Abstract
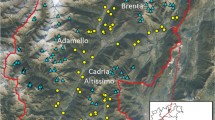
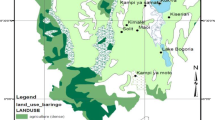
Many studies have inferred the way in which natural selection, genetic drift and gene flow shape the population genetic structures, but very few have quantified the population differentiation under spatially and temporally varying levels of selection pressure, population fluctuation and gene flow. In Nara Park (6.6 km2), central Japan, where several hundred sika deer (Cervus nippon) have been protected for more than 1,200 years, heavily- or moderately-haired nettle (Urtica thunbergiana) populations have evolved probably in response to intense deer browsing. Here, we analysed the genetic structure of two Nara Park populations and five surrounding populations using amplified fragment length polymorphism markers. A total of 546 marker loci were genotyped from 210 individuals. A Bayesian method estimated 5.5% of these loci to be outliers, which are putatively under natural selection. Neighbour-joining, principal coordinates and Bayesian clustering analyses using all-loci, non-outlier loci and outlier loci datasets showed that the Nara Park populations formed a cluster distinct from the surroundings. These results indicate the genome-wide differentiation of the Nara Park populations from the surroundings. Moreover, these imply the following: (1) gene flow is limited between these populations and thus genetic drift is a major factor causing the differentiation; and (2) natural selection imposed by intense deer browsing has contributed to some extent to the differentiation. In conclusion, sika deer seems to have counteracted genetic drift to drive the genetic differentiation of hairy nettles in Nara Park. This study suggests that a single herbivore species could lead to genetic differentiation among plant populations.






Similar content being viewed by others
References
Arany AM, de Jong TJ, van der Meijden E (2009) Herbivory and local genetic differentiation in natural populations of Arabidopsis thaliana (Brassicaceae). Plant Ecol 201:651–659. https://doi.org/10.1007/s11258-008-9530-y
Arrigo N, Holderegger R, Alvarez N (2012) Automated scoring of AFLPs using RawGeno v 20, a free R CRAN library. In: Pompanon F, Bonin A (eds) Data production and analysis in population genomics (methods in molecular biology, vol. 888). Humana Press, pp 155–175
Cheplick GP (2015) Approaches to plant evolutionary ecology. Oxford University Press
Di Pierro EA, Mosca E, Rocchini D, Binelli G, Neale DB, La Porta N (2016) Climate-related adaptive genetic variation and population structure in natural stands of Norway spruce in the South-Eastern Alps. Tree Genet Genomes 12:16. https://doi.org/10.1007/s11295-016-0972-4
Didiano TJ, Turley NE, Everwand G, Schaefer H, Crawley MJ, Johnson MT (2014) Experimental test of plant defence evolution in four species using long-term rabbit exclosures. J Ecol 102:584–594. https://doi.org/10.1111/1365-2745.12227
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Felsenstein J (2005) PHYLIP (Phylogeny Inference Package) Version 3.6. distributed by the author. Department of Genome Sciences, University of Washington
Foll M, Gaggiotti O (2008) A genome-scan method to identify selected loci appropriate for both dominant and codominant markers: a Bayesian perspective. Genetics 180:977–993. https://doi.org/10.1534/genetics.108.092221
Foundation for the Protection of Deer in Nara Park (2020) Census of the sika dee population in Nara Park. https://naradeer.com/learning/number.html.
Freeland JR (2020) Molecular ecology, 3rd edn. Wiley
Frei ER, Hahn T, Ghazoul J, Pluess AR (2014) Divergent selection in low and high elevation populations of a perennial herb in the Swiss Alps. Alp Bot 124:131–142. https://doi.org/10.1007/s00035-014-0131-1
Futuyma D, Kirkpatrick M (2018) Evolution, 4th edn. Oxford University Press
Garrido E, Andraca-Gómez G, Fornoni J (2012) Local adaptation: simultaneously considering herbivores and their host plants. New Phytol 193:445–453. https://doi.org/10.1111/j.1469-8137.2011.03923.x
Gonzalo-Turpin H, Hazard L (2009) Local adaptation occurs along altitudinal gradient despite the existence of gene flow in the alpine plant species Festuca eskia. J Ecol 97:742–751. https://doi.org/10.1111/j.1365-2745.2009.01509.x
Grdiša M, Radosavljević I, Liber Z, Stefkov G, Ralli P, Chatzopoulou PS, Carović-Stanko K, Šatović Z (2019) Divergent selection and genetic structure of Sideritis scardica populations from southern Balkan Peninsula as revealed by AFLP fingerprinting. Sci Rep 9:1–14. https://doi.org/10.1038/s41598-019-49097-x
Hamann E, Kesselring H, Armbruster GF, Scheepens JF, Stöcklin J (2016) Evidence of local adaptation to fine- and coarse-grained environmental variability in Poa alpina in the Swiss Alps. J Ecol 104:1627–1637. https://doi.org/10.1111/1365-2745.12628
Hirao AS, Shimono Y, Narita K, Wada N, Kudo G (2019) Ecotypic divergences of the alpine herb Potentilla matsumurae adapted to fellfield-snowbed habitats across a series of mountain sky islands. Am J Bot 106:772–787. https://doi.org/10.1002/ajb2.1290
Hirata R, Wasaka N, Fujii A, Kato T, Sato H (2019) Differences in flowering phenology, architecture, sexual expression and resource allocation between a heavily haired and a lightly haired nettle population: relationships with sika deer. Plant Ecol 220:255–266. https://doi.org/10.1007/s11258-019-00910-7
Holderegger R, Buehler D, Gugerli F, Manel S (2010) Landscape genetics of plants. Trends Plant Sci 15:675–683. https://doi.org/10.1016/j.tplants.2010.09.002
Iwamoto M, Horikawa C, Shikata M, Wasaka N, Kato T, Sato H (2014) Stinging hairs on the Japanese nettle Urtica thunbergiana have a defensive function against mammalian but not insect herbivores. Ecol Res 29:455–462. https://doi.org/10.1007/s11284-014-1137-2
Karkkäinen K, Ågren J (2002) Genetic basis of trichome production in Arabidopsis lyrata. Hereditas 136:219–226. https://doi.org/10.1034/j.1601-5223.2002.1360307.x
Karkkäinen K, Løe G, Ågren J (2004) Population structure in Arabidopsis lyrata: evidence for divergent selection on trichome production. Evolution 58:2831–2836. https://doi.org/10.1111/j.0014-3820.2004.tb01634.x
Kato T (2001) Nettles distributed in Nara Park. Nara Bot 23:11–14 (in Japanese)
Kato T, Ishida K, Sato H (2008) The evolution of nettle resistance to heavy deer browsing. Ecol Res 23:339–345. https://doi.org/10.1007/s11284-007-0387-7
Kivimäki M, Kärkkäinen K, Gaudeul M, Løe G, Ågren J (2007) Gene, phenotype and function: GLABROUS1 and resistance to herbivory in natural populations of Arabidopsis lyrata. Mol Ecol 16:453–462. https://doi.org/10.1111/j.1365-294X.2007.03109.x
Kohyama TI, Matsumoto K, Katakura H (2014) Deep phylogeographical structure and parallel host range evolution in the leaf beetle Agelasa nigriceps. Mol Ecol 23:421–434. https://doi.org/10.1111/mec.12597
Koskela T (2002) Variation in life-history traits among Urtica dioica populations with different history in parasitism by the holoparasitic plant Cuscuta europaea. Evol Ecol 16:433–454. https://doi.org/10.1023/A:1020860718125
Lehndal L, Ågren J (2015) Latitudinal variation in resistance and tolerance to herbivory in the perennial herb Lythrum salicaria is related to intensity of herbivory and plant phenology. J Evol Biol 28:576–589. https://doi.org/10.1111/jeb.12589
Marshall MM, Batten LC, Remington DL, Lacey EP (2019) Natural selection contributes to geographic patterns of thermal plasticity in Plantago lanceolata. Ecol Evol 9:2945–2963. https://doi.org/10.1002/ece3.4977
Martin LJ, Agrawal AA, Kraft CE (2015) Historically browsed jewelweed populations exhibit greater tolerance to deer herbivory than historically protected populations. J Ecol 103:243–249. https://doi.org/10.1111/1365-2745.12344
Meirmans PG (2015) Seven common mistakes in population genetics and how to avoid them. Mol Ecol 24:3223–3231. https://doi.org/10.1111/mec.13243
Miles LS, Rivkin LR, Johnson MT, Munshi-South J, Verrelli BC (2019) Gene flow and genetic drift in urban environments. Mol Ecol 28:4138–4151. https://doi.org/10.1111/mec.15221
Müller MH, Leppälä J, Savolainen O (2008) Genome-wide effects of postglacial colonization in Arabidopsis lyrata. Heredity 100:47–58. https://doi.org/10.1038/sj.hdy.6801057
Nei M (1972) Genetic distance between populations. Am Nat 106:283–292. https://doi.org/10.1086/282771
Nosil P (2012) Ecological speciation. Oxford University Press
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, Mcglinn D, Minchin PR, O’hara, RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2019) Package “Vegan”-community ecology package version 2.5–6. https://rdrr.io/cran/vegan/
Pollard AJ, Briggs D (1982) Genecological studies of Urtica dioica L. I. The nature of intraspecific variation in U. dioica. New Phytol 92:453–470. https://doi.org/10.1111/j.1469-8137.1982.tb03403.x
Pollard AJ, Briggs D (1984a) Genecological studies of Urtica dioica L. II. Patterns of variation at Wicken Fen, Cambridgeshire. England New Phytol 96:483–499. https://doi.org/10.1111/j.1469-8137.1984.tb03582.x
Pollard AJ, Briggs D (1984b) Genecological studies of Urtica dioica L. III. Stinging hairs and plant-herbivore interactions. New Phytol 97:507–522. https://doi.org/10.1111/j.1469-8137.1984.tb03615.x
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
R Core Team (2019) R ver. 3.6.2. R: A language and environment for statistical computing. R foundation for statistical computing. http://www.R-project.org/
Sampson JF, Byrne M (2012) Genetic diversity and multiple origins of polyploid Atriplex nummularia Lindl. (Chenopodiaceae). Biol J Linn Soc 105:218–230. https://doi.org/10.1111/j.1095-8312.2011.01787.x
Shikata M, Kato T, Shibata E, Sato H (2013) Among-population variation in resistance traits of a nettle and its relationship with deer habitat use frequency. Ecol Res 28:207–216. https://doi.org/10.1007/s11284-012-1007-8
Skaien CL, Arcese P (2020) Local adaptation in island populations of Plectritis congesta that differ in historic exposure to ungulate browsers. Ecology 101:e03054. https://doi.org/10.1002/ecy.3054
Spss IBM (2017) IBM SPSS Statistics 25 command syntax reference. IBM
Strasburg JL, Sherman NA, Wright KM, Moyle LC, Willis JH, Rieseberg LH (2012) What can patterns of differentiation across plant genomes tell us about adaptation and speciation? Phil Trans R Soc B Biol Sci 367:364–373. https://doi.org/10.1098/rstb.2011.0199
Tateishi Y (2006) Urticeae. In: Iwatsuki K, Boufford DE, Ohba H (eds) Flora of Japan, vol IIa. Angiospermae-Dicotyledoneae: Archichlamydeae (a). Kodansha, pp 78–81
Templeton AR (2006) Population genetics and microevolutionary theory. A John Wiley and Sons
The Scikit-Bio Development Team (2020). A bioinformatics library for data scientists, students, and developers. version 0.5.6. http://scikit-bio.org/
Torii H, Tatsuzawa S (2009) Sika deer in Nara Park: unique human-wildlife relations. In: McCullough DR, Tkatsuki S, Kaji K (eds) Sika deer: biology and management of native and introduced populations. Springer, pp 347–363
Vekemans X (2002) AFLP-SURV version 10. Distributed by the author. Laboratoire de Génétique et Ecologie Végétale, Université Libre de Bruxelles
Vourc’h G, Martin JL, Duncan P, Escarré J, Clausen TP (2001) Defensive adaptations of Thuja plicata to ungulate browsing: a comparative study between mainland and island populations. Oecologia 126:84–93. https://doi.org/10.1007/s004420000491
Westberg E, Ohali S, Shevelevich A, Fine P, Barazani O (2013) Environmental effects on molecular and phenotypic variation in populations of Eruca sativa across a steep climatic gradient. Ecol Evol 3:2471–2484. https://doi.org/10.1002/ece3.646
Wright S (1943) Isolation by distance. Genetics 28:114–138
Yamamoto K (1997) LIA32 for Windows95 ver. 0373. http://hp.vector.co.jp/authors/VA008416/index.html
Acknowledgements
We thank Todaiji Temple for the permission to research in the precincts. This study was supported by Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (No. 22570019).
Author information
Authors and Affiliations
Contributions
TK, MTK and HS conceived the study and planned study design. TK and HS collected materials from the field. MY grew nettles in the greenhouse and measured leaf characteristics. TK conducted the laboratory work and statistical analyses. HS wrote the early draft. All authors read, edited, and approved the final submitted version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Caroline Müller .
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kohyama, T.I., Yoshida, M., Kimura, M.T. et al. Intense browsing by sika deer (Cervus nippon) drives the genetic differentiation of hairy nettle (Urtica thunbergiana) populations. Oecologia 196, 1095–1106 (2021). https://doi.org/10.1007/s00442-021-04988-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-021-04988-7