuki GM, Coyne DL, Beek NV (2018) Effect of Trichoderma spp. and Purpureocillium lilacinum on Meloidogyne javanica in commercial pineapple production in Kenya. Biol Control 119:27–32. https://doi.org/10.1016/j.biocontrol.2018.01.005
Liu H, Brettell LE, Qiu Z, Singh BK (2020) Microbiome-mediated stress resistance in plants. Trends Plant Sci 25:733–743. https://doi.org/10.1093/nar/gks808
Lupatini M, Korthals GW, de Hollander M, Janssens TK, Kuramae EE (2017) Soil microbiome is more heterogeneous in organic than in conventional farming system. Front Microbiol 7:2064. https://doi.org/10.3389/fmicb.2016.02064
Marty F, Ghiglione J-F, Païssé S, Gueuné H, Quillet L, van Loosdrecht MC, Muyzer G (2012) Evaluation and optimization of nucleic acid extraction methods for the molecular analysis of bacterial communities associated with corroded carbon steel. Biofouling 28:363–380. https://doi.org/10.1080/08927014.2012.672644
Mendes R et al (2011) Deciphering the rhizosphere microbiome for disease-suppressive bacteria. Science 332:1097–1100. https://doi.org/10.1126/science.1203980
Mendes R, Garbeva P, Raaijmakers JM (2013) The rhizosphere microbiome: significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. FEMS Microbiol Rev 37:634–663. https://doi.org/10.1111/1574-6976.12028
Nguyen HP et al (2010) Methods to study PAMP-triggered immunity using tomato and Nicotiana benthamiana. Mol Plant Microbe Interact 23:991–999. https://doi.org/10.1094/MPMI-23-8-0991
Pocurull M et al (2020) Commercial formulates of Trichoderma induce systemic plant resistance to Meloidogyne incognita in tomato and the effect is additive to that of the Mi-1.2 resistance gene. Front Microbiol 10:3042. https://doi.org/10.3389/fmicb.2019.03042.
Pylro VS, Roesch LFW, Morais DK, Clark IM, Hirsch PR, Tótola MR (2014) Data analysis for 16S microbial profiling from different benchtop sequencing platforms. J Microbiol Methods 107:30–37. https://doi.org/10.1016/j.mimet.2014.08.018
Pylro VS, Morais DK, de Oliveira FS, dos Santos FG, Lemos LN, Oliveira G, Roesch LFW (2016) BMPOS: a flexible and user-friendly tool sets for microbiome studies. Microb Ecol 72:443–447. https://doi.org/10.1007/s00248-016-0785-x
Qiu M et al (2014) De-coupling of root–microbiome associations followed by antagonist inoculation improves rhizosphere soil suppressiveness. Biol Fertil Soils 50:217–224. https://doi.org/10.1007/s00374-013-0835-1
Raad M, Glare TR, Brochero HL, Müller C, Rostás M (2019) Transcriptional reprogramming of Arabidopsis thaliana defence pathways by the entomopathogen Beauveria bassiana correlates with resistance against a fungal pathogen but not against insects. Front Microbiol 10:615. https://doi.org/10.3389/fmicb.2019.00615
Rolfe SA, Griffiths J, Ton J (2019) Crying out for help with root exudates: adaptive mechanisms by which stressed plants assemble health-promoting soil microbiomes. Curr Opin Microbiol 49:73–82. https://doi.org/10.1016/j.mib.2019.10.003
Schoch CL et al (2012) Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc Nat Acad Sci 109:6241–6246. https://doi.org/10.1073/pnas.1117018109
Shukla N et al (2018) Transcriptome analysis of root-knot nematode (Meloidogyne incognita)-infected tomato (Solanum lycopersicum) roots reveals complex gene expression profiles and metabolic networks of both host and nematode during susceptible and resistance responses. Mol Plant Pathol 19:615–633. https://doi.org/10.1111/mpp.12547
Silva JCP, Medeiros FHVd, Campos VP (2018) Building soil suppressiveness against plant-parasitic nematodes. Biocontrol Sci Technol 28:423–445. http://orcid.org/0000-0002-1961-6695.
Stagnari F et al (2014) Long-term impact of farm management and crops on soil microorganisms assessed by combined DGGE and PLFA analyses. Front Microbiol 5:644. https://doi.org/10.3389/fmicb.2014.00644
Tao C et al (2020) Bio-organic fertilizers stimulate indigenous soil Pseudomonas populations to enhance plant disease suppression. Microbiome 8:1–14. https://doi.org/10.1186/s40168-020-00892-z
Tariq-Khan M, Munir A, Mukhtar T, Hallmann J, Heuer H (2017) Distribution of root-knot nematode species and their virulence on vegetables in northern temperate agro-ecosystems of the Pakistani-administered territories of Azad Jammu and Kashmir. J Plant Dis Prot 124:201–212. https://doi.org/10.1007/s41348-016-0045-9
Terra WC et al (2018) Volatile organic molecules from Fusarium oxysporum strain 21 with nematicidal activity against Meloidogyne incognita. Crop Prot 106:125–131. https://doi.org/10.1016/j.cropro.2017.12.022
Tian BY, Cao Y, Zhang K-Q (2015) Metagenomic insights into communities, functions of endophytes and their associates with infection by root-knot nematode, Meloidogyne incognita, in tomato roots. Sci Rep 5:1–15. srep17087.
Topalović O, Heuer H (2019) Plant-nematode interactions assisted by microbes in the rhizosphere. Curr Issues Mol Biol 30:75–88. https://doi.org/10.21775/cimb.030.075.
Topalović O, Bredenbruch S, Schleker ASS, Heuer H (2020a) Microbes attaching to endoparasitic phytonematodes in soil trigger plant defense upon root penetration by the nematode. Front Plant Sci 11:138. https://doi.org/10.3389/fpls.2020.00138
Topalović O, Hussain M, Heuer H (2020b) Plants and associated soil microbiota cooperatively suppress plant-parasitic nematodes. Front Microbiol 11:313. https://doi.org/10.3389/fmicb.2020.00313
Verdejo-Lucas S, Blanco M, Talavera M, Stchigel A, Sorribas F (2013) Fungi recovered from root-knot nematodes infecting vegetables under protected cultivation. Biocontrol Sci Technol 23:277–287. https://doi.org/10.1080/09583157.2012.756459
Weller DM, Raaijmakers JM, Gardener BBM, Thomashow LS (2002) Microbial populations responsible for specific soil suppressiveness to plant pathogens. Annu Rev Phytopathol 40:309–348. https://doi.org/10.1146/annurev.phyto.40.030402.110010
Wolfgang A, Taffner J, Guimarães RA, Coyne D, Berg G (2019) Novel strategies for soil-borne diseases: exploiting the microbiome and volatile-based mechanisms towards controlling Meloidogyne-based disease complexes. Front Microbiol 10:1296. https://doi.org/10.3389/fmicb.2019.01296
Yeates GW, Bongers T, De Goede R, Freckman DW, Georgieva S (1993) Feeding habits in soil nematode families and genera—an outline for soil ecologists. J Nematol. 25:315. PMC2619405.
Yergaliyev TM, Alexander-Shani R, Dimerets H, Pivonia S, Bird DM, Rachmilevitch S, Szitenberg A (2020) Bacterial community structure dynamics in meloidogyne incognita-infected roots and its role in worm-microbiome interactions. Msphere 5:e00306–e320. https://doi.org/10.1128/mSphere.00306-20
Zhai Y et al (2018) Multiple modes of nematode control by volatiles of Pseudomonas putida 1A00316 from Antarctic soil against Meloidogyne incognita. Front Microbiol 9:253. https://doi.org/10.3389/fmicb.2018.00253
Zhou D et al (2019) Rhizosphere microbiomes from root knot nematode non-infested plants suppress nematode infection. Microb Ecol 78:470–481. https://doi.org/10.1007/s00248-019-01319-5
Acknowledgements
We acknowledge the financial support provided by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) Grant 409611/2018-2 and also the support by the Brazilian Microbiome Project (http://brmicrobiome.org).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal participants
This article does not contain any studies with human participants or animals (vertebrates) performed by any of the authors.
Additional information
Communicated by Aurelio Ciancio.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Silva, J.C.P., Nunes, T.C.S., Guimarães, R.A. et al. Organic practices intensify the microbiome assembly and suppress root-knot nematodes. J Pest Sci 95, 709–721 (2022). https://doi.org/10.1007/s10340-021-01417-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-021-01417-9