Abstract—
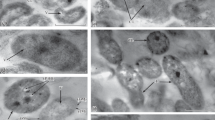
At an elevated copper content, the viability of Azospirillum baldaniorum on wheat seedling roots and the number of cells colonizing the roots and forming biofilms depended on the physicochemical properties of the lipopolysaccharides ( LPSs) synthesized by these bacteria. Compared to the strain Cal+ LpsI LpsII A. baldaniorum strain Sp245, its mutants Cal– LpsI– KM252, Lps II– KM139, and Cal– LpsII– Mot– KM018 colonized the roots less efficiently and formed thinner biofilms. Changes in the polysaccharide synthesis in KM252, KM139, and KM018 mediated increased copper accumulation by the cells and decreased the resistance of bacterial cultures to the negative effect of copper ions. Copper excess in planta and/or on the model polystyrene surface resulted in higher levels of the polysaccharide antigens in the biofilms of strain Sp245 and of its mutants with altered glycopolymer composition. Inoculation with strain Sp245 had a positive effect on the growth of wheat stems and leaves both at copper concentrations which had no noticeable effect on the growth of any of the partners (0.001 mM) and at elevated concentrations of copper ions (up to 0.5 mM). Strains КМ018, КМ139, and КМ252 had a positive effect on the seedlings only at high copper concentrations in the medium.





Similar content being viewed by others
REFERENCES
Abd El-Samad, H.M., The biphasic role of copper and counteraction with Azospirillum brasilense application on growth, metabolities, osmotic pressure and mineral of wheat plant, Amer. J. Plant Sci., 2017, vol. 8, pp. 1182–1195.
Baldani, V.L.D., Baldani, J.I., and Döbereiner, J., Effects of Azospirillum inoculation on root infection and nitrogen incorporation in wheat, Can. J. Microbiol., 1983, vol. 29, pp. 924–929.
Burdman, S., Jurkevitch, E., Schwartsburd, B., Hampel, M., and Okon, Y., Aggregation in Azospirillum brasilense: effects of chemical and physical factors and involvement of extracellular components, Microbiology (SGM), 1998, vol. 144, pp. 1989–1999.
Chang, W.C., Hu, G.S., Chiang, S.M., and Su, M.C., Heavy metal removal from aqueous solution by wasted biomass from a combined AS-biofilm process, Bioresour. Technol., 2006, vol. 97, pp. 1503–1508.
Döbereiner, J. and Day, J.M., Associative symbiosis in tropical grass: characterization of microorganisms and dinitrogen fixing sites, in Symposium on Nitrogen Fixation, Newton, W.E. and Nijmans, C.J., Eds., Pullman: Washington State Univ. Press, 1976, pp. 518–538.
Dos Santos Ferreira, N., Sant’Anna, F.H., Reis, V.M., Ambrosini, A., Volpiano, C.G., Rothballer, M., Schwab, S., Baura, V.A., Balsanelli, E., de Oliveira Pedrosa, F., Pereira Passaglia, L.M., de Souza, E.M., Hartmann, A., Cassan, F., and Zilli, J.E., Genome-based reclassification of Azospirillum brasilense Sp245 as the type strain of Azospirillum baldaniorum sp. nov., Int. J. Syst. Evol. Microbiol., 2020, vol. 70, pp. 6203–6212. https://doi.org/10.1099/ijsem.0.004517
Fedonenko, Yu.P., Zatonsky, G.V., Konnova, S.A., Zdorovenko, E.L., and Ignatov, V.V., Structure of the O-specific polysaccharide of the lipopolysaccharide of Azospirillum brasilense Sp245, Carbohydr. Res., 2002, vol. 337, pp. 869–872.
Fendrihan, S., Constantinescu, F., Sicuia, O., and Dinu, S., Azospirillum strains as biofertilizers and and biocontrol agents—a practical review, J. Adv. Agricult., 2017, vol. 7, pp. 1096–1108.
Fibach-Paldi, S., Burdman, S., and Okon, Y., Key physiological properties contributing to rhizosphere adaptation and plant growth promoting abilities of Azospirillum brasilense, FEMS Microbiol. Lett., 2012, vol. 326, pp. 99–108.
Flemming, H.-C. and Wingender, J., The biofilm matrix, Nat. Rev. Microbiol., 2010, vol. 8, pp. 623–633.
Jing, Y., He, Z., and Yang, X., Role of soil rhizobacteria in phytoremediation of heavy metal contaminated soils, J. Zhejiang Univ. Sci. B., 2007, vol. 8, pp. 192–207.
Kamnev, A.A., Tugarova, A.V., Antonyuk, L.P., Tarantilis, P.A., Polissiou, M.G., and Gardiner, P.H.Y., Effects of heavy metals on plant-associated rhizobacteria: comparison of endophytic and non-endophytic strains of Azospirillum brasilense, J. Trace Elem. Med. Biol., 2005, vol. 19, pp. 91–95.
Katsy, E.I. and Prilipov, A.G., Insertional mutation in the AZOBR_p60120 gene is accompanied by defects in the synthesis of lipopolysaccharide and calcofluor-binding polysaccharides in the bacterium Azospirillum brasilense Sp245, Russ. J. Genet., 2015, vol. 51, pp. 306–311.
Lino, A.R., Farinha, C.R., Pereira, S., and Bursakov, S.A., Desulfovibrio gigas: toxicity of copper and molybdenum, in Metal Ions in Biology and Medicine, Alpoim, M.C., Ed., Lisbon: John Libbey Eurotext, 2006, vol. 9, pp. 231–235.
Lugtenberg, B. and Kamilova, F., Plant-growth-promoting rhizobacteria, Annu. Rev. Microbiol., 2009, vol. 63, pp. 541–556.
Matora, L.Yu. and Shchegolev, S.Yu., Antigenic identity of the capsule lipopolysaccharides, exopolysaccharides, and O-specific polysaccharides in Azospirillum brasilense, Microbiology (Moscow), 2008, vol. 71, pp. 178–181.
Nies, D.H., Microbial heavy-metal resistance, Appl. Microbiol. Biotechnol., 1999, vol. 51, pp. 730–750.
Nocelli, N., Bogino, P.C., Banchio, E., and Giordano, W., Roles of extracellular polysaccharides and biofilm formation in heavy metal resistance of rhizobia, Materials, 2016, vol. 9, p. 418. https://doi.org/10.3390/ma9060418
O’Toole, G.A. and Kolter, R., Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: a genetic analysis, Mol. Microbiol., 1998, vol. 28, pp. 449–461.
Petrova, L.P., Yevstigneeva, S.S., Borisov, I.V., Shelud’ko, A.V., Burygin, G.L, and Katsy, E.I., Plasmid gene AZOBR_p60126 impacts biosynthesis of lipopolysaccharide II and swarming motility in Azospirillum brasilense Sp245, J. Basic Microbiol., 2020b, vol. 60, pp. 613–623.
Petrova, L.P., Yevstigneeva, S.S., Filip’echeva, Yu.A., Shelud’ko, A.V., Burygin, G.L, and Katsy, E.I., Plasmid gene for putative integral membrane protein affects formation of lipopolysaccharide and motility in Azospirillum brasilense Sp245, Folia Microbiol., 2020a, vol. 65, pp. 963–972.
Shelud’ko, A.V., Filip’echeva, Y.A., Telesheva, E.M., Burov, A.M., Evstigneeva, S.S., Burygin, G.L., and Petrova, L.P., Characterization of carbohydrate-containing components of Azospirillum brasilense Sp245 biofilms, Microbiology (Moscow), 2018, vol. 87, pp. 610–620.
Sheludko, A.V., Kulibyakina, O.V., Shirokov, A.A., Petrova, L.P., Matora, L.Yu., and Katsy, E.I., The effect of mutations affecting synthesis of lipopolysaccharides and calcofluor-binding polysaccharides on biofilm formation by Azospirillum brasilense, Microbiology (Moscow), 2008, vol. 77, pp. 313–317.
Shelud’ko, A.V., Mokeev, D.I., Evstigneeva, S.S., Filip’echeva, Yu.A., Burov, A.M., Petrova, L.P., Ponomareva, E.G., and Katsy, E.I., Cell ultrastructure in biofilms of Azospirillum brasilense, Microbiology (Moscow), 2020, vol. 89, pp. 50–63.
Shelud’ko, A.V., Shirokov, A.A., Sokolova, M.K., Sokolov, O.I., Petrova, L.P., Matora, L.Yu., and Katsy, E.I., Wheat root colonization by Azospirillum brasilense strains with different motility, Microbiology (Moscow), 2010, vol. 79, pp. 688–695.
Shelud’ko, A.V., Varshalomidze, O.E., Petrova, L.P., and Katsy, E.I., Effect of genomic rearrangement on heavy metal tolerance in the plant-growth-promoting rhizobacterium Azospirillum brasilense Sp245, Folia Microbiol., 2012, vol. 57, pp. 5–10.
Stoderegger, K. and Herndl, G.J., Production and release of bacterial capsular material and its subsequent utilization by marine bacterioplankton, Limnol. Oceanogr., 1998, vol. 43, pp. 877–884.
Tak, H.I., Ahmad, F., and Babalola, O.O., Advances in the application of plant growth-promoting rhizobacteria in phytoremediation of heavy metals, Rev. Environ. Contam. Toxicol., 2013, vol. 223, pp. 33–52.
ACKNOWLEDGMENTS
The authors thank the IBPPM RAS Collection of Rhizosphere Microorganisms for A. brasilense strain Sp245 (IBPPM 219) and the Simbioz Center for the Collective Use of Research Equipment in the Field of Physical–Chemical Biology and Nanobiotechnology (IBPPM RAS, Saratov, Russia).
Funding
This study was supported in part by the Russian Foundation for Basic Research, project no. 20-04-00006-a. The assessment of the respiratory activity of cells was supported in part by the Razumovsky Saratov State Medical University, project no. SSMU-2021-001.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests. The authors declare that they have no conflict of interest.
Statement on the welfare of animals. This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by A. Panyushkina
Rights and permissions
About this article
Cite this article
Petrova, L.P., Filip’echeva, Y.A., Telesheva, E.M. et al. Variations in Lipopolysaccharide Synthesis Affect Formation of Azospirillum baldaniorum Biofilms in planta at Elevated Copper Content. Microbiology 90, 470–480 (2021). https://doi.org/10.1134/S002626172104010X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002626172104010X