Abstract
Purpose
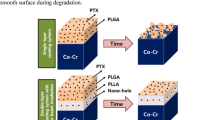
To explore the degradation, drug release, and mechanical properties of drug-incorporated films made of different ratios of poly(lactic-co-glycolic acid) (PLGA) and different amounts of paclitaxel (PTX), which may serve as the material platform for the manufacturing of biodegradable drug-eluting biliary stents.
Materials and Methods
PLGA of different lactic acid/glycolic acid ratios (50/50, 70/30, and 80/20) and 0%, 10, 20, and 30% weight by weight (w/w) PTX was mixed to make PLGA films, which were then cut into small pieces for further testing. Films were immersed in phosphate-buffered saline (pH 7.4) for a maximum of 11 weeks. Samples were taken randomly at Day 2, 4, 6, 8, 10, 12, 14, and weekly thereafter until Week 11 to test tensile strength, weight loss, pH value of the soaking solution, and drug release. The morphology of films was observed using scanning electron microscope (SEM).
Results
At Week 10 of degradation, PLGA 80/20 still withstood a tensile strength of 9.7 newton (N), while PLGA 50/50 and 70/30 cracked spontaneously since Day 4. At Week 11, weight loss of PLGA 50/50, 70/30, and 80/20 was 95.15, 82.32, and 16.17%, respectively; and the lowest pH value of soaking solution was 1.87, 1.95, and 6.58, respectively. Drug release of 10, 20, and 30% PTX groups was 3.52–4.48%, 1.90–2.26%, and 1.44–2.06%, respectively. SEM proved smooth films before degradation; however, after the tensile strength was lost, cracks could be seen.
Conclusion
The degradation rate of PLGA can be controlled by altering lactic acid/glycolic acid ratio. Overall, PLGA 50/50 and 70/30 degrade significantly faster than 80/20. PLGA can serve as an effective drug carrier for PTX while being the stent strut, and PTX can be slowly released as PLGA degrades.






Similar content being viewed by others
References
Judah JR, Draganov PV. Endoscopic therapy of benign biliary strictures. World J Gastroenterol. 2007;13(26):3531–9.
DePietro DM, et al. Long-term outcomes of a benign biliary stricture protocol. J Vasc Interv Radiol. 2015;26(7):1032–9.
Weber A, et al. Long-term follow-up of percutaneous transhepatic biliary drainage (PTBD) in patients with benign bilioenterostomy stricture. Endoscopy. 2009;41(4):323–8.
Kim JH, et al. Bile leak following living donor liver transplantation: clinical efficacy of percutaneous transhepatic treatment. Liver Transpl. 2008;14(8):1142–9.
Krokidis M, et al. Interventional radiology in the management of benign biliary stenoses, biliary leaks and fistulas: a pictorial review. Insights Imaging. 2013;4(1):77–84.
Ogura T, et al. Recanalization for tight bile duct-jejunum anastomosis stricture using peroral transliminal cholangioscopy (with video). Dig Dis. 2018;36(6):446–9.
Donelli G, et al. Plastic biliary stent occlusion: factors involved and possible preventive approaches. Clin Med Res. 2007;5(1):53–60.
Petrtyl J, et al. Management of benign intrahepatic bile duct strictures: initial experience with polydioxanone biodegradable stents. Endoscopy. 2010;42(Suppl 2):E89-90.
van Boeckel PG, Vleggaar FP, Siersema PD. Plastic or metal stents for benign extrahepatic biliary strictures: a systematic review. BMC Gastroenterol. 2009;9:96.
Dumonceau JM, et al. Biliary stenting: indications, choice of stents and results: european society of gastrointestinal endoscopy (ESGE) clinical guideline. Endoscopy. 2012;44(3):277–98.
Siriwardana HP, Siriwardena AK. Systematic appraisal of the role of metallic endobiliary stents in the treatment of benign bile duct stricture. Ann Surg. 2005;242(1):10–9.
Maccioni F, et al. Metallic stents in benign biliary strictures: three-year follow-up. Cardiovasc Intervent Radiol. 1992;15(6):360–6.
Xu X, et al. Feasibility of biodegradable PLGA common bile duct stents: an in vitro and in vivo study. J Mater Sci Mater Med. 2009;20(5):1167–73.
Meng B, et al. Study of biodegradable and self-expandable PLLA helical biliary stent in vivo and in vitro. J Mater Sci Mater Med. 2006;17(7):611–7.
Siiki A, et al. A pilot study of endoscopically inserted biodegradable biliary stents in the treatment of benign biliary strictures and cystic duct leaks. Gastrointest Endosc. 2018;87(4):1132–7.
Mauri G, et al. Benign biliary strictures refractory to standard bilioplasty treated using polydoxanone biodegradable biliary stents: retrospective multicentric data analysis on 107 patients. Eur Radiol. 2016;26(11):4057–63.
Siiki A, Sand J, Laukkarinen J. A systematic review of biodegradable biliary stents: promising biocompatibility without stent removal. Eur J Gastroenterol Hepatol. 2018;30(8):813–8.
Makadia HK, Siegel SJ. Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers (Basel). 2011;3(3):1377–97.
Shi J, et al. Interest of a new biodegradable stent coated with paclitaxel on anastomotic wound healing after biliary reconstruction. Eur J Gastroenterol Hepatol. 2013;25(12):1415–23.
Jang SI, et al. Porcine feasibility and safety study of a new paclitaxel-eluting biliary stent with a Pluronic-containing membrane. Endoscopy. 2012;44(9):825–31.
Almeida GG, Donato P. Biodegradable versus multiple plastic stent implantation in benign biliary strictures: a systematic review and meta-analysis. Eur J Radiol. 2020;125:108899.
Laukkarinen J, et al. A novel biodegradable biliary stent in the endoscopic treatment of cystic-duct leakage after cholecystectomy. Gastrointest Endosc. 2007;65(7):1063–8.
Bowen PK, et al. biodegradable metals for cardiovascular stents: from clinical concerns to recent Zn-alloys. Adv Healthc Mater. 2016;5(10):1121–40.
Hu T, et al. Biodegradable stents for coronary artery disease treatment: recent advances and future perspectives. Mater Sci Eng C Mater Biol Appl. 2018;91:163–78.
Grolich T, et al. Self-expandable biodegradable biliary stents in porcine model. J Surg Res. 2015;193(2):606–12.
Girard E, et al. Evaluation of a biodegradable PLA-PEG-PLA internal biliary stent for liver transplantation: in vitro degradation and mechanical properties. J Biomed Mater Res B Appl Biomater. 2021;109(3):410–9.
Tanimoto Y, et al. Radiopaque biodegradable stent for duct-to-duct biliary reconstruction in pigs. Langenbecks Arch Surg. 2016;401(4):513–7.
Anderloni A, et al. New biliary and pancreatic biodegradable stent placement: a single-center, prospective, pilot study (with video). Gastrointest Endosc. 2020;92(2):405–11.
Katsanos K, et al. Risk of Death Following Application of Paclitaxel-Coated Balloons and Stents in the Femoropopliteal Artery of the Leg: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J Am Heart Assoc. 2018;7(24):e011245.
Lee SS, et al. Histologic influence of paclitaxel-eluting covered metallic stents in a canine biliary model. Gastrointest Endosc. 2009;69(6):1140–7.
Xu Y, et al. Autologous urothelial cells transplantation onto a prefabricated capsular stent for tissue engineered ureteral reconstruction. J Mater Sci Mater Med. 2012;23(4):1119–28.
Acknowledgements
The authors thank Dr. Li-Gong Lu (Zhuhai People's Hospital, Zhuhai Hospital of Jinan University), Dr. Lei Chen (The Affiliated Suzhou Hospital of Nanjing Medical University), Dr. Jia-Jie Ji (Zhongda Hospital, Southeast University), Mr. Xiang-Man Zhang [Heng Rui Callisyn Biomedical (Suzhou) Co., Ltd.], and Mr. Xiao-Lei Gu [Heng Rui Callisyn Biomedical (Suzhou) Co., Ltd.] for their assistance in carrying out the study.
Funding
This study was supported by the National Natural Science Foundation of China (81441054, 81671796) and the Jiangsu Provincial Medical Youth Talent Program (ZDRCA2016078). The funders had no role in study design, data analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zeng, CH., Liu, LL., Zhu, HD. et al. The Exploration of a Novel Biodegradable Drug-Eluting Biliary Stent: Preliminary Work. Cardiovasc Intervent Radiol 44, 1633–1642 (2021). https://doi.org/10.1007/s00270-021-02892-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-021-02892-4