Abstract
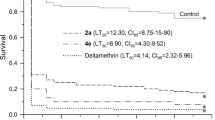
Plutella xylostella is a destructive cabbage pest responsible for huge economic losses. Heavy and continuous application of synthetic pesticides to control this pest has resulted in numerous environmental issues. Thus, discovering alternative methods to control P. xylostella, with reduced harmful effects has become crucial. In the present study we investigated the antifeedant activities and contact toxicities of two peptaibiotics, trichocellin A-I and B-II, produced by an endophytic fungus Trichoderma reesei against P. xylostella larvae. The two compounds were isolated using bioassay guided chromatographic methods from an ethyl acetate extract of T. reesei, isolated from the sedge Cyperus iria. The chemical structures of the two compounds were elucidated using mass spectrometric and NMR data. Both trichocellins showed 100% feeding deterrence activity against P. xylostella while the DC50 value for trichocellin A-I was 2.38 µg cm−2. The amount isolated was not sufficient to determine the DC50 value for trichocellin B-II. In addition, both compounds showed moderate contact toxicities against P. xylostella larvae having a mean motility around 40–50%. Trichocellin A-I and B-II, have previously been reported as having antibiotic properties. However, this is the first study to report the antifeedant and contact toxicities of trichocellin A-I and B-II against the economically important insect pest P. xylostella. The results of this study encourage further investigation of secondary metabolites of Trichoderma species for development as natural pest control agents in agriculture.




Similar content being viewed by others
Availability of data and material
Experimental procedures and NMR data are available under supplementary information.
References
Akhtar Y, Isman MB, Niehaus LA, Lee C, Lee H (2012) Antifeedant and toxic effects of naturally occurring and synthetic quinones to the cabbage looper, Trichoplusia ni. Crop Prot 31:8–14. https://doi.org/10.1016/j.cropro.2011.09.009
Akhtar Y, Shikano I, Isman MB (2009) Topical application of a plant extract to different life stages of Trichoplusia ni fails to influence feeding or oviposition behavior. Entomol Exp Appl 132:275–282. https://doi.org/10.1111/j.1570-7458.2009.00895.x
Akhtar Y, Yu Y, Isman MB, Plettner E (2010) Dialkoxybenzene and dialkoxyallylbenzene feeding and oviposition deterrents against the cabbage looper, Trichoplusia ni: potential insect behavior control agents. J Agric Food Chem 58:4983–4991. https://doi.org/10.1021/jf9045123
Alahmadi SS, Ouf SA, Ibrahim RA, El-Shaikh KA (2012) Possible control of date palm stag beetle, Lucanus cervus (L.) (Coleoptera: Lucanidae), using gut protease inhibitors of different biocontrol agents. Egypt J Biol Pest Control 22:93–101
Baron NC, Rigobelo EC, Zied DC (2019) Filamentous fungi in biological control: current status and future perspectives. Chil J Agric Res 79(2):307–315. https://doi.org/10.4067/S0718-58392019000200307
Campos EVR, de Oliveira JL, Pascoli M, de Lima R, Fraceto LF (2016) Neem oil and crop protection: from now to the future. Front Plant Sci 7:1494. https://doi.org/10.3389/fpls.2016.01494
Cantrell CL, Dayan FE, Duke SO (2012) Natural products as sources for new pesticides. J Nat Prod 75:1231–1242. https://doi.org/10.1021/np300024u
Coppola M, Cascone P, Di Lelio I, Woo SL, Lorito M, Rao R, Pennacchio F, Guerrier E, Digilio MC (2019) Trichoderma atroviride P1 colonization of tomato plants enhances both direct and indirect defense barriers against insects. Front Physiol 10:813. https://doi.org/10.3389/fphys.2019.00813
Daniel JFS, Filho ER (2007) Peptaibols of Trichoderma. Nat Prod Rep 24:1128–1141. https://doi.org/10.1039/B618086H
Frisvad JC, Moller LLH, Larsen TO, Kumar R, Arnau J (2018) Safety of the fungal workhorses of industrial biotechnology: update on the mycotoxin and secondary metabolite potential of Aspergillus niger, Aspergillus oryzae and Trichoderma reesei. Appl Microbiol Biotechnol 102:9481–9515. https://doi.org/10.1007/s00253-018-9354-1
Fujita T, Wada S, Iida A, Nishimura T, Kanai M, Toyama N (1994) Fungal Metabolites. XIII. Isolation and Structural Elucidation of New Peptaibols, Trichodecenins-I and -II, from Trichoderma viride. Chem Pharm Bull 42(3):489–494. https://doi.org/10.1248/cpb.42.489
Ikram M, Ali N, Jan G, Hamayun M, Jan FG, Iqbal A (2019) Novel antimicrobial and antioxidative activity by endophytic Penicillium roqueforti and Trichoderma reesei isolated from Solanum surattense. Acta Physiol Plant 41:164. https://doi.org/10.1007/s11738-019-2957-z
Kariyawasm GK, Mithrasena YJPK, Fernando THPS, Wijesundera RLC, Wijesundera WSS (2012) A new cost-effective method for extracting genomic DNA from fungi. Abstracts of Papers, 5th Annual Sessions of the Institute of Biochemistry, Molecular Biology and Biotechnology, Volume 5, Institute of Biochemistry, Molecular Biology and Biotechnology, Colombo, April 27, 2012. Pp49.
Khan RAA, Najeeb S, Hussain S, Xie B, Li Y (2020) Bioactive secondary metabolites from Trichoderma spp. Against Phytopathogenic Fungi Microorganisms 8:817. https://doi.org/10.3390/microorganisms8060817
Landowski CP, Huuskonen A, Wahl R, Westerholm-Parvinen A, Kanerva A, Hanninen A, Salovuori N, Penttila M, Natunen J, Ostermeier C, Helk B, Saarinen J, Saloheimo M (2015) Enabling low-cost biopharmaceuticals: A systematic approach to delete proteases from a well-known protein production host Trichoderma reesei. PLoS One 10
Landowski CP, Mustalahti E, Wahl R, Croute L, Sivasiddarthan D, Westerholm-Parvinen A, Sommer B, Ostermeier C, Helk B, Saarinen J, Saloheimo M (2016) Enabling low-cost biopharmaceuticals: high level interferon alpha-2b production in Trichoderma reesei. Microb Cell Fact 15:104. https://doi.org/10.1186/s12934-016-0508-5
Loiseleur O (2017) Natural products in the discovery of agrochemicals. Chimia 71:810–822. https://doi.org/10.2533/chimia.2017.810
Lorito M, Woo SL (2015) Trichoderma: A multi-purpose tool for integrated pest management. In: Lugtenberg B (ed) Principles of plant-microbe interactions, Springer International Publishing Switzerland. https://doi.org/10.1007/978-3-319-08575-3_36
Meletzus D, Eichenlaub R (1991) Transformation of the phytopathogenic bacterium Clavibacter michiganense subsp. michiganense by electroporation and development of a cloning vector. J Bacteriol 173:184–190. https://doi.org/10.1128/jb.173.1.184-190.1991
Mukherjee PK, Horwitz BA, Herrera-Estrella A, Schmoll M, Kenerley CM (2013a) Trichoderma research in the genome era. Annu Rev Phytopathol 51:105–129. https://doi.org/10.1146/annurev-phyto-082712-102353
Mukherjee PK, Horwitz BA, Singh US, Mukherjee M, Schmoll M (2013b) Trichoderma: Biology and Applications CABI Oxfordshire UK 13:9781780642475
Rantasalo A, Vitikainen M, Paasikallio T, Jantti J, Landowski CP, Mojzita D (2019) Novel genetic tools that enable highly pure protein production in Trichoderma reesei. Sci Rep 9:5032. https://doi.org/10.1038/s41598-019-41573-8
Ratnaweera PB, Jayasundara JMNM, Herath HHHMSD, Williams DE, Rajapaksha SU, Nishantha KMDWP, de Silva ED, Andersen RJ (2020) Antifeedant, contact toxicity and oviposition deterrent effects of phyllostine acetate and phyllostine isolated from the endophytic fungus Diaporthe miriciae against Plutella xylostella larvae. Pest Manag Sci 76:1541–1548. https://doi.org/10.1002/ps.5673
Ratnaweera PB, Walgama RC, Jayasundera KU, Herath SD, Abira S, Williams DE, Andersen RJ, de Silva ED (2018) Antibacterial activities of the endophytic fungi isolated from six Sri Lankan plants of the family Cyperaceae. Bangladesh J Pharmacol 13:264–272. https://doi.org/10.3329/bjp.v13i3.36716
Reino JL, Guerrero RF, Hernandez-Galan R, Collado IG (2008) Secondary metabolites from species of the biocontrol agent Trichoderma. Phytochem Rev 7:89–123. https://doi.org/10.1007/s11101-006-9032-2
Rodriguez-Gonzalez A, Carro-Huerga G, Mayo-Prieto S, Lorenzana A, Gutierrez S, Pelaez HJ, Casquero PA (2018) Investigations of Trichoderma spp. and Beauveria bassiana as biological control agent for Xylotrechus arvicola, a major insect pest in Spanish vineyards. J Econ Entomol 111(6):2585–2591. https://doi.org/10.1093/jee/toy256
Schmoll M, Dattenbock C, Carreras-Villasenor N et al (2016) The genomes of three uneven siblings: Footprints of the lifestyles of three Trichoderma species. Microbiol Mol Biol Rev 80:205–327. https://doi.org/10.1128/MMBR.00040-15
Shi M, Chen L, Wang XW, Zhang T, Zhao PB, Song XY, Sun CY, Chen XL, Zhou BC, Zhang YZ (2012) Antimicrobial peptaibols from Trichoderma pseudokoningii induce programmed cell death in plant fungal pathogens. Microbiology 158:166–175. https://doi.org/10.1099/mic.0.052670-0
Singh D, Raina TK, Singh J (2017) Entomopathogenic fungi: An effective biocontrol agent for management of insect populations naturally. Int J Pharm Sci Res 9(6):830–839. ISSN:0975–1459
Technical Bulletin of GoTaq (TBGT) (2019) PCR Core System. Promega, Madison, WI
Thapa S, Sotang N, Limbu A, Joshi A (2020) Impact of Trichoderma sp. in agriculture: A mini-review. J Biol Today’s World 9(7):227
USEPA (United States Environmental Protection Agency) (2019) https://www.epa.gov/
Vinale F, Marra R, Scala F, Ghisalberti EL, Lorito M, Sivasithamparam K (2006) Major secondary metabolites produced by two commercial Trichoderma strains active against different phytopathogens. Lett Appl Microbiol 43:143–148. https://doi.org/10.1111/j.1472-765X.2006.01939.x
Wada S, Iida A, Asami K, Tachikawa E, Fujita T (1997) Role of the Gln/Glu residues of trichocellins A-II/B-II in ion-channel formation in lipid membranes and catecholamine secretion from chromaffin cells. Biochim Biophys Acta 1325:209–214. https://doi.org/10.1016/S0005-2736(96)00260-X
Wada S, Nishimura T, Iida A, Toyama N, Fujita T (1994) Primary structures of antibiotic peptides, Trichocellins-A and -B from Trichoderma viride. Tetrahedron Lett 35(19):3095–3098. https://doi.org/10.1016/S0040-4039(00)76838-9
Yan SX, Shen QT, Xie ST, Chen XL, Sun CY, Zhang YZ (2006) Broad-spectrum antimicrobial activity and high stability of Trichokonins from Trichoderma koningii SMF2 against plant pathogens. FEMS Microbiol Lett 260:119–125. https://doi.org/10.1111/j.1574-6968.2006.00316.x
Yang S, Cao D, Wang G, Liu Y (2017) Identification genes involved in chemoreception in Plutella xylostella by antennal transcriptome analysis. Sci Rep 7:11941. https://doi.org/10.1038/s41598-017-11646-7
Yun BS, Yoo ID, Kim YH, Kim YS, Lee SJ, Kim KS, Yeo WH (2000) Peptaivirins A and B, two new antiviral peptaibols against TMV infection. Tetrahedron Lett 41:1429–1439. https://doi.org/10.1016/S0040-4039(99)02308-4
Zalucki MP, Shabbir A, Silva R, Adamson D, Shu-Sheng L, Furlong ML (2012) Estimating the economic cost of one of the world’s major insect pests, Plutella xylostella (Lepidoptera: Plutellidae): just how long is a piece of string? J Econ Entomol 105:1115–1129. https://doi.org/10.1603/EC12107
Zeilinger S, Gruber S, Bansal R, Mukherjee PK (2016) Secondary metabolism in Trichoderma – Chemistry meets genomics. Fungal Biol Rev 30:74–90. https://doi.org/10.1016/j.fbr.2016.05.001
Acknowledgements
This work was financially supported by the National Science Foundation, Sri Lanka research grant [RG/2016/EB/03] and Uva Wellassa University, Sri Lanka research grant [RG/2018/03] to PBR as well as Natural Sciences and Engineering Research Council (NSERC), Canada grant to RJA. Support and assistance provided by Agricultural Research Station, Seetha-eliya for insect larvae collection and rearing are also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Consent for publication
All authors give the consent for publication.
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ratnaweera, P.B., Madhushika, D.P.H., Jayasundara, J.M.N.M. et al. Antifeedant properties and contact toxicities of the trichocellins A-I and B-II from a Trichoderma reesei against Plutella xylostella larvae. Int J Trop Insect Sci 42, 845–854 (2022). https://doi.org/10.1007/s42690-021-00608-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-021-00608-2