Abstract
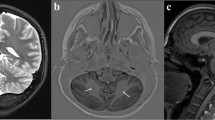
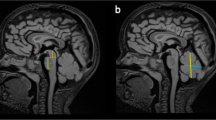
Dandy–Walker malformation (DWM) and Cerebellar vermis hypoplasia (CVH) are commonly recognized human cerebellar malformations diagnosed following ultrasound and antenatal or postnatal MRI. Specific radiological criteria are used to distinguish them, yet little is known about their differential developmental disease mechanisms. We acquired prenatal cases diagnosed as DWM and CVH and studied cerebellar morphobiometry followed by histological and immunohistochemical analyses. This was supplemented by laser capture microdissection and RNA-sequencing of the cerebellar rhombic lip, a transient progenitor zone, to assess the altered transcriptome of DWM vs control samples. Our radiological findings confirm that the cases studied fall within the accepted biometric range of DWM. Our histopathological analysis points to reduced foliation and inferior vermian hypoplasia as common features in all examined DWM cases. We also find that the rhombic lip, a dorsal stem cell zone that drives the growth and maintenance of the posterior vermis is specifically disrupted in DWM, with reduced proliferation and self-renewal of the progenitor pool, and altered vasculature, all confirmed by transcriptomics analysis. We propose a unified model for the developmental pathogenesis of DWM. We hypothesize that rhombic lip development is disrupted through either aberrant vascularization and/or direct insult which causes reduced proliferation and failed expansion of the rhombic lip progenitor pool leading to disproportionate hypoplasia and dysplasia of the inferior vermis. Timing of insult to the developing rhombic lip (before or after 14 PCW) dictates the extent of hypoplasia and distinguishes DWM from CVH.







Similar content being viewed by others
Data availability and materials
Normal human material was provided by the Joint Medical Research Council/Wellcome (MR/R006237/1) HDBR (www.hdbr.org), and the BDRL, University of Washington (NIH R24 HD000836 awarded to IAG). Human tissue used in this study was covered by a material transfer agreement between SCRI and HDBR/BDRL. Sequence data will be available upon request.
References
Aldinger KA, Doherty D (2016) The genetics of cerebellar malformations. Semin Fetal Neonatal Med 21:321–332. https://doi.org/10.1016/j.siny.2016.04.008
Aldinger KA, Lehmann OJ, Hudgins L, Chizhikov VV, Bassuk AG, Ades LC (2009) FOXC1 is required for normal cerebellar development and is a major contributor to chromosome 6p25.3 Dandy-Walker malformation. Nat Genet 41:1037–1042. https://doi.org/10.1038/ng.422
Aldinger KA, Thomson Z, Phelps IG, Haldipur P, Deng M, Timms AE et al (2021) Spatial and cell type transcriptional landscape of human cerebellar development. Nat Neurosci. https://doi.org/10.1038/s41593-021-00872-y
Aldinger KA, Timms AE, Thomson Z, Mirzaa GM, Bennett JT, Rosenberg AB et al (2019) Redefining the etiologic landscape of cerebellar Malformations. Am J Hum Genet 105:606–615. https://doi.org/10.1016/j.ajhg.2019.07.019
Barkovich AJ, Millen KJ, Dobyns WB (2009) A developmental and genetic classification for midbrain-hindbrain malformations. Brain 132:3199–3230. https://doi.org/10.1093/brain/awp247
Barkovich AJ, Millen KJ, Dobyns WB (2007) A developmental classification of malformations of the brainstem. Ann Neurol 62:625–639. https://doi.org/10.1002/ana.21239
Benda CE (1954) The Dandy-Walker syndrome or the so-called atresia of the foramen Magendie. J Neuropathol Exp Neurol 13:14–29. https://doi.org/10.1093/jnen/13.1.14
Bernardo S, Vinci V, Saldari M, Servadei F, Silvestri E, Giancotti A et al (2015) Dandy-Walker Malformation: is the ‘tail sign’ the key sign? Prenat Diagn 35:1358–1364. https://doi.org/10.1002/pd.4705
Cater SW, Boyd BK, Ghate SV (2020) Abnormalities of the fetal central nervous system: prenatal US diagnosis with postnatal correlation. Radiographics 40:1458–1472. https://doi.org/10.1148/rg.2020200034
Chapman T, Mahalingam S, Ishak GE, Nixon JN, Siebert J, Dighe MK (2015) Diagnostic imaging of posterior fossa anomalies in the fetus and neonate: part 2, Posterior fossa disorders. Clin Imaging 39:167–175. https://doi.org/10.1016/j.clinimag.2014.10.012
Conte G, Milani S, Palumbo G, Talenti G, Boito S, Rustico M et al (2018) Prenatal brain MR imaging: reference linear biometric centiles between 20 and 24 gestational weeks. AJNR Am J Neuroradiol 39:963–967. https://doi.org/10.3174/ajnr.A5574
Dandy W, Blackfan K (1914) Internal hydrocephalus: an experimental, clinical and pathological study. Am J Dis Child 8:406–482
Delahaye A, Khung-Savatovsky S, Aboura A, Guimiot F, Drunat S, Alessandri JL et al (2012) Pre- and postnatal phenotype of 6p25 deletions involving the FOXC1 gene. Am J Med Genet A 158A:2430–2438. https://doi.org/10.1002/ajmg.a.35548
Dobin A, Davis CA, Schlesinger F, Drenkow J, Zaleski C, Jha S et al (2013) STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29:15–21. https://doi.org/10.1093/bioinformatics/bts635
Gabriel E, Ramani A, Altinisik N, Gopalakrishnan J (2020) Human brain organoids to decode mechanisms of microcephaly. Front Cell Neurosci 14:115. https://doi.org/10.3389/fncel.2020.00115
Grinberg I, Northrup H, Ardinger H, Prasad C, Dobyns WB, Millen KJ (2004) Heterozygous deletion of the linked genes ZIC1 and ZIC4 is involved in Dandy-Walker malformation. Nat Genet 36:1053–1055. https://doi.org/10.1038/ng1420
Guidi S, Ciani E, Bonasoni P, Santini D, Bartesaghi R (2011) Widespread proliferation impairment and hypocellularity in the cerebellum of fetuses with down syndrome. Brain Pathol 21:361–373. https://doi.org/10.1111/j.1750-3639.2010.00459.x
Haldipur P, Aldinger KA, Bernardo S, Deng M, Timms AE, Overman LM et al (2019) Spatiotemporal expansion of primary progenitor zones in the developing human cerebellum. Science 366:454–460. https://doi.org/10.1126/science.aax7526
Haldipur P, Bharti U, Govindan S, Sarkar C, Iyengar S, Gressens P et al (2012) Expression of Sonic hedgehog during cell proliferation in the human cerebellum. Stem Cells Dev 21:1059–1068. https://doi.org/10.1089/scd.2011.0206
Haldipur P, Dang D, Aldinger KA, Janson OK, Guimiot F, Adle-Biasette H et al (2017) Phenotypic outcomes in Mouse and Human Foxc1 dependent Dandy-Walker cerebellar malformation suggest shared mechanisms. Elife. https://doi.org/10.7554/eLife.20898
Haldipur P, Gillies GS, Janson OK, Chizhikov VV, Mithal DS, Miller RJ et al (2014) Foxc1 dependent mesenchymal signalling drives embryonic cerebellar growth. Elife. https://doi.org/10.7554/eLife.03962
Kapur RP, Mahony BS, Finch L, Siebert JR (2009) Normal and abnormal anatomy of the cerebellar vermis in midgestational human fetuses. Birth Defects Res A Clin Mol Teratol 85:700–709. https://doi.org/10.1002/bdra.20589
Leto K, Arancillo M, Becker EB, Buffo A, Chiang C, Ding B et al (2016) Consensus paper: cerebellar development. Cerebellum 15:789–828. https://doi.org/10.1007/s12311-015-0724-2
Liao Y, Smyth GK, Shi W (2014) featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30:923–930. https://doi.org/10.1093/bioinformatics/btt656
Limperopoulos C, Soul JS, Gauvreau K, Huppi PS, Warfield SK, Bassan H et al (2005) Late gestation cerebellar growth is rapid and impeded by premature birth. Pediatrics 115:688–695. https://doi.org/10.1542/peds.2004-1169
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550. https://doi.org/10.1186/s13059-014-0550-8
Phillips JJ, Mahony BS, Siebert JR, Lalani T, Fligner CL, Kapur RP (2006) Dandy-Walker malformation complex: correlation between ultrasonographic diagnosis and postmortem neuropathology. Obstet Gynecol 107:685–693. https://doi.org/10.1097/01.AOG.0000200594.85483.8a
Poretti A, Boltshauser E, Huisman T (2016) Pre- and postnatal neuroimaging of congenital cerebellar abnormalities. Cerebellum 15:5–9. https://doi.org/10.1007/s12311-015-0699-z
Rakic P, Sidman RL (1970) Histogenesis of cortical layers in human cerebellum, particularly the lamina dissecans. J Comp Neurol 139:473–500. https://doi.org/10.1002/cne.901390407
Russo R, Fallet-Bianco C (2007) Isolated posterior cerebellar vermal defect: a morphological study of midsagittal cerebellar vermis in 4 fetuses–early stage of Dandy-Walker continuum or new vermal dysgenesis? J Child Neurol 22:492–500. https://doi.org/10.1177/0883073807299862
Schlatterer SD, Sanapo L, du Plessis AJ, Whitehead MT, Mulkey SB (2021) The role of fetal MRI for suspected anomalies of the posterior Fossa. Pediatr Neurol 117:10–18. https://doi.org/10.1016/j.pediatrneurol.2021.01.002
Taggart JK, Walker AE (1942) Congenital Atresia of the foramens of Luschka and Magendie. Arch NeurPsych 48:583–612. https://doi.org/10.1001/archneurpsyc.1942.02290100083008
Vera Alvarez R, Pongor LS, Marino-Ramirez L, Landsman D (2019) TPMCalculator: one-step software to quantify mRNA abundance of genomic features. Bioinformatics 35:1960–1962. https://doi.org/10.1093/bioinformatics/bty896
Volpe JJ (2009) Cerebellum of the premature infant: rapidly developing, vulnerable, clinically important. J Child Neurol 24:1085–1104. https://doi.org/10.1177/0883073809338067
Volpe P, Contro E, De Musso F, Ghi T, Farina A, Tempesta A et al (2012) Brainstem-vermis and brainstem-tentorium angles allow accurate categorization of fetal upward rotation of cerebellar vermis. Ultrasound Obstet Gynecol 39:632–635. https://doi.org/10.1002/uog.11101
Acknowledgements
We thank the staff at Human Developmental Biology Resource and Birth Defects Research Laboratory; Xuemei Deng, Conrad Winter, Jennifer Forrer (Seattle Children’s Hospital) and Armelle De Mauduit (APHP) for their technical help and support.
Funding
National Institutes of Health R01 NS080390 and R01 NS095733 (KJM). National Ataxia Foundation Young Investigator Research Grant, Brain and Behavior Research Foundation Young Investigator Award #28956, and Franklin Research Award by the American Philosophical Society (PH).
Author information
Authors and Affiliations
Contributions
PH and KJM conceived, designed, and oversaw the execution of the study. Data collection and analysis was performed by PH, TS, SB, DD, AHS, JM, DD, MD, AET, BDD, and JTP. Samples and radiological data were provided by SB, LM, KM, OO, FG, IAG, HAB, RR, JRS, DK, FR, GP, NR, CDG, and ES. KAA designed and oversaw LCM and RNA-seq data collection and analysis. PH and KJM wrote the first draft. All authors reviewed and critiqued the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
401_2021_2355_MOESM1_ESM.pdf
Supplementary file1 (PDF 1241 KB) Suppl. Fig. S1: List of DWM and CVH cases used in the study (H&E). H&E stained midsagittal sections of the DWM cerebella included in the study. A subset of cases has been described previously [13, 18, 20]. Suppl. Fig. 2: Autopsy images of cases used in the study. Suppl. Table S1: List of normal, DWM and CVH cases used in the study. Suppl. Table S2: Specimen and sample characteristics for LCM RNA-seq. Suppl. Table S3: Rhombic lip LCM RNA-seq differential expression analysis and gene expression.
Rights and permissions
About this article
Cite this article
Haldipur, P., Bernardo, S., Aldinger, K.A. et al. Evidence of disrupted rhombic lip development in the pathogenesis of Dandy–Walker malformation. Acta Neuropathol 142, 761–776 (2021). https://doi.org/10.1007/s00401-021-02355-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-021-02355-7