Abstract
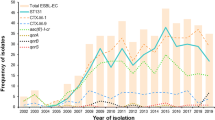
The emergence of carbapenem-resistant (CR) Escherichia coli obliges an assessment of such strains’ molecular epidemiology. Accordingly, we characterized in detail a globally distributed collection of CR E. coli isolates, then explored for associations between geographical origin and bacterial traits, and between different bacterial traits. We used established PCR-based assays and broth microdilution MIC determinations to characterize 343 global CR (i.e., non-susceptible to ≥ 1 carbapenem) extraintestinal E. coli isolates (2002–2017) for diverse molecular traits—including phylogroups, sequence types (STs), beta-lactamase genes, and 51 virulence genes—and susceptibility to 12 relevant antimicrobial agents. The study population was tremendously diverse according to all assessed variables. Nonetheless, certain geographically aligned, unifying themes emerged. These included an association of an Asia/West Pacific origin with non-B2/D/F phylogroups and STs, lower molecularly inferred virulence, more extensive resistance, and specific resistance genes (notably, metallo-beta-lactamases). Likewise, U.S. isolates from the central region, vs. other regions, were more virulent-appearing and more often from phylogroup B2 and ST131, but less extensively resistant and more often carbapenemase-gene negative. The global CR E. coli population is highly diverse according to multiple characteristics and varies significantly by geographical region. This predictably will pose challenges for prevention and management, and obliges ongoing surveillance.
Similar content being viewed by others
Availability of data and material
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Russo TA, Johnson JR (2003) Medical and economic impact of extraintestinal infections due to Escherichia coli: an overlooked epidemic. Microbes Infect 5:449–456
Bonten M, JR J, van den Biggelaar A, Georgalis L, Geurtsen J, Ibarra de Palacios P, Gravenstein S, Verstraeten T, Hermans P, Poolman J, (2021) Epidemiology of Escherichia coli bacteremia: a systematic literature review. Clin Infect Dis 72:1211–1229
Vihta K-D, Stoesser N, Llewelyn MJ, Quan TP, Davies T, Fawcett NJ, Dunn L, Jeffery K, Butler CC, Hayward G, Andersson M, Morgan M, Oakley S, Mason A, hopkins S, Wyllie DH, Crook DW, Wilcox MH, Johnson AP, Peto TEA, Walker AS, (2018) Trends over time in Escherichia coli bloodstream infections, urinary tract infections, and antibiotic susceptibilities in Oxfordshire, UK, 1998–2016: a study of electronic health records. Lancet Infect Dis 18:1138–1149
Peirano G, Bradfor PA, Kazmierczak KM, Badal RE, Hackel M, Hoban DJ, Pitout JDD (2014) Global incidence of carbapenemase-producing Escherichia coli ST131. Emerg Inf Dis 20:1928–1931
Mushtaq S, Irfan S, Sarma J, Doumith M, Pike R, Pitout J, Livermore D, Woodford N (2011) Phylogenetic diversity of Escherichia coli strains producing NDM-type carbapenemases. J Antimicrob Chemother 66:2002–2005
Liu X, Thungrat K, Boothe D (2016) Occurrence of OXA-48 carbapenemase and other β-lactamase genes in ESBL-producing multidrug resistant Escherichia coli from dogs and cats in the United States, 2009–2013. Front Microbiol 7:1057. https://doi.org/10.3389/fmicb.2016.01057
Cai J, Zhang R, Hu Y, Zhou H, Chen G (2014) Emergence of Escherichia coli sequence type 131 isolates producing KPC-2 carbapenemase in China. Antimicrob Agents Chemother 58:1146–1152
Livorsi DJ, Chorazy ML, Schweizer MS, Balkenende EC, Blevins AE, Nair R, Samore MH, Nelson RE, Khader K, Perencevich EN (2018) A systematic review of the epidemiology of carbapenem-resistant Enterobacteriaceae in the United States. Antimicrob Resist Infect Control 7:55
Colpan A, Johnston B, Porter S, Clabots C, Anway R, Thao L, Kuskowski MA, Tchesnokova V, Sokurenko EV, Johnson JR, VICTORY (Veterans Influence of Clonal Types on Resistance: Year, (2011) Investigators (2013) Escherichia coli sequence type 131 (ST131) as an emergent multidrug-resistant pathogen among U.S. veterans. Clin Infect Dis 57:1256–1265
CDC (2019) Antibiotic resistance threats in the United States, 2019. In: U.S. Department of Health and Human Services. https://doi.org/10.15620/cdc:82532
Hawkey PM, Warren RE, Livermore DM, McNulty CAM, Enoch DA, Otter JA, Wilson PR (2018) Treatment of infections caused by multidrug-resistant Gram-negative bacteria: report of the British society for antimicrobial chemotherapy/healthcare infection society/british infection association joint working party. J Antimicrob Chemother 73(suppl_3):iii2–iii78. https://doi.org/10.1093/jac/dky027.
Chang Y-T, Siu LK, Wantg J-T, Wu T-L, Chen Y-H, Chuang Y-C, Lin J-C, Lu P-L (2019) Resistance mechanisms and molecular epidemiology of carbapenem-nonsusceptible Escherichia coli in Taiwan, 2012–2015. Infect Drug Resist 12:2113–2123
Ellaby N, Doumith M, Hopkins K, Woodford N, Ellington M (2019) Emergence of diversity in carbapenemase-producing emopenEscherichia coliemclose ST131, England, January 2014 to June 2016. Euro Surveill 24:1800627. https://doi.org/10.2807/1560-7917.ES.2019.24.37.1800627
Patiño-Navarrete R, Rosinski-Chupin I, Cabanel N, Gauthier L, Takissian J, Madec J-Y, Hamze M, Bonnin R, A, Naas T, Glaser P, (2020) Stepwise evolution and convergent recombination underlie the global dissemination of carbapenemase-producing Escherichia coli. Genome Med 12:10
Tian X, Zheng X, Sun Y, Fang R, Zhang S, Zhang X, Lin J, Cao J, Zhou T (2020) Molecular mechanisms and epidemiology of carbapenem-resistant Escherichia coli isolated from Chinese patients during 2002–2017. Infect Drug Resist 13:501–512
Johnston B, Thuras P, Porter S, Anacker M, VonBank B, Vagnone Snippes P, Witwer M, Castanheira M, Johnson J (2020) Activity of imipenem/relebactam against carbapenem-resistant Escherichia coli isolates from the United States in relation to clonal background, resistance genes, co-resistance, and region. Antimicrob Agents Chemother 64:e02408-02419
Clinical and Laboratory Standards Institute (2020) M100: performance standards for antimicrobial susceptibility testing, 30th edn. CLSI, Wayne, PA
Clermont O, Christenson JK, Denamur E, Gordon DM (2012) The Clermont Escherichia coli phylo-typing method revisited: improvement of specificity and detection of new phylo-groups. Environ Microbiol Rep 5:58–65
Johnson JR, Johnston BD, Porter SB, Clabots C, Bender TL, Thuras P, Trott DJ, Cobbold R, Mollinger J, Ferrieri P, Drawz S, Banerjee R (2019) Rapid emergence, subsidence, and molecular detection of Escherichia coli sequence type 1193-fimH64 (ST1193-H64), a new disseminated multidrug-resistant commensal and extraintestinal pathogen. J Clin Microbiol 57:e01664-e1618
Clermont O, Christenson JK, Daubie A, Gordon DM, Denamur E (2014) Development of an allele-specific PCR for Escherichia coli B2 sub-typing, a rapid and easy to perform substitute of multilocus sequence typing. J Microbiol Methods 101:24–27
Johnson JR, Porter S, Thuras P, Castanheira M (2017) Epidemic emergence in the United States of Escherichia coli sequence type 131-H30 (ST131-H30), 2000–2009. Antimicrob Agents Chemother 61:pii: e00732–00717
Johnson JR, Davis G, Clabots C, Johnston BD, Porter S, Debroy C, Pomputius W, Ender PT, Cooperstock M, Slater BS, Banerjee R, Miller S, Kisiela D, Sokurenko E, Aziz M, Price LB (2016) Household clustering of Escherichia coli sequence type 131 clinical and fecal isolates according to whole genome sequence analysis. Open Forum Infect Dis 3:ofw129
Campbell I (2007) Chi-squared and Fisher-Irwin tests of two-by-two tables with small sample recommendations. Statist Med 26:3661–3675
Munoz-Price LS, Poirel L, Bonomo RA, Schwaber MJ, Daikos G, Cormican M, Cornaglia G, Garau J, Gniadkowski M, Hayden MK, Kumaraswamy K, Livermore DM, Maya JJ, Nordmann P, Patel JB, Paterson D, Pitout J, Villegas MV, Wang H, Woodford N, Quinn JP (2013) Clinical epidemiology of the global expansion of Klebsiella pneumoniae carbapenemases. Lancet Infect Dis 13:785–796
van Duin D, Doi Y (2017) The global epidemiology of carbapenemase-producing Enterobacteriaceae. Virulence 8:460–469
Logan LK, Weinstein RA (2017) The epidemiology of carbapenem-resistant Enterobacteriaceae: the impact and evolution of a global menace. J Infect Dis 215(S1):S28–S36
Matsumura Y, Johnson JR, Yamamoto M, Nagao M, Tanaka K, Takakura S, Ichiyama S, Kyoto-Shiga Clinical Microbiology Study Group (2015) CTX-M-27- and CTX-M-14-producing, ciprofloxacin-resistant Escherichia coli of the H30 subclonal group within ST131 drive a Japanese regional ESBL epidemic. J Antimicrob Chemother 70:1639–1649
Matsumura Y, Pitout J, Gommi R, Matsuda T, Noguchi T, Yamamoto M, Peirano G, DeVinney R, Bradfor PA, Motyl M, Tanaka M, Nagao M, Takakura S, Ichiyama S (2016) Global Escherichia coli sequence type 131 clade with blaCTX-M-27 gene. Emerg Infect Dis 22:1900–1907
Nicolas-Chanoine M-H, Petitjean M, Mora A, Mayer N, Lavigne JP, Boulet O, Leflon-Guibout V, Blanco J, Hocquet D (2017) The ST131 Escherichia coli H22 subclone from human intestinal microbiota: comparison of genomic and phenotypic traits with those of the globally successful H30 subclone. BMC Microbiol 17:71
Liu CM, Stegger M, Aziz M, Johnson TJ, Waits K, Nordstrom L, Gauld L, Weaver B, Rolland D, Statham S, Horwinski J, Sariya S, David GS, Sokurenko E, Keim P, Johnson JR, Price LB (2018) Escherichia coli ST131-H22 as a foodborne uropathogen. mBio 9:e00470–00418
Riley LW (2014) Pandemic lineages of extraintestinal pathogenic Escherichia coli. Clin Microbiol Infect 20:380–390
Ewers C, Bethe A, Stamm I, Grobbel M, Kopp PA, Guerra B, Stubbe M, Doi Y, Zong Z, Kola A, Schaufler K, Semmler T, Fruth A, Wieler LH, Guenther S (2014) CTX-M-15-D-ST648 emopenEscherichia coliemclose from companion animals and horses: another pandemic clone combining multiresistance and extraintestinal virulence? J Antimicrob Chemother 69:1224–1230
Matsumura Y, Yamamoto M, Nagao M, Ito Y, Takakura S, Ichiyama S (2013) Association of fluoroquinolone resistance, virulence genes, and IncF plasmids wtih extended-spectrum-β-lactamase-producing Escherichia coli sequence type 131 (ST131) and ST405 clonal groups. Antimicrob Agents Chemother 57:4736–4742
Johnson JR, Johnston BD, Gordon DM (2017) Rapid and specific detection of the Escherichia coli sequence type 648 complex within phylogroup F. J Clin Microbiol 55:1116–1121
Vila J, Simon K, Ruiz J, Horcajada JP, Velasco M, Barranco M, Moreno A, Mensa J (2002) Are quinolone-resistant uropathogenic Escherichia coli less virulent? J Infect Dis 186:1039–1042
Goodman KE, Simner PJ, Tamma PD, Milstone AM (2016) Infection control implications of heterogeneous resistance mechanisms in carbapenem-resistant Enterobacteriaceae (CRE). Expert Rev Anti Infect Ther 14:95–108
Bailey JK, Pinyon JL, Anantham S, Hall RM (2010) Distribution of human commensal emopenEscherichia coliemclose phylogenetic groups. J Clin Microbiol 48:3455–3456
Skurkink D, Bonnet D, Bernède-Bauduin C, Michel R, Guette C, Becker J-M, Balaire C, Chau F, Mohler J, Jarlier V, Boutin J-P, Moreau B, Guillemot D, Denamur E, Andremont A, Ruimy R (2008) Characteristics of human intestinal emopenEscherichia coliemclose with changing environments. Environ Microbiol 10:2132–2137
Duriez P, Clermont O, Bonacorsi S, Bingen E, Chaventre A, Elion J, Picard B, Denamur E (2001) Commensal Escherichia coli isolates are phylogenetically distributed among geographically distinct human populations. Microbiol 147:1671–1676
Tenaillon O, Skurnik D, Picard B, Denamur E (2010) The population genetics of commensal emopenEscherichia coliemclose. Nat Rev Microbiol 8:207–217
Acknowledgements
The Minnesota Department of Health and JMI Laboratories provided the study isolates.
Funding
This work was supported in part by investigator-initiated grants from Allergan, Cipla/Achaogen, Melinta, Merck, Shionogi, and Tetraphase. It was also supported by Office of Research and Development, Department of Veterans Affairs. The sponsors had no role in study design, data collection, data analysis, writing the manuscript, or the decision to publish.
Author information
Authors and Affiliations
Contributions
Brian Johnston: data collection and validation, laboratory procedures, data analysis, construction of tables, manuscript writing and editing.
Paul Thuras: statistical analysis.
Stephen B. Porter: isolate collection, data collection, manuscript editing.
Melissa Anacker: provision of isolates, data collection, manuscript editing.
Brittany VonBank: provision of isolates, data collection, manuscript editing.
Paula Snippes Vagnone: provision of isolates, data collection, manuscript editing.
Medora Witwer: provision of isolates, data collection, manuscript editing.
Mariana Castanheira: provision of isolates, data collection, manuscript editing.
James R. Johnson: concept, funding, project oversight, manuscript writing and editing.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the MVAMC Institutional Review Board.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
James R. Johnson has had grants and/or consultancies with Allergan/Actavis, Cipla/Achaogen, Janssen/Crucell, Melinta/The Medicines Company, Merck, Shionogi, Syntiron, and Tetraphase.
Mariana Castanheira is an employee of JMI Laboratories. JMI Laboratories contracted to perform services in 2019 for Achaogen, Inc., Albany College of Pharmacy and Health Sciences, Allecra Therapeutics, Allergan, AmpliPhi Biosciences Corp., Amicrobe Advanced Biomaterials, Amplyx, Antabio, American Proficiency Institute, Arietis Corp., Arixa Pharmaceuticals, Inc., Astellas Pharma Inc., Athelas, Basilea Pharmaceutica Ltd., Bayer AG, Becton, Dickinson and Company, bioMerieux SA, Boston Pharmaceuticals, Bugworks Research Inc., CEM-102 Pharmaceuticals, Cepheid, Cidara Therapeutics, Inc., CorMedix Inc., DePuy Synthes, Destiny Pharma, Discuva Ltd., Dr. Falk Pharma GmbH, Emery Pharma, Entasis Therapeutics, Eurofarma Laboratorios SA, U.S. Food and Drug Administration, Fox Chase Chemical Diversity Center, Inc., Gateway Pharmaceutical LLC, GenePOC Inc., Geom Therapeutics, Inc., GlaxoSmithKline plc, Harvard University, Helperby, HiMedia Laboratories, F. Hoffmann-La Roche Ltd., ICON plc, Idorsia Pharmaceuticals Ltd., Iterum Therapeutics plc, Laboratory Specialists, Inc., Melinta Therapeutics, Inc., Merck & Co., Inc., Microchem Laboratory, Micromyx, MicuRx Pharmaceuticals, Inc., Mutabilis Co., Nabriva Therapeutics plc, NAEJA-RGM, Novartis AG, Oxoid Ltd., Paratek Pharmaceuticals, Inc., Pfizer, Inc., Polyphor Ltd., Pharmaceutical Product Development, LLC, Prokaryotics Inc., Qpex Biopharma, Inc., Roivant Sciences, Ltd., Safeguard Biosystems, Scynexis, Inc., SeLux Diagnostics, Inc., Shionogi and Co., Ltd., SinSa Labs, Spero Therapeutics, Summit Pharmaceuticals International Corp., Synlogic, T2 Biosystems, Inc., Taisho Pharmaceutical Co., Ltd., TenNor Therapeutics Ltd., Tetraphase Pharmaceuticals, Theravance Biopharma, University of Colorado, University of Southern California-San Diego, University of North Texas Health Science Center, VenatoRx Pharmaceuticals, Inc., Viosera Therapeutics, Vyome Therapeutics Inc., Wockhardt, Yukon Pharmaceuticals, Inc., Zai Lab, and Zavante Therapeutics, Inc. She has no speakers’ bureaus or stock options to declare.
The other authors report no financial conflicts of interest.
Disclaimer
The opinions expressed here are strictly those of the authors and do not necessarily represent those of the Department of Veteran Affairs or the authors' institutions.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Johnston, B.D., Thuras, P., Porter, S.B. et al. Global molecular epidemiology of carbapenem-resistant Escherichia coli (2002–2017). Eur J Clin Microbiol Infect Dis (2021). https://doi.org/10.1007/s10096-021-04310-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10096-021-04310-6