Abstract
Background
The original description of Pachysentis canicola Meyer, 1931 was based on an unknown number of specimens from an undetermined species of Canis in Brazil from the Berlin Museum. It has since been reported from other carnivores in South and North America. Our specimens from the maned wolf, Chrysocyon brachyurus (Illiger, 1815), in Texas, represent a new host record, and has shed more light on morphometric characteristics missing from the original description, and expanded the range of variations in characters that remained fixed since 1931 and that have been repeated in other taxonomic accounts. We have found additional specimens in striped skunk, Mephitis mephitis Schreber, also in Texas.
Methods
We have performed metal analysis on hooks using EDXA (energy dispersive X-ray analysis). Sequences for the 18S gene and ITS1-5.8-ITS2 region of rDNA were generated to molecularly characterize the species for the first time.
Results
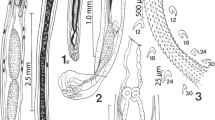
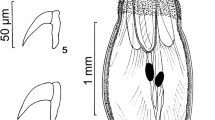
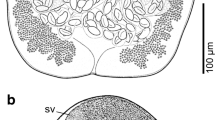
Worms with a massive trunk and a globular proboscis with prominent dome-like apical organ and 12 irregular spiral rows of 4–5 hooks deeply embedded in cuticular folds each, totaling 48–60 hooks. We have included line drawings of the male and female reproductive systems, among other structures, also missing from the original and subsequent descriptions. We describe a new population of P. canicola from Texas and report on the metal analysis of its hooks using EDXA. We also assess the phylogenetic position of P. canicola supporting its independent status in the family Oligacanthorhynchidae, inferred from the two molecular markers.
Conclusions
This is the foremost molecular characterization of any species of Pachysentis and will provide significant insights and reference for future molecular study of species of Pachysentis, especially from this newly described Texas population.





Similar content being viewed by others
References
Meyer A (1931) Neue Acanthocephalen aus dem Berliner Museum. Burgru¨ndung eines neue Acanthocephalen systems auf Grund einer Untersuchung der Berliner Sammlung. Zoologische Jahrbu¨cher Abteilung fu¨r Systematik, O¨ kologie und Geographie der Tiere 62: 53–108. (in German)
Meyer A (1933) Acanthocephala. In: Bronn's Klassen und Ordungen des Tierreichas. Vol. 4. Akademische Verlagsgesellschaft, Leipzg, Germany, Bd. 4, 2 Abt., 2 Buch, pp 333–582. (in German)
Petrochenko VI (1958) Acanthocephala of domestic and wild animals. Isdatel’stvo Akad. Nauk SSSR, Moscow, Russia. (English translation by Israel Program for Scientific Translations, 1971, p 478)
Van Cleave HJ (1953) Acanthocephala of North American mammals Ill. Biol Monogr 23:1–79
Buechner HK (1944) Helminth parasites of the gray fox. J Mammal 25:185–188
Neiswenter SA, Pence DB, Dowler RC (2006) Helminths of sympatric striped, hog-nosed, and spotted skunks in West-Central Texas. J Wildl Dis 42:511–517. https://doi.org/10.7589/0090-3558-42.3.511
Drewek J Jr (1980) Behavior, population structure, parasitism, and other aspects of coyote ecology in southern Arizona. Ph.D. dissertation. University of Arizona, Tucson, Arizona, pp 261
Bolette DP (1997) First record of Pachysentis canicola (Acanthocephala: Oliacanthorhynchidae) and the occurrence of Mesocestoides sp. Tetrathyridia (Cestoidea: Cyclophyllidea) in the western diamondback rattlesnake, Crotalus atrox (Serpentes: Viperidae). J Parasitol 83:751–752
Macedo LC, De Vasconcelos Melo FT, Ávila-Pires TCS, Giese EG, Dos Santos JN (2016) Acanthocephala larvae parasitizing Ameiva ameiva ameiva (Linnaeus, 1758) (Squamata: Teiidae). Rev Bras Parasitol Vet 25:119–123. https://doi.org/10.1590/S1984-29612016007
Mobedi I, Mowlavi Gh, Rahno A, Turner Mobedi K, Mojaradi A (2007) Natural infection of Pachysentis canicola (Acanthocephala: Oligacanthorhynchidae) in fox from Persian Gulf coastal area in Iran. Iran J Parasitol 2:44–47
Haefele HJ, Swenson J, Kinsella M, Amin OM, Garner M (2020) Acanthocephalans in captive maned wolves (Chrysocyon brachyurus). American Association of Zoo Veterinarians 52nd Annual conference (virtual), Nashville, Tennessee, 20–24 Sept 2020
Lee RE (1992) Scanning electron microscopy and X-ray microanalysis. Prentice Hall, Englewood Cliffs, p 458
Suzuki N, Hoshino K, Murakami K, Takeyama H, Chow S (2008) Molecular diet analysis of phyllosoma larvae of the Japanese spiny lobster Palinurus japonicus (Decapoda: Crustacea). Mar Biotechnol 10:49–55. https://doi.org/10.1007/s10126-007-9038-9
Galazzo DE, Dayanandan S, Marcogliese DJ, McLaughlin JD (2002) Molecular systematics of some North American species of Diplostomum (Digenea) based on rDNA-sequence data and comparisons with European congeners. Can J Zool 80:2207–2217. https://doi.org/10.1139/z02-198
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/ NT. Nucleic Acids Symp Ser 41:95–98
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680. https://doi.org/10.1093/nar/22.22.4673
Tamura K, Stecher G, Peterson D, Filipski A, Kumar s, (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. https://doi.org/10.1093/molbev/mst197
Milne I, Lindner D, Bayer M, Husmeier D, Mcguire G, Marshall DF, Wright F (2009) TOPALiv2: a rich graphical interface for evolutionary analyses of multiple alignments on HPC clusters and multi-core desktops. Bioinformatics 25:126–127. https://doi.org/10.1093/bioinformatics/btn575
Posada D (2008) jModelTest: phylogenetic model averaging. Mol Biol Evol 25:1253–1256. https://doi.org/10.1093/molbev/msn083
Schmidt GD (1972) Revision of the class Archiacanthocephala Meyer, 1931 (Phylum Acanthocephala), with emphasis on Oliganthorhynchidae Southwell et Macfie, 1925. J Parasitol 58:290–297
Nickol BB, Dunagan TT (1989) Reconsideration of the acanthocephalan genus Echinopardalis, with a description of adult E. atrata and a key to genera of the Oligacanthorhynchidae. Proc Helminthol Soc Wash 56:8–13
Gomes APN, Amin OM, Olifiers N, de Bianchi CR, Souza JGR, Barbosa HS, Maldonado A Jr (2019) A new species of Pachysentis Meyer, 1931 (Acanthocephala: Oligacanthorhynchidae) in the brown-nosed coati Nasua nasua (Carnivora: Procyonidae) from Brazil, with notes on the genus and a key to species. Acta Parasitol 64:587–595. https://doi.org/10.2478/s11686-019-00080-6
Near JT, Garey JR, Nadler SA (1998) Phylogenetic relationships of the acanthocephala inferred from 18S ribosomal DNA sequences. Mol Phylogenet Evol 10:287–298. https://doi.org/10.1006/mpev.1998.0569
Kamimura K, Yonemitsu K, Maeda K, Sakaguchi S, Setsuda A, Varcasia A, Sato H (2018) An unexpected case of a Japanese wild boar (Sus scrofa leucomystax) infected with the giant thorny-headed worm (Macracanthorhynchus hirudinaceus) on the mainland of Japan (Honshu). Parasitol Res 117:2315–2322. https://doi.org/10.1007/s00436-018-5922-7
García-Varela M, Pérez-Ponce de León G, de la Torre P, Cummings MP, Sarma SSS, Laclette JP (2000) Phylogenetic relationships of Acanthocephala based on analysis of 18S ribosomal RNA gene sequences. J Mol Evol 50:532–540
Amin OM, Heckmann RA, Osama M, Evans RP (2016) Morphological and molecular descriptions of Moniliformis saudi sp. n. (Acanthocephala: Moniliformidae) from the desert hedgehog, Paraechinus aethiopicus (Ehrenberg) in Saudi Arabia, with a key to species and notes on histopathology. Folia Parasitol 26:63. https://doi.org/10.14411/fp.2016.014
Amin OM, Heckmann RA, Sharifdini M, Albayati NY (2019) Moniliformis cryptosaudi n. sp., (Acanthocephala: Moniliformidae) from the long-eared hedgehog Hemiechinus auratus (Gmelin) (Erinaceidae) in Iraq; a case of incipient cryptic speciation related to M. saudi in Saudi Arabia. Acta Parasitol 64:195–204. https://doi.org/10.2478/s11686-018-00021-9
Amin OM, Evans P, Heckmann RA, El-Naggar AM (2013) The description of Mediorhynchus africanus n. sp. (Acanthocephala: Gigantorhynchidae) from galliform birds in Africa. Parasitol Res 112:2897–2906. https://doi.org/10.1007/s00436-013-3461-9
García-Varela M, Cummings MP, Pérez-Ponce de León G, Gardner SL, Laclette JP (2002) Phylogenetic analysis based on 18S ribosomal RNA gene sequences supports the existence of class polyacanthocephala (acanthocephala). Mol Phylogenet Evol 23:288–292. https://doi.org/10.1016/S1055-7903(02)00020-9
García-Varela M, Pérez-Ponce de León G, Aznar FJ, Nadler SA (2013) Phylogenetic relationship among genera of Polymorphidae (Acanthocephala), inferred from nuclear and mitochondrial gene sequences. Mol Phylogenet Evol 68:176–184. https://doi.org/10.1016/j.ympev.2013.03.029
Giribet G, Sørensen MV, Funch P, Kristensen RM, Sterrer W (2004) Investigations into the phylogenetic position of Micrognathozoa using four molecular loci. Cladistics 20:1–13. https://doi.org/10.1111/j.1096-0031.2004.00004.x
Nakao M (2016) Pseudoacanthocephalus toshimai sp. nov. (Palaeacanthocephala: Echinorhynchidae), a common acanthocephalan of anuran and urodelan amphibians in Hokkaido, Japan, with a finding of its intermediate host. Parasitol Int 65:323–332. https://doi.org/10.1016/j.parint.2016.03.011
Irena VS, Damir V, Damir K, Zrinka D, Emil G, Helena C, Emin T (2013) Molecular characterisation and infection dynamics of Dentitruncus truttae from trout (Salmo trutta and Oncorhynchus mykiss) in Krka River, Croatia. Vet Parasitol 197:604–613. https://doi.org/10.1016/j.vetpar.2013.07.014
García-Varela M, Nadler SA (2005) Phylogenetic relationships of Palaeacanthocephala (Acanthocephala) inferred from SSU and LSU rDNA gene sequences. J Parasitol 91:1401–1409. https://doi.org/10.1645/GE-523R.1
Sørensen MV, Giribet G (2006) A modern approach to rotiferan phylogeny: combining morphological and molecular data. Mol Phylogenet Evol 40:585–608. https://doi.org/10.1016/j.ympev.2006.04.001
Heckmann RA, Amin OM, El Naggar AM (2013) Micropores of Acanthocephala, a scanning electron microscopy study. Scientia Parasitol 14:105–113
Amin OM, Heckmann RA, Radwan NA, Mantuano JS, Alcivar MAZ (2009) Redescription of Rhadinorhynchus ornatus (Acanthocephala: Rhadinorhynchidae) from skipjack tuna, Katsuwonus pelamis, collected in the Pacific Ocean off South America, with special reference to new morphological features. J Parasitol 95:656–664. https://doi.org/10.1645/GE-1804.1
Wright RD, Lumsden RD (1969) Ultrastructure of the tegumentary pore-canal system of the acanthocephalan Moniliformis dubius. J Parasitol 55:993–1003
Byram JE, Fisher FM Jr (1973) The absorptive surface of Moniliformis dubius (Acanthocephala). 1 Fine structure. Tissue Cell 5:553–579. https://doi.org/10.1016/S0040-8166(73)80045-X
Whitfield PJ (1979) The biology of parasitism: an introduction to the study of associating organisms. University Park Press, Baltimore, p 277
Amin OM, Heckmann RA, Halajian A, El-Naggar AM (2011) The morphology of an unique population of Corynosoma strumosum (Acanthocephala, Polymorphidae) from the Caspian seal, Pusacaspica, in the land-locked Caspian Sea using SEM, with special notes on Histopathology. Acta Parasitol 56:438–445. https://doi.org/10.2478/s11686-011-0070-6
Tkach I, Sarabeev V, Shvetsova L (2014) Taxonomic status of Neoechinorhynchus agilis (Acanthocephala, Neoechinorhynchidae), with a description of two new species of the genus from the Atlantic and Pacific mullets (Teleostei, Mugilidae). Vestn Zool 48:291–306. https://doi.org/10.2478/vzoo-2014-0035
Amin OM, Sharifdini M, Heckmann RA, Rubtsova N, Chine HJ (2020) On the Neoechinorhynchus agilis (Acanthocephala: Neoechinorhynchidae) complex, with the description of Neoechinorhynchus ponticus n. sp. from Chelon auratus Risso in the Black Sea. Parasite 27:48. https://doi.org/10.1051/parasite/2020044
Heckmann RA, Amin OM, Standing MD (2007) Chemical analysis of metals in acanthocephalans using energy dispersive X-ray analysis (EDXA, XEDS) in conjunction with a scanning electron microscope (SEM). Comp Parasitol 74:388–391. https://doi.org/10.1654/4258.1
Heckmann RA, Amin OM, Radwan NAE, Standing MD, Eggett DL, El Naggar AM (2012) Fine structure and energy dispersive X-ray analysis (EDXA) of the proboscis hooks of Radinorynchus ornatus, Van Cleave 1918 (Rhadinorynchidae: Acanthocephala). Scientia Parasitol 13:37–43
Standing MD, Heckmann RA (2014) Features of acanthocephalan hooks using dual beam preparation and XEDS phase maps. Microscopy and Microanalysis Meeting: Hartford, Connecticut, Poster. Subm. No. 0383-00501
Amin OM, Heckmann RA, Bannai M (2018) Cavisoma magnum (Cavisomidae), a unique Pacific acanthocephalan redescribed from an unusual host, Mugil cephalus (Mugilidae), in the Arabian Gulf, with notes on histopathology and metal analysis. Parasite 25:5. https://doi.org/10.1051/parasite/2018006
Raynaud S, Champion E, Benache-Assollant D, Thomas P (2000) Calcium phosphate apatites with variable Ca. P. atomic ratio synthesis, characterization and thermal stability of powder. Biomaterials 23:1065–1072. https://doi.org/10.1016/s0142-9612(01)00218-6
Amin OM, Chaudhary A, Heckmann RA, Ha NV, Singh HS (2019) The Morphological and Molecular Description of Acanthogyrus (Acanthosentis) fusiformis n. sp. (Acanthocephala: Quadrigyridae) from the Catfish Arius sp. (Ariidae) in the Pacific Ocean off Vietnam, with Notes on Zoogeography. Acta Parasitol 64:779–796. https://doi.org/10.2478/s11686-019-00102-3
Rubtsova NYu, Heckmann RA, Smit WS, Luus-Powell WJ, Halajian A, Roux F (2018) Morphological studies of developmental stages of Oculotrema hippopotami (Monogenea: Polystomatidae) infecting the eye of Hippopotamus amphibius (Mammalia: Hippopotamidae) using SEM and EDXA with notes on histopathology. Korean J Parasitol 56:463–475. https://doi.org/10.3347/kjp.2018.56.5.463
Rubtsova NYu, Heckmann RA (2019) Structure and morphometrics of Ancyrocephalus paradoxus (Monogenea: Ancyrocephalidae) from Sander lucioperca (Percidae) in Czechia. Helminthologia 56:11–21. https://doi.org/10.2478/helm-2018-0037
Rubtsova NYu, Heckmann RA (2020) Morphological features and structural analysis of plerocercoids of Spirometra erinaceieuropaei (Cestoda: Diphyllobothriidae) from European pine marten, Martes martes (Mammalia: Mustelidae) in Ukraine. Comp Parasitol 87:100–117. https://doi.org/10.1654/1525-2647-87.1.109
Acknowledgements
This project was supported by the Department of Biology, Brigham Young University (BYU), Provo, Utah, and by an Institutional Grant from the Parasitology Center, Inc. (PCI), Scottsdale, Arizona. We thank Madison Laurence, Bean Museum (BYU) and Nataliya Rubtsova (PCI) for expert help in the preparation and organization of plates and figures and to Michael Standing, Electron Optics Laboratory (BYU), for his technical help and expertise. We would like to acknowledge the laboratory facilities provided by the Department of Zoology, Chaudhary Charan Singh University, Meerut, India. The authors would also like to thank the staff at Fossil Rim Wildlife Center, Glen Rose, Texas, especially Dr. Holly Haefele and Allyssa Roberts, LVT, for procuring the specimens that were used for this project as well as to thank Dr. Michael Kinsella (HelmWest Laboratory, Missoula, Montana) and Dr. Michael Garner (Northwest Zoo Path, Monroe, Washington) for their assistance with their original recognition of these parasites as acanthocephalans.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
The authors declare that they have observed all applicable ethical standards.
Additional information
Richard A. Heckmann—deceased.
Rights and permissions
About this article
Cite this article
Amin, O.M., Chaudhary, A., Heckmann, R.A. et al. Redescription and Molecular Characterization of Pachysentis canicola Meyer, 1931 (Acanthocephala: Oligacanthorhynchidae) from the Maned Wolf, Chrysocyon brachyurus (Illiger, 1815) in Texas. Acta Parasit. 67, 275–287 (2022). https://doi.org/10.1007/s11686-021-00458-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11686-021-00458-5