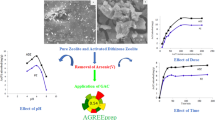
Abstract
Purpose
Pollution of surface water and groundwater by bulky molecules such as pesticides has been recognized as a major problem in many countries due to their persistence in aquatic environment and potential adverse health effects. The main purpose of this study is the development of a capable adsorbent to remove these bulky molecules from wastewater such as the pesticide Mesosulfuron-Methyl (MM) by reducing the diffusion path, to overcome the problems of diffusional limitations on microporous adsorbents.
Methods
The adsorption of mesosulfuron-methyl (MM) from aqueous solution is curried out using treated acid HY zeolite. Batch sorption equilibrium and kinetic experiments are conducted to evaluate the efficiency of these materials. Parent zeolites and their derivatives have been characterized by nitrogen adsorption–desorption, pyridine chemisorption followed by infrared spectroscopy and X-ray fluorescence.
Results
The acid treatment leads to an increase in the specific surface from 691 to 853 m2 g− 1 for HY(30) and from 631 to 806 m2 g− 1 for the HY(16.6) zeolites. It also leads to a reduction in Lewis acidity from 74 to 25 µmol g− 1 and from 135 to 31 µmol g− 1 for HY(30) and HY(16.6) zeolites respectively, and increases the adsorbent-adsorbate interaction. The adsorption capacity increased from 83 to 99 % after acid treatment. The equilibrium adsorption time is decreased from 15 h to 10 min for the HY(30)_A and from 20 h to 20 min for the HY(16.6)_A for an initial concentration of 20 mg L− 1. The adsorption capacity depends on the pH solution, and the neutral form of the MM is more easily adsorbed into zeolite than the dissociated form via the framework bridged oxygen atoms. For all the samples, the pseudo-second-order kinetic model fits very well with the experimental data. In the case of the modified zeolites, the approaching equilibrium factor Rw decreases from 0.08183 to 0.00008 when the Lewis acid sites decrease; indicating that the equilibrium is reached more quickly. S-shape adsorption isotherms indicates that cooperative adsorption phenomena. Nevertheless, the shape of acid treated zeolites evolves to an L type indicating a significant enhancement of the adsorbent – adsorbate interactions inducing better adsorption efficiency.
Conclusions
Mesosulfuron-methyl adsorption has been successfully enhanced after acid treatments of zeolites HY.







Similar content being viewed by others
References
Tanga Z, Zhang W, Chen Y. Adsorption and desorption characteristics of monosulfuron in Chinese soils. J Hazard Mater. 2009;166:1351–6.
VenkataLaxma Reddy P, Kim KH. A review of photochemical approaches for the treatment of a wide range of pesticides. J Hazard Mater. 2015;285:325–35.
Fadaei A, Dehghani MH, Nasseri S, Mahvi AH, Rastkari N, Shayeghi M. Organophosphorous pesticides in surface water of Iran. Bull Environ Contam Toxicol. 2012;88:867–9.
Mirzadeh SS, Khezri SM, Rezaei S, Forootanfar H, Mahvi AH, Faramarzi MA. Decolorization of two synthetic dyes using the purified laccase of Paraconiothyriumvariabile immobilized on porous silica beads. J Environ Health Sci Eng. 2014;12:6.
Bazrafshan E, Mahvi AH, Nasseri S, Shaieghi M. Performance evaluation of electrocoagulation process for diazinon removal from aqueous environments by using iron electrodes. Iran J Environ Health Sci Eng. 2007;4:127–32.
Asadgol Z, Forootanfar H, Rezaei S, Mahvi AH, Faramarzi MA. Removal of phenol and bisphenol-a catalyzed by laccase in aqueous solution. J Environ Health Sci Eng. 2014. https://doi.org/10.1186/2052-336X-12-93.
Kamani H, Safari GH, Asgari G, Ashrafi SD. Data on modeling of enzymatic elimination of Direct Red 81 using response surface methodology. Data Brief. 2018;18:80–6.
Ashrafi SD, Rezaei S, Forootanfar H, Mahvi AH, Faramazi MA. The enzymatic decolorization and detoxification of synthetic dyes by the laccase from a soil-isolated ascomycete Paraconiothyrium variabile. Int Biodeterior Biodegrad. 2013;85:173–81.
Selcuk H. Decolorization and detoxification of textile wastewater by ozonation and coagulation processes. Dyes Pigment. 2005;64:217–22.
Kasmi T, Soualah A, Mignard S, Batonneau-Gener I. Effect of Brønsted acidity of HY zeolites in adsorption of methylene blue and comparative study with bentonite. J Environ Health Sci Eng. 2018;16:239–47.
Shirmardi M, Mesdaghinia A, Mahvi AH, Nasseri S, Nabizadeh R. Kinetics and equilibrium studies on adsorption of acid red 18 (Azo-Dye) using multiwall carbon nanotubes (MWCNTs) from aqueous solution. E-J Chem. 2012;9:2371–83.
Ashrafi SD, Kamani H, Mahvi AH. The optimization study of direct red 81 and methylene blue adsorption on NaOH-modified rice husk. Des Water Treat. 2014;57(2):738–46.
Andrejkovičová S, Sudagar A, Rocha J, Patinha C, Hajjaji W, Ferreira da Silva E, Velosa A, Rocha F. The effect of natural zeolite on microstructure, mechanical and heavy metals adsorption properties of metakaolin based geopolymers. Appl Clay Sci. 2016;126:141–52.
Ganiyu SO, Bispo C, Bion N, Ferreira P, Batonneau-Gener I. Periodic mesoporous organosilicas as adsorbents for the organic pollutants removal in aqueous phase. Microporous Mesoporous Mater. 2014;200:117–23.
Bernardoni F, Y.Fadeev A. Adsorption and wetting characterization of hydrophobic SBA-15 silicas. J Colloid Interface Sci. 2011;356:690–8.
Hamoudi S, El-Nemr A, Belkacemi K. Adsorptive removal of dihydrogenphosphate ion from aqueous solutions using mono, di- and tri-ammonium-functionalized SBA-15. J Colloid Interface Sci. 2010;343:615–21.
Jamil TS, Abdel-Ghafar HH, Ibrahim HS, Abd-El-Maksoud IH. Removal of methylene blue by two zeolites prepared from naturally occurring Egyptian kaolin as cost effective technique. Solid State Sci. 2011;13:1844–51.
Martucci A, Pasti L, Marchetti N, Cavazzini A, Dondi F, Alberti A. Adsorption of pharmaceuticals from aqueous solutions on synthetic zeolites. Microporous Mesoporous Mater. 2012;148:174–83.
Mirodatos C, Barthomeuf DJ. Superacid sites in zeolites. J Chem Soc Chem Commun. 1981;2:39–40.
Mirandatos C, Ha BH, Otsuka K, Barthomeuf D. Proceedings of the 5th International Conference on Zeolites, In: Rees, editor, Heyden, London; 1980. p. 382.
Kondo JN, Nishitani R, Yoda E, Yokoi T, Tatsumi T, Domen K. A comparative IR characterization of acidic sites on HY zeolite by pyridine and CO probes with silica-alumina and γ-alumina references. Phys Chem Chem Phys. 2010;12(37):115 76-86.
Boréave A, Auroux A, Guimon C. Nature and strength of acid sites in HY zeolites: a multitechnical approach. Microporous Mater. 1997;11(5–6):275–91.
Barthomeuf D. A general hypothesis on zeolites physicochemical properties. Applications to adsorption, acidity, catalysis, and electrochemistry. J Phys Chem. 1979;83:249–256.
Henriques CA, Monteiro JLF, Magnoux P, Guisnet M. Characterization of the cokeformed during-xylene isomerisationoverMordenites at various temperatures. J Catal. 1997;172:436–45.
Wu P, Komatsu T, Yashima TJ, Chem SOC. Faraday Trans. 1996;92(5):861.
Yonli AH, Batonneau-Gener I, Koulidiati J. Adsorptive removal of α-endosulfan from water by hydrophobic zeolites. An isothermal study. J Hazard Mater. 2012;204–203:357–62.
Marques JP, Gener I, Ayrault P, Bordado JC, Lopes JM, Ribeiro FR, Guisnet M. Dealumination of HBEA zeolite by steaming and acid leaching: distribution of the various aluminic species and identification of the hydroxyl groups. C R Chimie. 2005;8:399–410.
Guisnet M, Gilson JP. In: Zeolites for Cleaner Technologies. Catalytic Science series. GJ Hutchings editor. London: Imperial College Press. 2002;3:5.
Guisnet M, Ayrault P, Datka J. Acid properties of dealuminated mordenites studied by IR spectroscopy. 2. Concentration, acid strength and heterogeneity of OH groups. Pol J Chem. 1997;71:1455–61.
Yousefi M, Nabizadeh R, Alimohammadi M, Mohammadi AA, Mahvi AH. Removal of phosphate from aqueous solutions using granular ferric hydroxide process optimization by response surface methodology. Desalin Water Treat. 2019;158:290–300.
SharifiS, S, Nabizadeh R, Akbarpour B, Azari A, Ghaffari HR, Nazmara B, Mahmoudi B, Shiri L, Yousefi M. Modeling and optimizing parameters affecting hexavalent chromium adsorption from aqueous solutions using Ti-XAD7 nanocomposite: RSM-CCD approach, kinetic, and isotherm studies. J Environ Health Sci Eng. 2019;17:873–88.
Serrano DP, Garcia RA, Linares M, Gil B. Influence of the calcination treatment on the catalytic properties of hierarchical ZSM-5. Catal Today. 2012;179:91–101.
Koubaissy B, Joly G, Batonneau-Gener I, Magnoux P. Adsorptive removal of aromatic compounds present in wastewater by using dealuminated faujasite zeolite. Ind Eng Chem Res. 2011;50(9):5705–13.
Mrozik W, Stefanska J. Adsorption and biodegradation of antidiabetic pharmaceuticals in soils. Chemosphere. 2014;95:281–8.
Ghadiri SK, Nabizadeh R, Mahvi AH, Naseri S, Kazemian H, Mesdaghinia AR, Nazmara SH.Methyl tert-butyl ether adsorption on surfactant modified natural zeolites. Iran J Environ Health Sci Eng 2010;7:241–252.
Hay JV. Chemistry of Sulfonylurea Herbicides. J Pestic Sci. 1990;29:247–61.
Pusino A, Fiori MG, Braschi I, Gessa C. Adsorption and desorption of triasulfuron by soil. J Agric Food Chem. 2003;51:5350–4.
Mirzaei N, Ghaffari HR, Sharafi K, Velayati A, Hoseindoost G, Rezaei S, Mahvi AH, Azari A, Dindarloo K. Modified natural zeolite using ammonium quaternary based material for Acid red 18 removals from aqueous solution. J Environ Chem Eng. 2017;15(4):3151–60.
Shirmardi M, Mahvi AH, Hashemzadeh B, Naeimabadi A, Hassani G, Vosoughi Niri M. The adsorption of malachite green (MG) as a cationic dye onto functionalized multi walled carbon nanotubes. Korean J Chem Eng. 2013;30:1603–8.
Dogan M, Alkan M, Türkyilmaz A, Özdemir Y. Kinetics and mechanism of removal of methylene blue by adsorption onto perlite. J Hazard Mater. 2004;B109:141–8.
Balarak D, Mostafapour FK, Bazrafshan E, Mahvi AH, The equilibrium, kinetic, and thermodynamic parameters of the adsorption of the fluoride ion on to synthetic nanosodalite zeolite. Fluoride. 2017;50(2):223–34.
Wu FC, Tseng RL, Huang SC, Juang RS. Characteristics of pseudo-second-order kinetic model for liquid-phase adsorption: A mini-review. Chem Eng J. 2009;151:1–9.
Batonneau-gener I, Yonli A, Hazael-pascal S, Marques JP, Lopes JM, Guisnet M, Ribeiro FR, Mignard S. Influence of steaming and acid-leaching treatments on the hydrophobicity of HBEA zeolite determined under static conditions. Microporous Mesoporous Mater. 2008;110:480–7.
Weng CH, Pan YF. Adsorption of a cationic dye (methylene blue) onto spent activated clay. J Hazard Mater. 2007;144:355–62.
Giles CH, Smith D, Huitson A. A general treatment and classification of the soluteadsorption isotherm. J Colloid Interface Sci. 1974;47:755–65.
Jin X, Yu B, Chen Z, Arocena MJ, Thring RW. Adsorption of Orange II dye in aqueous solution onto surfactant-coated zeolite: Characterization, kinetic and thermodynamic studies. J Colloid Interface Sci. 2014;435:15–20.
Juang LC, Wang CC, Lee CK. Adsorption of basic dyes onto MCM-41. Chemosphere. 2006;64:1920–8.
Dada AO, Olalekan AP, Olatunya AM, Dada O, Langmuir F. Temkin and Dubinin–Radushkevich isotherms studies of equilibrium sorption of Zn2+ unto phosphoric acid modified rice husk. J Appl Chem. 2012;3:38–45.
Acknowledgements
The authors would like to acknowledge the support provided by both the Algerian and French governments for funding this work through project Tassili No. 12-MDU/859. We are grateful to Faculty of technology, Abderrahmane Mira University of Bejaia for its support. We also present our thanks to Miss Merieme Manseur (teacher of English language at secondary level) for her support.
Funding
This work was supported by both the Algerian and French governments [project Tassili No. 12-MDU/859].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kasmi-Belouzir, T., Soualah, A., Kouachi, K. et al. Effect of acid treated HY zeolites in adsorption of mesosulfuron-methyl. J Environ Health Sci Engineer 19, 1435–1445 (2021). https://doi.org/10.1007/s40201-021-00698-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40201-021-00698-7