Abstract
Purpose
Grade 3 neuroendocrine tumor (NET G3) is a novel pathologic category within gastro-entero-pancreatic (GEP) neuroendocrine neoplasms (NENs) but its clinical behavior and therapeutic management still remain challenging. Prognostic and predictive factors aiding NET G3 management are needed.
Patients and methods
We performed a retrospective analysis from 2015 to 2020 of all patients with > 20% Ki-67, well-differentiated NETs evaluated within our NEN-dedicated multidisciplinary team. We divided the sample according the timing of NET G3 diagnosis, the radiotracers distribution and Ki-67. We analyzed the correlation between these NET G3 features and clinical outcomes.
Results
Among 3238 multidisciplinary discussion reports, we selected 55 patients, 48 from GEP and 7 from an occult GEP origin. In 45 patients, NET G3 diagnosis occurred at the beginning of clinical history (upfront-NET G3), whereas in 10, during the NET G1-G2 clinical history (late-NET G3).
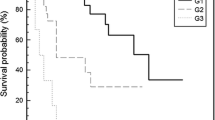
Patients with ≤ 30% (34/55) vs. > 30% Ki-67 (21/55) had a better overall survival (OS) (p = 0.042); patients with a homogeneous vs. inhomogeneous/negative 68Gallium(68Ga)-DOTA-Peptide Positron Emission Tomography (PET)/computed tomography (CT) showed a trend to a better OS, and a significant better progression-free survival (PFS) (p = 0.033). A better OS was observed for negative/inhomogeneous vs. homogeneous 18-fluorodeoxyglucose (18FDG)-PET/CT (p = 0.027). A trend to a better OS was reported in late- vs. upfront-NET G3, while the latter showed a significantly better response rate (RR) (p = 0.048).
Conclusion
Our findings suggested that Ki-67 cutoff, functional imaging and the timing to NET G3 diagnosis may help clinicians in more accurate selection of NET G3 management. Prospective studies are needed.




Similar content being viewed by others
Data availability
IEO shall be classified as autonomous Data Controllers pursuant to Regulation (EU) 2016/679 of the European Parliament and Council of 27 April 2016 (GDPR).
References
Basturk O, Yang Z, Tang Lh, Hruban Rh, Adsay V, Cm M et al (2015) The High-grade (who g3) pancreatic neuroendocrine tumor category is morphologically and biologically heterogenous and includes both well differentiated and poorly differentiated neoplasms. Am J Surg Pathol 39(5):683–690
Heetfeld M, Chougnet CN, Olsen IH, Rinke A, Borbath I, Crespo G et al (2015) Characteristics and treatment of patients with g3 gastroenteropancreatic neuroendocrine neoplasms. Endocr Relat Cancer 22(4):657–664
Velayoudom-Cephise Fl, Duvillard P, Foucan L, Hadoux J, Chougnet Cn, Leboulleux S et al (2013) Are G3 enets neuroendocrine neoplasms heterogeneous? Endocr Relat Cancer. 20(5):649–657
Sorbye H, Welin S, Langer SW, Vestermark LW, Holt N, Osterlund P et al (2013) Predictive and prognostic factors for treatment and survival in 305 patients with advanced gastrointestinal neuroendocrine carcinoma (who g3): the nordic nec study. Ann Oncol. 24(1):152–160
Milione M, Maisonneuve P, Spada F, Pellegrinelli A, Spaggiari P, Albarello L et al (2017) The clinicopathologic heterogeneity of grade 3 gastroenteropancreatic neuroendocrine neoplasms: morphological differentiation and proliferation identify different prognostic categories. Neuroendocrinology 104(1):85–93
Or LR, Kloppel G, Rosai J et al (2017) Who classification of tumours of endocrine organs (World Health Organization Classification Of Tumors), 4th edn. Iarc Press, Lyons France
Or NI, Klimstra D, Paradis V, Rugge M, Schirmacher P, Washington M, Carneiro F, Cree I (2019) Who classification of tumor editorial board; digestive system tumours, who classification of tumours, 5th edn. Iarc Press, France
Konukiewitz B, Am S, Jesinghaus M, Pfister D, Steiger K, Segler A et al (2017) Somatostatin receptor expression related to Tp53 and Rb1 alterations in pancreatic and extrapancreatic neuroendocrine neoplasms with A Ki67-index above 20. Mod Pathol 30(4):587–598
Yachida S, Vakiani E, Cm W, Zhong Y, Saunders T, Morgan R et al (2012) Small cell and large cell neuroendocrine carcinomas of the pancreas are genetically similar and distinct from well-differentiated pancreatic neuroendocrine tumors. Am J Surg Pathol 36(2):173–184
Dr S, Fc R, Ej S (2012) Molecular and cellular biology of neuroendocrine lung tumors: evidence for separate biological entities. Biochim Biophys Acta 1826(2):255–271
Ao V, Ia L, Mj M, Cy W, Pham T, Ee F et al (1997) Concordance of genetic alterations in poorly differentiated colorectal neuroendocrine carcinomas and associated adenocarcinomas. J Natl Cancer Inst 89(19):1448–1453
Kloppel G (2019) Neuroendocrine neoplasms: two families with distinct features unified in one classification (German version). Pathologe 40(3):211–219
Gm BJ, Wittekind C (2016) Tnm classification of malignant tumours, 8th edn. Wiley
Dy L, Yi K (2019) Prognostic value of maximum standardized uptake value in 68ga-somatostatin receptor positron emission tomography for neuroendocrine tumors: a systematic review and meta-analysis. Clin Nucl Med 44(10):777–783
Zhang P, Yu J, Li J, Shen L, Li N, Zhu H et al (2018) Clinical and prognostic value of pet/ct imaging with combination of (68)Ga-Dotatate and (18)F-Fdg in gastroenteropancreatic neuroendocrine neoplasms. Contrast Media Mol Imaging 2018:2340389
Karfis I, Marin G, Levillain H, Drisis S, Muteganya R, Critchi G et al (2020) Prognostic value of a three-scale grading system based on combining molecular imaging with (68)ga-dotatate and (18)f-fdg pet/ct in patients with metastatic gastroenteropancreatic neuroendocrine neoplasias. Oncotarget 11(6):589–599
Strosberg J, El-Haddad G, Wolin E, Hendifar A, Yao J, Chasen B et al (2017) Phase 3 trial of (177)lu-dotatate for midgut neuroendocrine tumors. N Engl J Med 376(2):125–135
Severi S, Nanni O, Bodei L, Sansovini M, Ianniello A, Nicoletti S et al (2013) Role of 18fdg pet/ct in patients treated with 177lu-Dotatate for advanced differentiated neuroendocrine tumours. Eur J Nucl Med Mol Imaging 40(6):881–888
Binderup T, Knigge U, Loft A, Federspiel B, Kjaer A (2010) 18f-fluorodeoxyglucose positron emission tomography predicts survival of patients with neuroendocrine tumors. Clin Cancer Res 16(3):978–985
Sorbye H, Baudin E, Borbath I, Caplin M, Chen J, Cwikla JB et al (2019) Unmet needs in high-grade gastroenteropancreatic neuroendocrine neoplasms (Who G3). Neuroendocrinology 108(1):54–62
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
All authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no financial or personal relationships that may have inappropriately influenced them in this study.
Ethical approval
The retrospective analysis complies with the Declaration of Helsinki and was approved by our local internal review board (IRB), and the IEO Data Protection Officer, with the U-ID of 2259.
Research involving human participants and/or animals
The retrospective analysis complies with the Declaration of Helsinki and was approved by our local internal review board (IRB), and the European Institute of Oncology Data Protection Officer, with the U-ID of 2259.
Informed consent
As a retrospective study, a specific informed consent was not needed and all collected data were anonymized. Every patient gave the consent for the research, as required by the Institution regulation.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Laffi, A., Spada, F., Bagnardi, V. et al. Gastroenteropancreatic grade 3 neuroendocrine tumors: a single entity or a heterogeneous group? A retrospective analysis. J Endocrinol Invest 45, 317–325 (2022). https://doi.org/10.1007/s40618-021-01642-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-021-01642-0