Abstract
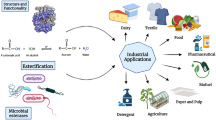
The successfulness of a lipase-catalyzed industrial process depends on a proper lipase selection. In this work, an alternative screening platform for industrially important biotransformations catalyzed by microbial lipases was proposed. Thus, the reactivity of sixty lipase activities from spore-forming microorganisms towards hydrolytic and transesterification reactions by using p-nitrophenyl palmitate as a chromogenic acyl donor substrate was explored. Only three biocatalysts were capable of catalyzing all reactions tested. Fourteen biocatalysts did not show hydrolytic activity at all; however, they displayed transesterification activities using ethanol, starch, low-methoxyl (LM) pectin, high-methoxyl (HM) pectin, or vitamin C as acyl acceptors. Using heat-treated biocatalysts, hydrolytic activities were not highly correlated with the corresponding transesterification activities using ethanol (r = −0.058, p = 0.660), starch (r = 0.431, p = 0.001), LM pectin (r = −0.010, p = 0.938), HM pectin (r = 0.167, p = 0.202), and vitamin C (r = −0.048, p = 0.716) as acyl acceptor. In addition, to the best of our knowledge, several transesterification activities produced from microorganisms of the genus Bacillus, Brevibacillus, Lysinibacillus, Geobacillus, or Sporosarcina were reported for first time. Finally, the global lipase market was presented and segmented by date, application, geography and player highlighting the commercial contribution of microbial lipases.
Graphical abstract









Similar content being viewed by others
References
Adak S, Banerjee R (2016) A green approach for starch modification: esterification by lipase and novel imidazolium surfactant. Carbohydr Polym 150:359–368. https://doi.org/10.1016/j.carbpol.2016.05.038
Alissandratos A, Halling PJ (2012) Enzymatic acylation of starch. Bioresour Technol 115:41–47. https://doi.org/10.1016/j.biortech.2011.11.030
Ansorge-Schumacher MB, Thum O (2013) Immobilised lipases in the cosmetics industry. Chem Soc Rev 42:6475–6490. https://doi.org/10.1039/C3CS35484A
Berlemont R, Spee O, Delsaute M, Lara Y, Schuldes J, Simon C, Power P, Daniel R, Galleni M (2013) Novel organic solvent-tolerant esterase isolated by metagenomics: insights into the lipase/esterase classification. Rev Argent Microbiol 45:3–12
Bezbradica D, Stojanović M, Veličković D, Dimitrijević A, Carević M, Mihailović M, Milosavić N (2013) Kinetic model of lipase-catalyzed conversion of ascorbic acid and oleic acid to liposoluble vitamin C ester. Biochem Eng J 71:89–96. https://doi.org/10.1016/j.bej.2012.12.001
Chandra P, Enespa SR, Aroa PK (2020) Microbial lipases and their industrial applications: a comprehensive review. Microb Cell Factories 19:169. https://doi.org/10.1016/j.scp.2020.100231
Chaves S, Pera LM, Avila CM, Romero CM, Baigorí M, Morán Vieyra FE, Borsarelli CD, Chehin R (2016) Towards efficient biocatalysts: photo-immobilization of a lipase on novel lysozyme amyloid-like nanofibrils. RSC Adv 6:8528–8538. https://doi.org/10.1039/C5RA19590J
Chen J, Liu W, Liu C-M, Li T, Liang R-H, Luo S-J (2015) Pectin modifications: a review. Crit Rev Food Sci Nutr 55:1684–1698. https://doi.org/10.1080/10408398.2012.718722
Ćorović M, Milivojević A, Simović M, Banjanac K, Pjanović R, Bezbradica RD (2020) Enzymatically derived oil-based L-ascorbyl esters: synthesis, antioxidant properties and controlled release from cosmetic formulations. Sustain Chem Pharm 15:100231. https://doi.org/10.1016/j.scp.2020.100231
Costas L, Bosio VE, Pandey A, Castro GR (2008) Effects of organic solvents on immobilized lipase in pectin microspheres. Appl Biochem Biotechnol 151:578–586. https://doi.org/10.1007/s12010-008-8233-0
Dettori L, Jelsch C, Guiavarc’h Y, Delaunay S, Framboisier X, Chevalot I, Humeau C (2018) Molecular rules for selectivity in lipase-catalyzed acylation of lysine. Process Biochem 74:50–60. https://doi.org/10.1016/j.procbio.2018.07.021
Dong-Mei L, Chen J, Yan-Ping S (2018) Advances on methods and easy separated support materials for enzymes immobilization. Trends Anal Chem 102:332–342. https://doi.org/10.1016/j.trac.2018.03.011
Geoffry K, Achur RN (2018) Screening and production of lipase from fungal organisms. Biocatal Agric Biotechnol 14:241–253. https://doi.org/10.1016/j.bcab.2018.03.009
Guncheva M, Zhiryakova D (2011) Catalytic properties and potential applications of Bacillus lipases. J Mol Catal B Enzym 68:1–21. https://doi.org/10.1016/j.molcatb.2010.09.002
Hasan F, Shah AA, Hameed A (2009) Methods for detection and characterization of lipases: a comprehensive review. Biotechnol Adv 27:782–798. https://doi.org/10.1016/j.biotechadv.2009.06.001
Huang X, Chen F, Sun B, Zhang H, Tian Y, Zhu C (2017) Isolation of a fluoroglycofen-degrading KS-1 strain and cloning of a novel esterase gene fluE. FEMS Microbiol Lett 364:16. https://doi.org/10.1093/femsle/fnx168
Javed S, Azeem F, Hussain S, Rasul I, Siddique MH, Riaz M, Afzal M, Kouser A, Nadeem H (2018) Bacterial lipases: a review on purification and characterization. Prog Biophys Mol Biol 132:23–34. https://doi.org/10.1016/j.pbiomolbio.2017.07.014
Jesionowski T, Zdarta J, Krajewska B (2014) Enzyme immobilization by adsorption: a review. Adsorption 20:801–821. https://doi.org/10.1007/s10450-014-9623-y
Karaki N, Aljawish A, Humeau C, Muniglia L, Jasniewski J (2016) Enzymatic modification of polysaccharides: mechanisms, properties, and potential applications: a review. Enzyme Microb Technol 90:1–18. https://doi.org/10.1016/j.enzmictec.2016.04.004
Karmee SK (2009) Biocatalytic synthesis of ascorbyl esters and their biotechnological applications. Appl Microbiol Biotechnol 81:1013–1022. https://doi.org/10.1007/s00253-008-1781-y
Li D, Wang W, Durrani R, Li X, Yang B, Wang Y (2016) Simplified enzymatic upgrading of high-acid rice bran oil using ethanol as a novel acyl acceptor. J Agric Food Chem 64:6730–6737. https://doi.org/10.1021/acs.jafc.6b02518
Li Q, Xu J, Du W, Li Y, Liu D (2013) Ethanol as the acyl acceptor for biodiesel production. Renew Sust Energ Rev 25:742–748. https://doi.org/10.1016/j.rser.2013.05.043
MarketsandMarkets™ (2018) Microbial lipase market by application (cleaning agents, animal feed, dairy products, bakery products, and confectionery products), form (powder and liquid), source (fungi and bacteria), and region - global forecast to 2023. https://www.marketsandmarkets.com/Market-Reports/microbial-lipase-market-248464055.html. Accessed 7 Apr 2021
Maurer S, Bayer R, Budde M, Kerber M, Farivar-Memar F, Däuwel J, Berg T, Rollie S (2016) Method for immobilizing and drying enzymes. Applicant: BASF SE [DE]. US2016312209 (A1)
Mesa M, Pereañez JA, Preciado LM, Bernal C (2018) How the Triton X-100 modulates the activity/stability of the Thermomyces lanuginose lipase: insights from experimental and molecular docking approaches. Int J Biol Macromol 120:2410–2417. https://doi.org/10.1016/j.ijbiomac.2018.09.009
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4325. https://doi.org/10.1093/nar/8.19.4321
O’Loughlin IB, Murray BA, Kelly PM, FitzGerald RJ, Brodkorb A (2012) Enzymatic hydrolysis of heat-induced aggregates of whey protein isolate. J Agric Food Chem 60:4895–4904. https://doi.org/10.1021/jf205213n
Pencreac`h G, Baratti JC, (2001) Comparison of hydrolytic activity in water and heptane for thirty-two commercial lipase preparations. Enzyme Microb Tecnol 28:473–479. https://doi.org/10.1016/S0141-0229(00)00355-0
Posocco B, Dreussi E, de Santa J, Toffoli G, Abrami M, Musiani F, Grassi M, Farra R, Tonon F, Grassi G, Dapas B (2015) Polysaccharides for the delivery of antitumor drugs. Materials 8:2569–2615. https://doi.org/10.3390/ma8052569
Rúa ML, Schmidt-Dannert C, Wahl S, Sprauer A, Schmid RD (1997) Thermoalkalophilic lipase of Bacillus thermocatenulatus large-scale production, purification and properties: aggregation behaviour and its effect on activity. J Biotechnol 56:89–102. https://doi.org/10.1016/S0168-1656(97)00079-5
Salvatierra HN, Regner EL, Baigori MD, Pera LM (2021) Orchestration an extracellular lipase production from Aspergillus niger MYA 135: biomass morphology and fungal physiology. AMB Express. https://doi.org/10.1186/s13568-021-01202-y
Talekar S, Joshi A, Joshi G, Kamat P, Haripurkar R, Kambale S (2013) Parameters in preparation and characterization of cross linked enzyme aggregates (CLEAs). RSC Adv 3:12485–12511. https://doi.org/10.1039/C3RA40818C
Tallur PN, Sajjan DB, Mulla SI, Talwar MP, Pragasam A, Nayak VM, Ninnekar HZ, Bhat SS (2016) Characterization of antibiotic resistant and enzyme producing bacterial strains isolated from the Arabian Sea. 3 Biotech 6:28. https://doi.org/10.1007/s13205-015-0332-3
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703. https://doi.org/10.1128/jb.173.2.697-703.1991
Winkler UK, Stuckmann M (1979) Glycogen, hyaluronate, and some other polysaccharides. J Bacteriol 138:663–670
WIPO (2021) PATENTSCOPE. https://www.wipo.int/patentscope/en/. Accessed 7 Apr 2021
Yadav AN, Sachan SG, Verma P, Kaushik R, Saxena AK (2016) Cold active hydrolytic enzymes production by psychrotrophic Bacilli isolated from three sub-glacial lakes of NW Indian Himalayas. J Basic Microbiol 56:294–307. https://doi.org/10.1002/jobm.201500230
Zhao J, Liu S, Gao Y, Ma M, Yan X, Cheng D, Wan D, Zeng Z, Yu P, Gong D (2021a) Characterization of a novel lipase from Bacillus licheniformis NCU CS-5 for applications in detergent industry and biodegradation of 2,4-D butyl ester. Int J Biol Macromol 176:126–136. https://doi.org/10.1016/j.ijbiomac.2021.01.214
Zhao J, Ma M, Zeng Z, Yu P, Gong D (2021b) Production, purification and biochemical characterization of a novel lipase from a newly identified lipolytic bacterium Staphylococcus caprae NCU S6. J Enzym Inhib Med Chem 36:248–256. https://doi.org/10.1080/14756366.2020.1861607
Zhao TT, No DS, Kim Y, Kim YS, Kim I-H (2014) Novel strategy for lipase-catalyzed synthesis of biodiesel using blended alcohol as an acyl acceptor. J Mol Catal B Enzym 107:17–22. https://doi.org/10.1016/j.molcatb.2014.05.002
Funding
This work was supported by FONCyT (PICT 2015–2596) and UNT (PIUNT 2020 D 681).
Author information
Authors and Affiliations
Contributions
Performed the research: MBA, LC. Analyzed the data: MBA, FdVL, MDB, LMP. Conceived of or designed the study: MDB, LMP. Wrote the paper: MDB, LMP.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abdulhamid, M.B., Costas, L., del Valle Loto, F. et al. Industrial biotransformations catalyzed by microbial lipases: screening platform and commercial aspects. Folia Microbiol 66, 1009–1022 (2021). https://doi.org/10.1007/s12223-021-00900-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-021-00900-1