Abstract
Background
There is evidence that post-training exposure to nicotine, cocaine, and their conditioned stimuli (CS), enhance memory consolidation in rats. The present study assessed the effects of blocking noradrenergic and dopaminergic receptors on nicotine and cocaine unconditioned and conditioned memory modulation.
Methods
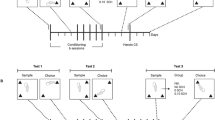
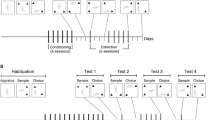
Males Sprague–Dawley rats tested on the spontaneous object recognition task received post-sample exposure to 0.4 mg/kg nicotine, 20 mg/kg cocaine, or their CSs, in combination with 5–10 mg/kg propranolol (PRO; beta-adrenergic antagonist) or 0.2–0.6 mg/kg pimozide (PIM; dopamine D2 receptor antagonist). The CSs were established by confining rats in a chamber (the CS +) after injections of 0.4 mg/kg nicotine, or 20 mg/kg cocaine, for 2 h and in another chamber (the CS −) after injections of vehicle, repeated over 10 days (5 drug/CS + and 5 vehicle/CS − pairings in total). Object memory was tested 72 h post sample in drug-free animals.
Results
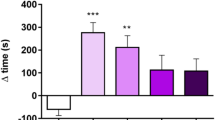
Co-administration of PRO or PIM blocked the memory-enhancing effects of post-training injections of nicotine, cocaine, and, importantly, exposure to their CSs.
Conclusions
These data suggest that nicotine, cocaine as well as their conditioned stimuli share actions on overlapping noradrenergic and dopaminergic systems to modulate memory consolidation.




Similar content being viewed by others
References
Albasser MM, Olarte-Sánchez CM, Amin E et al (2015) Perirhinal cortex lesions in rats: novelty detection and sensitivity to interference. Behav Neurosci 129(3):227–243. https://doi.org/10.1037/bne0000049
Arqueros L, Naquira D, Zunino E (1978) Nicotine-induced release of catecholamines from rat hippocampus and striatum. Biochem Pharmacol 27(23):2667–2674. https://doi.org/10.1016/0006-2952(78)90040-0
Atkins A, Mashoon Y, Kantak KM (2012) Hippocampal regulation of contextual cue-induced reinstatement of cocaine-seeking behavior. Bone 23(1):1–7. https://doi.org/10.1038/jid.2014.371
Baidoo N, Wolter M, Holahan MR, et al. (2020) The effects of morphine withdrawal and conditioned withdrawal on memory consolidation and c-Fos expression in the central amygdala. Addiction Biology (January 2020): 1–11. DOI: https://doi.org/10.1111/adb.12909
Balderas I, Moreno-Castilla P, Bermudez-Rattoni F (2013) Dopamine D1 receptor activity modulates object recognition memory consolidation in the perirhinal cortex but not in the hippocampus. Hippocampus 23(10):873–878. https://doi.org/10.1002/hipo.22143
Barros DM, Ramirez MR, Izquierdo I (2005) Modulation of working, short- and long-term memory by nicotinic receptors in the basolateral amygdala in rats. Neurobiol Learn Mem 83(2):113–118. https://doi.org/10.1016/j.nlm.2004.10.001
Beninger RJ, Phillips AG (1980) The effect of pimozide on the establishment of conditioned reinforcement. Psychopharmacology 153:147–153
Blackburn JR, Phillips AG, Fibiger HC (1987) Dopamine and preparatory behavior: I. Effects of Pimozide 101(3):352–360
Bocklisch C, Pascoli V, Wong JCY et al (2013) Cocaine disinhibits dopamine neurons by potentiation of GABA transmission in the ventral tegmental area christina. Science 341:1521–1526. https://doi.org/10.1126/science.1142365
Brazell MP, Mitchell SN, Gray JA (1991) Effect of acute administration of nicotine on in vivo release of noradrenaline in the hippocampus of freely moving rats: a dose-response and antagonist study. Neuropharmacology 30(8):823–833. https://doi.org/10.1016/0028-3908(91)90116-S
Brophey A, Raptis H (2003) Cholinergic neurotransmission is essential for perirhinal cortical plasticity and recognition memory. Neuron 38:237–252
Brown EE, George S, Robertson S et al (1992) Evidence for conditional neuronal activation following exposure to a cocaine-paired environment: role of forebrain limbic structures. J Neurosci 12(10):4112–4121
Cahill L, Prins B, Weber M et al (1994) β-Adrenergic activation and memory for emotional events. Nature. https://doi.org/10.1038/371702a0
Cahill L, Pham CA, Setlow B (2000) Impaired memory consolidation in rats produced with β-adrenergic blockade. Neurobiol Learn Mem 74(3):259–266. https://doi.org/10.1006/nlme.1999.3950
Castellano C, Cestari V, Cabib S et al (1991) Post-training dopamine receptor agonists and antagonists affect memory storage in mice irrespective of their selectivity for D1 or D2 receptors. Behav Neural Biol 56(3):283–291. https://doi.org/10.1016/0163-1047(91)90439-W
Castellano C, Cestari V, Cabib S et al (1994) The effects of morphine on memory consolidation in mice involve both D1 and D2 dopamine receptors. Behav Neural Biol 61(2):156–161. https://doi.org/10.1016/S0163-1047(05)80069-X
Cestari V, Mele A, Oliverio A et al (1996) Amygdala lesions block the effect of cocaine on memory in mice. Brain Res 713(1–2):286–289
Dani JA, Bertrand D (2007) Nicotinic acetylcholine receptors and nicotinic cholinergic mechanisms of the central nervous system. Annu Rev Pharmacol Toxicol 47(1):699–729. https://doi.org/10.1146/annurev.pharmtox.47.120505.105214
de Lima MNM, Presti-Torres J, Dornelles A et al (2011) Modulatory influence of dopamine receptors on consolidation of object recognition memory. Neurobiol Learn Mem 95(3):305–310. https://doi.org/10.1016/j.nlm.2010.12.007
Deroche-Gamonet V, Martinez A, Le Moal M et al (2003) Relationships between individual sensitivity to CS- and cocaine-induced reinstatement in the rat. Psychopharmacology 168(1–2):201–207. https://doi.org/10.1007/s00213-002-1306-9
Di Ciano P, Everitt BJ (2004) Direct interactions between the basolateral amygdala and nucleus accumbens core underlie cocaine-seeking behavior by rats. J Neurosci 24(32):7167–7173. https://doi.org/10.1523/JNEUROSCI.1581-04.2004
Duszkiewicz AJ, Mcnamara CG, Takeuchi T, et al. (2019) Novelty and dopaminergic modulation of memory persistence: a tale of two systems. Trends Neurosci 42(2). Elsevier Ltd: 102–114. https://doi.org/10.1016/j.tins.2018.10.002
Ennaceur A, Delacour J (1988) A new one-trial test for neurobiological studies of memory in rats. 1: Behavioral data. Behav Brain Res 31:47–59. https://doi.org/10.1016/S0166-4328(05)80315-8
Ferry B, Roozendaal B, McGaugh JL (1999) Basolateral amygdala noradrenergic influences on memory storage are mediated by an interaction between β- and α1-adrenoceptors. J Neurosci. https://doi.org/10.1523/jneurosci.19-12-05119.1999
Fitzgerald PJ (2013) Elevated norepinephrine may be a unifying etiological factor in the abuse of a broad range of substances: alcohol, nicotine, marijuana, heroin, cocaine, and caffeine. Subst Abuse 7:171–183. https://doi.org/10.4137/SART.S13019
Florin SM, Kuczenski R, Segal DS (1994) Regional extracellular norepinephrine responses to amphetamine and cocaine and effects of clonidine pretreatment. Brain Res 654(1):53–62. https://doi.org/10.1016/0006-8993(94)91570-9
Fotros A, Casey KF, Larcher K, et al. (2013) Cocaine cue-induced dopamine release in amygdala and hippocampus: a high-resolution PET 18Fallypride study in cocaine dependent participants. Neuropsychopharmacology 38(9). Nature Publishing Group: 1780–1788. https://doi.org/10.1038/npp.2013.77
Fu Y, Matta SG, Kane VB et al (2003) Norepinephrine release in amygdala of rats during chronic nicotine self-administration: an in vivo microdialysis study. Neuropharmacology 45(4):514–523. https://doi.org/10.1016/S0028-3908(03)00201-6
Fuchs RA, Evans KA, Ledford CC et al (2005) The role of the dorsomedial prefrontal cortex, basolateral amygdala, and dorsal hippocampus in contextual reinstatement of cocaine seeking in rats. Neuropsychopharmacology 30(2):296–309. https://doi.org/10.1038/sj.npp.1300579
Gibbs ME, Hutchinson DS and Summers RJ (2010) Noradrenaline release in the locus coeruleus modulates memory formation and consolidation; roles for α- and β-adrenergic receptors. Neuroscience 170(4). Elsevier Inc.: 1209–1222. https://doi.org/10.2147/CIA.S118152
Goode TD, Leong KC, Goodman J, et al. (2016) Enhancement of striatum-dependent memory by conditioned fear is mediated by beta-adrenergic receptors in the basolateral amygdala. Neurobiology of Stress 3. Elsevier Inc: 74–82. https://doi.org/10.1016/j.ynstr.2016.02.004
Hadjiconstantinou M and Neff NH (2011) Nicotine and endogenous opioids: neurochemical and pharmacological evidence. Neuropharmacology 60(7–8). Elsevier Ltd: 1209–1220. https://doi.org/10.1016/j.neuropharm.2010.11.010.
Hansen N (2017) The longevity of hippocampus-dependent memory is orchestrated by the locus coeruleus–noradrenergic system. Neural Plasticity 2017. https://doi.org/10.1155/2017/2727602
Heath FC, Jurkus R, Bast T et al (2015) Dopamine D1-like receptor signalling in the hippocampus and amygdala modulates the acquisition of contextual fear conditioning. Psychopharmacology 232(14):2619–2629. https://doi.org/10.1007/s00213-015-3897-y
Holahan MR and White NM (2002) Conditioned memory modulation, freezing, and avoidance as measures of amygdala-mediated conditioned fear. 275: 250–275. https://doi.org/10.1006/nlme.2001.4012
Holahan MR, White NM (2004) Amygdala inactivation blocks expression of conditioned memory modulation and the promotion of avoidance and freezing. Behav Neurosci 118(1):24–35. https://doi.org/10.1037/0735-7044.118.1.24
Holmes NM, Parkes SL, Killcross AS et al (2013) The basolateral amygdala is critical for learning about neutral stimuli in the presence of danger, and the perirhinal cortex is critical in the absence of danger. J Neurosci 33(32):13112–13125. https://doi.org/10.1523/JNEUROSCI.1998-13.2013
Horvitz JC, Ettenberg A (1991) Conditioned incentive properties of a food-paired conditioned stimulus remain intact during dopamine receptor blockade. Behav Neurosci 105(4):536–541. https://doi.org/10.1037/0735-7044.105.4.536
Hsu EH, Schroeder JP, Packard MG (2002) The amygdala mediates memory consolidation for an amphetamine conditioned place preference. Behav Brain Res 129(1–2):93–100. https://doi.org/10.1016/S0166-4328(01)00376-X
Ichihara K, Nabeshima T, Kameyama T (1989) Differential effects of pimozide and SCH 23390 on acquisition of learning in mice. Eur J Pharmacol 164:189–195
Introini-Collison IB, Baratti CM (1986) Opioid peptidergic systems modulate the activity of β-adrenergic mechanisms during memory consolidation processes. Behav Neural Biol 46(2):227–241. https://doi.org/10.1016/S0163-1047(86)90710-7
Keshavarzian E, Ghasemzadeh Z and Rezayof A (2018) The basolateral amygdala dopaminergic system contributes to the improving effect of nicotine on stress-induced memory impairment in rats. Progress in Neuro-Psychopharmacology and Biological Psychiatry 86(May). Elsevier: 30–35. https://doi.org/10.1016/j.pnpbp.2018.05.008
Khaled MATM, Pushparaj A, Di Ciano P, et al. (2014) Dopamine D3 receptors in the basolateral amygdala and the lateral habenula modulate cue-induced reinstatement of nicotine seeking. Neuropsychopharmacology 39(13). Nature Publishing Group: 3049–3058. https://doi.org/10.1038/npp.2014.158
Kim KM, Baratta MV, Yang A, et al. (2012) Optogenetic mimicry of the transient activation of dopamine neurons by natural reward is sufficient for operant reinforcement. PLoS ONE 7(4). https://doi.org/10.1371/journal.pone.0033612
Kobayashi K, Yasoshima Y (2001) The central noradrenaline system and memory consolidation. Neuroscientist 7(5):371–376. https://doi.org/10.1177/107385840100700506
Koch G, Esposito Z, Codecà C, et al. (2011) Altered dopamine modulation of LTD-like plasticity in Alzheimer’s disease patients. Clinical Neurophysiology 122(4). International Federation of Clinical Neurophysiology: 703–707. https://doi.org/10.1016/j.clinph.2010.10.033.
Kramar CP, Chefer VI, Wise RA, et al. (2014) Dopamine in the dorsal hippocampus impairs the late consolidation of cocaine-associated memory. Neuropsychopharmacology 39(7). Nature Publishing Group: 1645–1653. https://doi.org/10.1038/npp.2014.11
Kutlu MG, Gould TJ (2016) Effects of drugs of abuse on hippocampal plasticity and hippocampus-dependent learning and memory: contributions to development and maintenance of addiction. Learn Mem 23(10):515–533. https://doi.org/10.1101/lm.042192.116
Laing M and Bashir ZI (2014) β-Adrenoceptors and synaptic plasticity in the perirhinal cortex. Neuroscience 273. IBRO: 163–173. https://doi.org/10.1016/j.neuroscience.2014.04.070
Lee EH, Ma YL (1995) Amphetamine enhances memory retention and facilitates norepinephrine release from the hippocampus in rats. Brain Res Bull 37(4):411–416
Liang KC, Mcgaugh JL and Yao H-Y (1990) Involvement of amygdala pathways in the influence of post-training intra-amygdala norepinephrine and peripheral epinephrine on memory storage. Brain Research. Available at: https://ac-els-cdn-com.subzero.lib.uoguelph.ca/0006899390904006/1-s2.0-0006899390904006-main.pdf?_tid=8aebb597-37a1-4b4c-8a80-ae7334baa68e&acdnat=1547578826_9e20f835a592a55a4eeb9e7af566a132 (accessed 15 January 2019)
Lisman JE, Grace AA (2005) The hippocampal-VTA loop: controlling the entry of information into long-term memory. Neuron 46(5):703–713. https://doi.org/10.1016/j.neuron.2005.05.002
Lodge DJ, Grace AA (2008) Amphetamine activation of hippocampal drive of mesolimbic dopamine neurons: a mechanism of behavioral sensitization. J Neurosci 28(31):7876–7882. https://doi.org/10.1523/JNEUROSCI.1582-08.2008
McGaugh JL (2000) Memory—a century of consolidation. Science 287(5451):248–251. https://doi.org/10.1126/science.287.5451.248
McGaugh JL (2004) The amygdala modulates the consolidation of memories of emotionally arousing experiences. Annu Rev Neurosci 27:1–28. https://doi.org/10.1146/annurev.neuro.27.070203.144157
McGaugh JL (2013) Making lasting memories: remembering the significant. Proc Natl Acad Sci USA 110(SUPPL2):10402–10407. https://doi.org/10.1073/pnas.1301209110
McGaugh JL, Roozendaal B (2002) Role of adrenal stress hormones in forming lasting memories in the brain. Curr Opin Neurobiol 12(2):205–210
Melichercik AM, Elliott KS, Bianchi C, et al. (2012) Nicotinic receptor activation in perirhinal cortex and hippocampus enhances object memory in rats. Neuropharmacology 62(5–6). Elsevier Ltd: 2096–2105. https://doi.org/10.1016/j.neuropharm.2012.01.008.
Mishra A, Singh S and Shukla S (2018) Physiological and functional basis of dopamine receptors and their role in neurogenesis: possible implication for Parkinson’s disease. J Exp Neurosci 12. https://doi.org/10.1177/1179069518779829
Mitchell SN, Brazell MP, Joseph MH et al (1989) Regionally specific effects of acute and chronic nicotine on rates of catecholamine and 5-hydroxytryptamine synthesis in rat brain. Eur J Pharmacol 167(3):311–322
Placzek AN, Zhang TA, Dani JA (2009) Nicotinic mechanisms influencing synaptic plasticity in the hippocampus. Acta Pharmacol Sin 30(6):752–760. https://doi.org/10.1038/aps.2009.39
Rescorla RA, Solomon RL (1975) Two process learning theory: relationships between Pavlovian conditioning and instrumental learning. Psychol Rev 74(3):151–182
Rkieh N, Cloke JM, Gallagher N et al (2014) Drugs of abuse as memory modulators: a study of cocaine in rats. Psychopharmacology 231(11):2339–2348. https://doi.org/10.1007/s00213-013-3390-4
Roozendaal B, Nguyen BT, Power AE et al (1999) Basolateral amygdala noradrenergic influence enables enhancement of memory consolidation induced by hippocampal glucocorticoid receptor activation. Proc Natl Acad Sci 96(20):11642–11647. https://doi.org/10.1073/pnas.96.20.11642
Roozendaal B, Carmi O and McGaugh JL (2002) Adrenocortical suppression blocks the memory-enhancing effects of amphetamine and epinephrine. Proceedings of the National Academy of Sciences 93(4). National Academy of Sciences: 1429–1433. https://doi.org/10.1073/pnas.93.4.1429.
Roozendaal B, Castello NA, Vedana G, et al. (2008) Noradrenergic activation of the basolateral amygdala modulates consolidation of object recognition memory. Neurobiology of Learning and Memory 90(3). NIH Public Access: 576–579. https://doi.org/10.1016/j.nlm.2008.06.010.
Rossato JI, Radiske A, Kohler CA, et al. (2013) Consolidation of object recognition memory requires simultaneous activation of dopamine D1/D5 receptors in the amygdala and medial prefrontal cortex but not in the hippocampus. Neurobiology of Learning and Memory 106. Elsevier Inc.: 66–70. https://doi.org/10.1016/j.nlm.2013.07.012
Rossi S, Singer S, Shearman E et al (2005) The effects of cholinergic and dopaminergic antagonists on nicotine-induced cerebral neurotransmitter changes. Neurochem Res 30(4):541–558. https://doi.org/10.1007/s11064-005-2689-x
Sharp BM (2019) Basolateral amygdala, nicotinic cholinergic receptors, and nicotine: pharmacological effects and addiction in animal models and humans. Eur J Neurosci 50(3):2247–2254. https://doi.org/10.1111/ejn.13970
Stern SA, Alberini CM (2013) Mechanisms of memory enhancement. Wiley Interdiscip Rev Syst Biol Med 5(1):37–53. https://doi.org/10.1002/wsbm.1196
Stewart J, de Wit H, Eikelboom R (1984) Role of unconditioned and conditioned drug effects in the self-administration of opiates and stimulants. Psychol Rev 91(2):251–268. https://doi.org/10.1037/0033-295X.91.2.251
Tang W, Kochubey O, Kintscher M et al (2020) A VTA to basal amygdala dopamine projection contributes to signal salient somatosensory events during fear learning. J Neurosci 40(20):3969–3980. https://doi.org/10.1523/JNEUROSCI.1796-19.2020
Tessari M, Catalano A, Pellitteri M et al (2007) Correlation between serum ghrelin levels and cocaine-seeking behaviour triggered by cocaine-associated conditioned stimuli in rats. Addict Biol 12(1):22–29. https://doi.org/10.1111/j.1369-1600.2007.00052.x
Tunstall BJ, Kearns DN (2017) Cocaine can generate a stronger conditioned reinforcer than food despite being a weaker primary reinforcer. Addict Biol 21(2):282–293. https://doi.org/10.1111/adb.12195.Cocaine
Tzschentke TM (1998) Measuring reward with the conditioned place preference paradigm: a comprehensive review of drug effects, recent progress and new issues. Prog Neurobiol 56(6):613–672. https://doi.org/10.1016/S0301-0082(98)00060-4
Verheij MMM, Karel P, Cools AR, et al. (2014) Reduced cocaine-induced serotonin, but not dopamine and noradrenaline, release in rats with a genetic deletion of serotonin transporters. European Neuropsychopharmacology 24(11). Elsevier: 1850–1854. https://doi.org/10.1016/j.euroneuro.2014.09.004.
Villain H, Benkahoul A, Drougard A et al (2016) Effects of propranolol, a β-noradrenergic antagonist, on memory consolidation and reconsolidation in mice. Front Behav Neurosci 10(March):1–14. https://doi.org/10.3389/fnbeh.2016.00049
White N (1996) Addictive drugs as reinforcers: multiple partial actions on memory systems. Addiction 91(7):921–949. https://doi.org/10.1111/j.1360-0443.1996.tb03586.x
White N (2002) The psychobiology of reinforcers. Annu Rev Psychol. https://doi.org/10.1146/annurev.psych.43.1.443
White N, Major R (1978) Effect of pimozide on the improvement in learning produced by self-stimulation and by water reinforcement I. Pharmacol Biochem Behav 8:565–571
Winters BD, Bussey TJ (2005) Glutamate receptors in perirhinal cortex mediate encoding, retrieval, and consolidation of object recognition memory. J Neurosci 25(17):4243–4251. https://doi.org/10.1523/JNEUROSCI.0480-05.2005
Winters BD, Forwood SE, Cowell RA et al (2004) Double dissociation between the effects of peri-postrhinal cortex and hippocampal lesions on tests of object recognition and spatial memory: heterogeneity of function within the temporal lobe. J Neurosci 24(26):5901–5908. https://doi.org/10.1523/JNEUROSCI.1346-04.2004
Winters BD, Saksida LM, Bussey TJ (2008) Object recognition memory: neurobiological mechanisms of encoding, consolidation and retrieval. Neurosci Biobehav Rev 32(5):1055–1070. https://doi.org/10.1016/j.neubiorev.2008.04.004
Wolter M, Huff E, Speigel T et al (2019) Cocaine, nicotine, and their conditioned contexts enhance consolidation of object memory in rats. Learn Mem 26:46–55. https://doi.org/10.1101/lm.048579.118.Freely
Wolter M, Huff AE, Baidoo N, et al. (2020) Modulation of object memory consolidation by heroin and heroin-conditioned stimuli: role of opioid and noradrenergic systems. European Neuropsychopharmacology 33. Elsevier B.V.: 146–157. https://doi.org/10.1016/j.euroneuro.2020.01.010
Yamasaki M and Takeuchi T (2017) Locus coeruleus and dopamine-dependent memory consolidation. Neural Plasticity 2017. Hindawi. https://doi.org/10.1155/2017/8602690
Zavala AR, Browning JR, Dickey ED et al (2008) Region-specific involvement of AMPA/Kainate receptors in Fos protein expression induced by cocaine-conditioned cues. Eur Neuropsychopharmacol 18(8):600–611. https://doi.org/10.1016/j.euroneuro.2008.04.010
Acknowledgements
This research was funded by a Discovery Grant from the Natural Sciences and Engineering Research Council of Canada (NSERC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wolter, M., Lapointe, T., Melanson, B. et al. Memory enhancing effects of nicotine, cocaine, and their conditioned stimuli; effects of beta-adrenergic and dopamine D2 receptor antagonists. Psychopharmacology 238, 2617–2628 (2021). https://doi.org/10.1007/s00213-021-05884-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-021-05884-x