Abstract
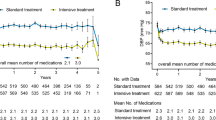
Upstream therapy of atrial remodeling may decrease atrial fibrillation and associated thromboembolism. We examined the impact of intensive BP lowering on ECG-defined left atrial abnormalities in the SPRINT. SPRINT was a randomized clinical trial comparing outcomes when a systolic BP of <120 mmHg (standard treatment) was the target. We included SPRINT participants without baseline atrial fibrillation who had a technically interpretable baseline ECG and at least one follow-up ECG. The primary outcome was incident left atrial abnormality, defined as P-wave terminal force in V1 (PTFV1) > 4000 μV × ms. Secondary outcomes were regression of the left atrial abnormality and the change in PTFV1 from baseline across follow-up ECGs. Cox regression was used to examine the associations between treatment assignment and incident left atrial abnormality and its regression. We used linear mixed models to examine the changes in PTFV1. Of 9361 SPRINT participants, 7738 qualified for this analysis, of whom 5544 did not have baseline left atrial abnormalities. Intensive BP management was not associated with incident left atrial abnormality (HR, 0.96; 95% CI, 0.87–1.07) or regression of the baseline left atrial abnormality (HR, 1.09; 95% CI, 0.98–1.21). The change in PTFV1 from baseline through follow-up did not differ significantly between treatment groups (difference in μV × ms per year, 6; 95% CI, −67 to 79). Thus, among patients in a randomized clinical trial, we found no difference in the progression or regression of ECG-defined left atrial abnormalities with intensive BP management compared to standard BP management.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Iwasaki YK, Nishida K, Kato T, Nattel S. Atrial fibrillation pathophysiology: implications for management. Circulation. 2011;124:2264–74.
Heijman J, Voigt N, Nattel S, Dobrev D. Cellular and molecular electrophysiology of atrial fibrillation initiation, maintenance, and progression. Circulation Res. 2014;114:1483–99.
Abed HS, Wittert GA, Leong DP, Shirazi MG, Bahrami B, Middeldorp ME, et al. Effect of weight reduction and cardiometabolic risk factor management on symptom burden and severity in patients with atrial fibrillation: a randomized clinical trial. JAMA. 2013;310:2050–60.
Kamel H, Okin PM, Longstreth WT Jr., Elkind MS, Soliman EZ. Atrial cardiopathy: a broadened concept of left atrial thromboembolism beyond atrial fibrillation. Future Cardiol. 2015;11:323–31.
Andlauer R, Seemann G, Baron L, Dossel O, Kohl P, Platonov P, et al. Influence of left atrial size on p-wave morphology: differential effects of dilation and hypertrophy. Europace. 2018;20:iii36–44.
Morris JJ Jr, Estes EH Jr, Whalen RE, Thompson HK Jr, McIntosh HD. P-wave analysis in valvular heart disease. Circulation. 1964;29:242–52.
Scott CC, Leier CV, Kilman JW, Vasko JS, Unverferth DV. The effect of left atrial histology and dimension on p wave morphology. J Electrocardiol. 1983;16:363–6.
Soliman EZ, Prineas RJ, Case LD, Zhang ZM, Goff DC Jr. Ethnic distribution of ecg predictors of atrial fibrillation and its impact on understanding the ethnic distribution of ischemic stroke in the atherosclerosis risk in communities (aric) study. Stroke. 2009;40:1204–11.
Eranti A, Aro AL, Kerola T, Anttonen O, Rissanen HA, Tikkanen JT, et al. Prevalence and prognostic significance of abnormal p terminal force in lead v1 of the ECG in the general population. Circulation Arrhythmia Electrophysiol. 2014;7:1116–21.
Tereshchenko LG, Henrikson CA, Sotoodehnia N, Arking DE, Agarwal SK, Siscovick DS, et al. Electrocardiographic deep terminal negativity of the p wave in v(1) and risk of sudden cardiac death: the atherosclerosis risk in communities (aric) study. J Am Heart Assoc. 2014;3:e001387.
Hancock EW, Deal BJ, Mirvis DM, Okin P, Kligfield P, Gettes LS, et al. Aha/accf/hrs recommendations for the standardization and interpretation of the electrocardiogram: part V—electrocardiogram changes associated with cardiac chamber hypertrophy: a scientific statement from the american heart association electrocardiography and arrhythmias committee, council on clinical cardiology; the american college of cardiology foundation; and the heart rhythm society. Endorsed by the international society for computerized electrocardiology. J Am Coll Cardiol. 2009;53:992–1002.
Kamel H, Hunter M, Moon YP, Yaghi S, Cheung K, Di Tullio MR, et al. Electrocardiographic left atrial abnormality and risk of stroke: Northern Manhattan study. Stroke. 2015;46:3208–12.
Kamel H, Soliman EZ, Heckbert SR, Kronmal RA, Longstreth WT Jr., Nazarian S, et al. P-wave morphology and the risk of incident ischemic stroke in the multi-ethnic study of atherosclerosis. Stroke. 2014;45:2786–8.
Kamel H, O’Neal WT, Okin PM, Loehr LR, Alonso A, Soliman EZ. Electrocardiographic left atrial abnormality and stroke subtype in aric. Ann Neurol. 2015;78:670–8.
Wright JT Jr, Williamson JD, Whelton PK, Snyder JK, Sink KM, Rocco MV, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373:2103–16.
Ambrosius WT, Sink KM, Foy CG, Berlowitz DR, Cheung AK, Cushman WC, et al. The design and rationale of a multicenter clinical trial comparing two strategies for control of systolic blood pressure: the Systolic Blood Pressure Intervention Trial (SPRINT). Clin Trials. 2014;11:532–46.
Hommel G. A stagewise rejective multiple test procedure based on a modified bonferroni test. Biometrika. 1988;75:383–6.
Staszewsky L, Wong M, Masson S, Raimondi E, Gramenzi S, Proietti G, et al. Left atrial remodeling and response to valsartan in the prevention of recurrent atrial fibrillation: the GISSI-AF echocardiographic substudy. Circ Cardiovasc Imaging. 2011;4:721–8.
Kapelios CJ, Murrow JR, Nuhrenberg TG, Montoro Lopez MN. Effect of mineralocorticoid receptor antagonists on cardiac function in patients with heart failure and preserved ejection fraction: a systematic review and meta-analysis of randomized controlled trials. Heart Fail Rev. 2019;24:367–77.
McNamara DA, Aiad N, Howden E, Hieda M, Link MS, Palmer D, et al. Left atrial electromechanical remodeling following 2 years of high-intensity exercise training in sedentary middle-aged adults. Circulation. 2019;139:1507–16.
Edelmann F, Gelbrich G, Dungen HD, Frohling S, Wachter R, Stahrenberg R, et al. Exercise training improves exercise capacity and diastolic function in patients with heart failure with preserved ejection fraction: results of the ex-dhf (exercise training in diastolic heart failure) pilot study. J Am Coll Cardiol. 2011;58:1780–91.
Chen LY, Bigger JT, Hickey KT, Chen H, Lopez-Jimenez C, Banerji MA, et al. Effect of intensive blood pressure lowering on incident atrial fibrillation and p-wave indices in the accord blood pressure trial. Am J Hypertens. 2016;29:1276–82.
Snyder ML, Soliman EZ, Whitsel EA, Gellert KS, Heiss G. Short-term repeatability of electrocardiographic p wave indices and pr interval. J Electrocardiol. 2014;47:257–63.
Soliman EZ, Rahman AF, Zhang ZM, Rodriguez CJ, Chang TI, Bates JT, et al. Effect of intensive blood pressure lowering on the risk of atrial fibrillation. Hypertension. 2020;75:1491–6.
Funding
The SPRINT is funded with federal funds from the National Institutes of Health (NIH), including the National Heart, Lung, and Blood Institute (NHLBI), the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), the National Institute on Aging (NIA), and the National Institute of Neurological Disorders and Stroke (NINDS), under contract numbers HHSN268200900040C, HHSN268200900046C, HHSN268200900047C, HHSN268200900048C, HHSN268200900 049C, and Inter-Agency Agreement Number A-HL-13-002-001. It was also supported in part with resources and the use of facilities through the Department of Veterans Affairs. SPRINT investigators acknowledge the contribution of the study. Medications (azilsartan and azilsartan combined with chlorthalidone) were from Takeda Pharmaceuticals International, Inc. All components of the SPRINT study protocol were designed and implemented by the investigators. The investigative team collected, analyzed, and interpreted the data. All aspects of article writing and revision were carried out by the coauthors. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH, the US Department of Veterans Affairs, or the United States Government. For a full list of contributors to SPRINT, please see the supplementary acknowledgment list: https://www.sprinttrial.org/public/dspScience.cfm. We also acknowledge the support from the following Clinical and Translational Science Awards funded by the National Center for Advancing Translational Sciences of the NIH: Case Western Reserve University: UL1TR000439, Ohio State University: UL1RR025755, University of Pennsylvania: UL1RR024134 and UL1TR000003, Boston University: UL1RR025771, Stanford University: UL1TR000093, Tufts: UL1RR025752, UL1TR000073 and UL1TR001064, University of Illinois: UL1TR000050, University of Pittsburgh: UL1TR000005, UT Southwestern University: 9U54TR000017-06, University of Utah: UL1TR000105-05, Vanderbilt University: UL1 TR000445, George Washington University: UL1TR000075, University of California, Davis: UL1 TR000002, University of Florida: UL1 TR000064, University of Michigan: UL1TR000433, and Tulane University: P30GM103337 COBRE Award NIGMS. The views expressed in this article are those of the authors and do not necessarily represent the views of the National Heart, Lung, and Blood Institute; National Institutes of Health; or the United States Department of Health and Human Services or of the Veterans Administration.
Author information
Authors and Affiliations
Contributions
Conception and design of study: all authors. Data collection: CEL, EZS. Statistical analysis: AFR. Interpretation of data: all authors. Drafting of manuscript: HK. Revision of manuscript for important intellectual content.
Corresponding author
Ethics declarations
Conflict of interest
HK serves as co-PI for the NIH-funded ARCADIA trial (NINDS U01NS095869), which receives an in-kind study drug, Eliquis®, from the BMS-Pfizer Alliance and ancillary study support from Roche Diagnostics; serves as a Deputy Editor for JAMA Neurology; serves as a steering committee member of Medtronic’s Stroke AF trial (uncompensated); serves on an endpoint adjudication committee for a trial of empagliflozin for Boehringer-Ingelheim; and has served on an advisory board for Roivant Sciences related to factor XI inhibition.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kamel, H., Rahman, A.F., O’Neal, W.T. et al. Effect of intensive blood pressure lowering on left atrial remodeling in the SPRINT. Hypertens Res 44, 1326–1331 (2021). https://doi.org/10.1038/s41440-021-00713-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-021-00713-2
Keywords
This article is cited by
-
Denervation or stimulation? Role of sympatho-vagal imbalance in HFpEF with hypertension
Hypertension Research (2023)
-
P-wave changes as an index of hypertensive organ damage and a predictor of cardiovascular events: can the P wave be used to assess atrial reverse remodeling?
Hypertension Research (2022)
-
Update on Hypertension Research in 2021
Hypertension Research (2022)