Abstract
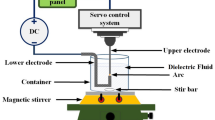
In this study, the electric spark discharge method was used to prepare a cuprous iodide nanocolloid (CuINC); specifically, an electrical discharge machine was used to prepare a CuINC under five sets of pulse width modulation (Ton–Toff) parameters, and ultraviolet–visible spectrophotometry and a zetasizer were used to evaluate the most suitable parameter set. Copper wires were used as electrodes (copper content = 99.7%, diameter = 1 mm), and deionized water mixed with iodine was used as the dielectric fluid. The analysis results indicated that the CuINC prepared under Ton–Toff = 10–10 µs had absorbance of 1.8 and a zeta potential of − 31.9 mV. The resultant CuINC had the highest concentration and suspension stability; this indicated that Ton–Toff = 10–10 µs is the most suitable parameter combination for preparing a CuINC. X-ray diffraction revealed a complete CuI crystal structure. Transmission electron microscopy images showed that most of the CuI nanoparticles were smaller than 5 nm and that the nanoparticles were evenly dispersed. The electric-discharge-based production process employed in this study is rapid and simple, and the end products have favorable suspension power. The method is a safe, environmentally friendly, and rapid method of preparing CuINCs.








Similar content being viewed by others
Abbreviations
- CuINC:
-
Cuprous iodide nanocolloid
- DC:
-
Direct current
- NC:
-
Nanocolloid
- CuINPs:
-
Cuprous iodide nanoparticles
- ESDM:
-
Electrical Spark Discharge Method
- EDX:
-
Energy-dispersive X-ray spectroscopy
- DW:
-
Deionized water
- UV–Vis:
-
Ultraviolet–Visible spectrophotometry
- NPs:
-
Nanoparticles
- XRD:
-
X-ray diffraction
- EDMs:
-
Electrical discharge machines
- TEM:
-
Transmission electron microscopy
References
D. G. Barceloux and D. Barceloux (1999). Copper. J. Toxicol. 37 (2), 217–230.
J. Wolff (1969). Iodide goiter and the pharmacologic effects of excess iodide. Am. J Med. 47 (1), 101–124.
A. Klapars and S. L. Buchwald (2002). Copper-catalyzed halogen exchange in aryl halides: an aromatic Finkelstein reaction. J. Am. Chem. Soc. 124 (50), 14844–14845.
C. Chen, P. Liu, and C. Lu (2008). Synthesis and characterization of nano-sized ZnO powders by direct precipitation method. Chem. Eng. J. 144 (3), 509–513.
S. X. Liu, X. Y. Chen, X. Chen, and C. L. Sun (2006). A novel high active TiO2/AC composite photocatalyst prepared by acid catalyzed hydrothermal method. Chin. Chem. Lett. 17 (4), 529–532.
D. G. Gromov, L. M. Pavlova, A. I. Savitskii, and A. Y. Trifonov (2015). Investigation of the early stages of condensation of Ag and Au on the amorphous carbon surface during thermal evaporation under vacuum. Phys. Solid State 57 (1), 173–180.
Q. Ye, P. Y. Liu, Z. F. Tang, and L. Zhai (2007). Hydrophilic properties of nano-TiO2 thin films deposited by RF magnetron sputtering. Vacuum 81 (5), 627–631.
K. H. Tseng, M. Y. Chung, and J. L. Chiu (2018). Application of nano-ag fabricated by the electrical spark discharge method for restraining aspergillus niger. Mater. Trans. 59 (7), 1101–1105.
M. Yang, J. Z. Xu, S. Xu, J. J. Zhu, and H. Y. Chen (2004). Preparation of porous spherical CuI nanoparticles. Inorg. Chem. Commun. 7 (5), 628–630.
Y. Zhou, G. Zhou, S. Wang, and S. Wang (2006). Preparation and photoluminescence of γ-CuI nanoparticles. Mater. Lett. 60 (17–18), 2184–2186.
P. M. Sirimanne, M. Rusop, T. Shirata, T. Soga, and T. Jimbo (2003). Characterization of CuI thin films prepared by different techniques. Mater. Chem. Phys. 80 (2), 461–465.
P. M. Sirimanne, M. Rusop, T. Shirata, T. Soga, and T. Jimbo (2002). Characterization of transparent conducting CuI thin films prepared by pulse laser deposition technique. Chem. Phys. Lett. 366 (5–6), 485–489.
Y. Yang, X. Li, B. Zhao, H. Chen, and X. Bao (2004). Nano-particulate CuI film formed on porous copper substrate by iodination. Chem. Phys. Lett. 387 (4–6), 400–404.
K. H. Ho and S. T. Newman (2003). State of the art electrical discharge machining (EDM). Int. J. Mach. Tools Manuf. 43 (13), 1287–1300.
K. H. Tseng, M. Y. Chung, and C. Y. Chang (2017). Parameters for fabricating nano-Au colloids through the electric spark discharge method with micro-electrical discharge machining. Nanomaterials 7 (6), 133.
M. Gostimirovic, P. Kovac, M. Sekulic, and B. Skoric (2012). Influence of discharge energy on machining characteristics in EDM. J. Mech. Sci. Technol. 26 (1), 173–179.
K. H. Tseng, Y. H. Lin, D. C. Tien, H. C. Ku, and L. Stobinski (2018). Stability analysis of platinum nanoparticles prepared by ESDM in deionized water. Micro Nano Lett. 13, 1545.
K. H. Tseng, H. C. Ku, D. C. Tien, and L. Stobinski (2019). Novel preparation of reduced graphene oxide–silver complex using an electrical spark discharge method. Nanomaterials 9 (7), 979.
K. H. Tseng, H. C. Ku, D. C. Tien, and L. Stobinski (2019). Parameter control and concentration analysis of graphene colloids prepared by electric spark discharge method. Nanotechnol. Rev. 8 (1), 201–209.
K. H. Tseng, C. Y. Chang, M. Y. Chung, and T. S. Cheng (2017). Fabricating TiO2 nanocolloids by electric spark discharge method at normal temperature and pressure. Nanotechnology 28 (46), 465701.
E. Hontañón, J. M. Palomares, M. Stein, X. Guo, R. Engeln, H. Nirschl, and F. E. Kruis (2013). The transition from spark to arc discharge and its implications with respect to nanoparticle production. J. Nanopart. Res. 15 (9), 1957.
H. E. De Bruyn and A. J. Pekelharing (1982). Has the «Delay Time» influence on the EDM-process? CIRP Ann. 31 (1), 103–106.
Y. S. Wong, M. Rahman, H. S. Lim, et al. (2003). Investigation of micro-EDM material removal characteristics using single RC-pulse discharges. J. Mater. Process. Technol. 140 (1), 303–307.
A. H. Qiang, L. M. Zhao, C. J. Xu, and M. Zhou (2007). Effect of dispersant on the colloidal stability of nano-sized CuO suspension. J. Dispers. Sci. Technol. 28 (7), 1004–1007.
R. K. Sahu, S. S. Hiremath, P. V. Manivannan, and M. Singaperumal (2014). Generation and characterization of copper nanoparticles using micro-electrical discharge machining. Mater. Manuf. Process. 29 (4), 477–486.
Acknowledgements
The authors would like to thank the Precision Research and Analysis Center, National Taipei University of Technology for technically supporting this research.
Funding
The authors would like to thank the Ministry of Science and Technology (MOST 108-2221-E-027-050-) for financial support of this research.
Author information
Authors and Affiliations
Contributions
Data curation, MC and DT; Formal analysis, WL; Funding acquisition, KT; Methodology, DT and WL; Project administration, KT; Resources, KT; Supervision, KT and LS; Validation, DT and LS; Writing—original draft, WL and DT; Writing—review & editing, LS and MC. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tseng, KH., Lin, WJ., Chung, MY. et al. Preparing Cuprous Iodide Nanocolloid by the Electrical Spark Discharge Method. J Clust Sci 33, 2069–2075 (2022). https://doi.org/10.1007/s10876-021-02127-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-021-02127-z