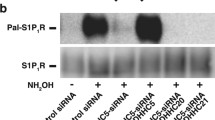
Abstract
Lysophosphatidic acid receptor 1 (LPA1) belongs to the G protein-coupled receptor family. The ligand for LPA1 is LPA, the simplest lysophospholipid. LPA is considered a growth factor and induces cell proliferation, anti-apoptosis, and cell migration. The pro-inflammatory and pro-fibrotic roles of LPA have also been well-demonstrated. Most of the biological functions of LPA are mostly executed through LPA1. The mature form of LPA1 is glycosylated and localized on the plasma membrane. LPA1 is bound to heterotrimetric G proteins and transduces intracellular signaling in response to ligation to LPA. Desensitization of LPA1 negatively regulates LPA1-mediated signaling and the resulting biological functions. Phosphorylation and ubiquitination are well-demonstrated posttranslational modifications of GPCR. In this review, we will discuss our knowledge of LPA1 glycosylation, maturation, and trafficking from the endoplasmic reticulum (ER)/Golgi to the plasma membrane. Moreover, in light of recent findings, we will also discuss molecular regulation of LPA1 internalization and stability.

Similar content being viewed by others
Abbreviations
- LPA:
-
lysophosphatidic acid
- GPCR:
-
G protein-coupled receptor
- ER:
-
endoplasmic reticulum
- ERK:
-
extracellular signal-regulated kinase
- PI3K:
-
phosphatidylinositol-3-kinase
- COPII:
-
coat protein complex II
- SNPs:
-
single nucleotide polymorphisms
- GRK2:
-
GPCR kinase 2
- EGFR:
-
epidermal growth factor receptor
- TrkA:
-
tropomyosin receptor kinase A
- ERAD:
-
ER-associated degradation
- PKC:
-
protein kinase C
- FFA4:
-
free fatty acid receptor 4
- PMA:
-
phorbol ester
- LPS:
-
lipopolysaccharide
- PROTACs:
-
proteolysis-targeting chimeras.
References
Selbie, L. A., & Hill, S. J. (1998). G protein-coupled-receptor cross-talk: the fine-tuning of multiple receptor-signalling pathways. Trends in Pharmacological Sciences, 19, 87–93.
Stevens, R. C., Cherezov, V., Katritch, V., Abagyan, R., Kuhn, P., Rosen, H., & Wuthrich, K. (2013). The GPCR Network: a large-scale collaboration to determine human GPCR structure and function. Nature Reviews Drug Discovery, 12, 25–34.
Pavlos, N. J., & Friedman, P. A. (2017). GPCR Signaling and Trafficking: the Long and Short of It. Trends in Endocrinology & Metabolism, 28, 213–226.
Zhang, D., Zhao, Q., & Wu, B. (2015). Structural Studies of G Protein-Coupled Receptors. Molecular Cells, 38, 836–842.
Becker, O. M., Shacham, S., Marantz, Y., & Noiman, S. (2003). Modeling the 3D structure of GPCRs: advances and application to drug discovery. Current Opinion in Drug Discovery & Development, 6, 353–361.
Fang, Y., Lahiri, J., & Picard, L. (2003). G protein-coupled receptor microarrays for drug discovery. Drug Discovery Today, 8, 755–761.
Congreve, M., Dias, J. M., & Marshall, F. H. (2014). Structure-based drug design for G protein-coupled receptors. Progress in Medicinal Chemistry, 53, 1–63.
Congreve, M., de Graaf, C., Swain, N. A., & Tate, C. G. (2020). Impact of GPCR Structures on Drug Discovery. Cell, 181, 81–91.
Barak, L. S., Wilbanks, A. M., & Caron, M. G. (2003). Constitutive desensitization: a new paradigm for g protein-coupled receptor regulation. Assay and Drug Development Technologies, 1, 339–346.
Rajagopal, S., & Shenoy, S. K. (2018). GPCR desensitization: acute and prolonged phases. Cell Signaling, 41, 9–16.
Evron, T., Daigle, T. L., & Caron, M. G. (2012). GRK2: multiple roles beyond G protein-coupled receptor desensitization. Trends in Pharmacological Sciences, 33, 154–164.
Hla, T., Jackson, A. Q., Appleby, S. B., & Maciag, T. (1995). Characterization of edg-2, a human homologue of the Xenopus maternal transcript G10 from endothelial cells. Biochimica et Biophysica Acta, 1260, 227–229.
An, S., Dickens, M. A., Bleu, T., Hallmark, O. G., & Goetzl, E. J. (1997). Molecular cloning of the human Edg2 protein and its identification as a functional cellular receptor for lysophosphatidic acid. Biochemical and Biophysical Research Communications, 231, 619–622.
Fukushima, N., Kimura, Y., & Chun, J. (1998). A single receptor encoded by vzg-1/lpA1/edg-2 couples to G proteins and mediates multiple cellular responses to lysophosphatidic acid. Proceedings of the National Academy of Sciences of the U S A, 95, 6151–6156.
Moolenaar, W. H. (1994). LPA: a novel lipid mediator with diverse biological actions. Trends in Cell Biology, 4, 213–219.
Sengupta, S., Wang, Z., Tipps, R., & Xu, Y. (2004). Biology of LPA in health and disease. Seminars in Cell and Development Biology, 15, 503–512.
Pages, C., Simon, M. F., Valet, P., & Saulnier-Blache, J. S. (2001). Lysophosphatidic acid synthesis and release. Prostaglandins & Other Lipid Mediators, 64, 1–10.
Xie, Y., & Meier, K. E. (2004). Lysophospholipase D and its role in LPA production. Cell Signaling, 16, 975–981.
Zhao, Y., & Natarajan, V. (2009). Lysophosphatidic acid signaling in airway epithelium: role in airway inflammation and remodeling. Cell Signaling, 21, 367–377.
Zhao, Y., & Natarajan, V. (2013). Lysophosphatidic acid (LPA) and its receptors: role in airway inflammation and remodeling. Biochimica et Biophysica Acta, 1831, 86–92.
Daaka, Y. (2002). Mitogenic action of LPA in prostate. Biochimica et Biophysica Acta, 1582, 265–269.
Fang, X., Schummer, M., Mao, M., Yu, S., Tabassam, F. H., Swaby, R., Hasegawa, Y., Tanyi, J. L., LaPushin, R., Eder, A., Jaffe, R., Erickson, J., & Mills, G. B. (2002). Lysophosphatidic acid is a bioactive mediator in ovarian cancer. Biochimica et Biophysica Acta, 1582, 257–264.
Valdes-Rives, S. A., & Gonzalez-Arenas, A. (2017). Autotaxin-Lysophosphatidic Acid: from Inflammation to Cancer Development. Mediators of Inflammation, 2017, 9173090.
Murph, M. M., Nguyen, G. H., Radhakrishna, H., & Mills, G. B. (2008). Sharpening the edges of understanding the structure/function of the LPA1 receptor: expression in cancer and mechanisms of regulation. Biochimica et Biophysica Acta, 1781, 547–557.
Yung, Y. C., Stoddard, N. C., & Chun, J. (2014). LPA receptor signaling: pharmacology, physiology, and pathophysiology. Journal of Lipid Research, 55, 1192–1214.
Kim, M. K., Lee, H. Y., Park, K. S., Shin, E. H., Jo, S. H., Yun, J., Lee, S. W., Yoo, Y. H., Lee, Y. S., Baek, S. H., & Bae, Y. S. (2005). Lysophosphatidic acid stimulates cell proliferation in rat chondrocytes. Biochemical Pharmacology, 70, 1764–1771.
Billon-Denis, E., Tanfin, Z., & Robin, P. (2008). Role of lysophosphatidic acid in the regulation of uterine leiomyoma cell proliferation by phospholipase D and autotaxin. Journal of Lipid Research, 49, 295–307.
Du, J., Sun, C., Hu, Z., Yang, Y., Zhu, Y., Zheng, D., Gu, L., & Lu, X. (2010). Lysophosphatidic acid induces MDA-MB-231 breast cancer cells migration through activation of PI3K/PAK1/ERK signaling. PLoS ONE, 5, e15940.
Liu, S., Jiang, H., Min, L., Ning, T., Xu, J., Wang, T., Wang, X., Zhang, Q., Cao, R., Zhang, S. & Zhu, S. (2020). Lysophosphatidic acid mediated PI3K/AKT activation contributed to esophageal squamous cell cancer progression. Carcinogenesis, 42, 611–620.
Liu, Y., Chen, F., Ji, L., Zhang, L., Xu, Y. J., & Dhalla, N. S. (2020). Role of lysophosphatidic acid in vascular smooth muscle cell proliferation. Canadian Journal of Physiology and Pharmacology, 98, 103–110.
Van Leeuwen, F. N., Olivo, C., Grivell, S., Giepmans, B. N., Collard, J. G., & Moolenaar, W. H. (2003). Rac activation by lysophosphatidic acid LPA1 receptors through the guanine nucleotide exchange factor Tiam1. Journal of Biological Chemistry, 278, 400–406.
Wei, J., Mialki, R. K., Dong, S., Khoo, A., Mallampalli, R. K., Zhao, Y., & Zhao, J. (2013). A new mechanism of RhoA ubiquitination and degradation: roles of SCF(FBXL19) E3 ligase and Erk2. Biochimica et Biophysica Acta, 1833, 2757–2764.
Zhao, J., Mialki, R. K., Wei, J., Coon, T. A., Zou, C., Chen, B. B., Mallampalli, R. K., & Zhao, Y. (2013). SCF E3 ligase F-box protein complex SCF(FBXL19) regulates cell migration by mediating Rac1 ubiquitination and degradation. FASEB Journal, 27, 2611–2619.
Kim, D., Li, H. Y., Lee, J. H., Oh, Y. S., & Jun, H. S. (2019). Lysophosphatidic acid increases mesangial cell proliferation in models of diabetic nephropathy via Rac1/MAPK/KLF5 signaling. Experimental and Molecular Medicine, 51, 1–10.
Pilquil, C., Dewald, J., Cherney, A., Gorshkova, I., Tigyi, G., English, D., Natarajan, V., & Brindley, D. N. (2006). Lipid phosphate phosphatase-1 regulates lysophosphatidate-induced fibroblast migration by controlling phospholipase D2-dependent phosphatidate generation. Journal of Biological Chemistry, 281, 38418–38429.
Schmitz, U., Thommes, K., Beier, I., & Vetter, H. (2002). Lysophosphatidic acid stimulates p21-activated kinase in vascular smooth muscle cells. Biochemical and Biophysical Research Communications, 291, 687–691.
van Nieuw Amerongen, G. P., Vermeer, M. A., & van Hinsbergh, V. W. (2000). Role of RhoA and Rho kinase in lysophosphatidic acid-induced endothelial barrier dysfunction. Arteriosclerosis Thrombosis and Vascular Biology, 20, E127–133.
Wang, J., Sun, Y., Qu, J., Yan, Y., Yang, Y., & Cai, H. (2016). Roles of LPA receptor signaling in breast cancer. Expert Review of Molecular Diagnostics, 16, 1103–1111.
Park, J., Jang, J. H., Oh, S., Kim, M., Shin, C., Jeong, M., Heo, K., Park, J. B., Kim, S. R., & Oh, Y. S. (2018). LPA-induced migration of ovarian cancer cells requires activation of ERM proteins via LPA1 and LPA2. Cell Signaling, 44, 138–147.
Plastira, I., Bernhart, E., Goeritzer, M., Reicher, H., Kumble, V. B., Kogelnik, N., Wintersperger, A., Hammer, A., Schlager, S., Jandl, K., Heinemann, A., Kratky, D., Malle, E., & Sattler, W. (2016). 1-Oleyl-lysophosphatidic acid (LPA) promotes polarization of BV-2 and primary murine microglia towards an M1-like phenotype. Journal of Neuroinflammation, 13, 205.
Kim, J., Hwang, Y. S., Chung, A. M., Chung, B. G., & Khademhosseini, A. (2012). Liver cell line derived conditioned medium enhances myofibril organization of primary rat cardiomyocytes. Molecular Cells, 34, 149–158.
Zhao, J., Wei, J., Weathington, N., Jacko, A. M., Huang, H., Tsung, A., & Zhao, Y. (2015). Lysophosphatidic acid receptor 1 antagonist ki16425 blunts abdominal and systemic inflammation in a mouse model of peritoneal sepsis. Translation Research, 166, 80–88.
Kwon, J. H., Gaire, B. P., Park, S. J., Shin, D. Y., & Choi, J. W. (2018). Identifying lysophosphatidic acid receptor subtype 1 (LPA1) as a novel factor to modulate microglial activation and their TNF-alpha production by activating ERK1/2. Biochimica et Biophysica Acta - Molecular and Cell Biology of Lipids, 1863, 1237–1245.
Tager, A. M., LaCamera, P., Shea, B. S., Campanella, G. S., Selman, M., Zhao, Z., Polosukhin, V., Wain, J., Karimi-Shah, B. A., Kim, N. D., Hart, W. K., Pardo, A., Blackwell, T. S., Xu, Y., Chun, J., & Luster, A. D. (2008). The lysophosphatidic acid receptor LPA1 links pulmonary fibrosis to lung injury by mediating fibroblast recruitment and vascular leak. Nature Medicine, 14, 45–54.
Zhao, J., He, D., Su, Y., Berdyshev, E., Chun, J., Natarajan, V., & Zhao, Y. (2011). Lysophosphatidic acid receptor 1 modulates lipopolysaccharide-induced inflammation in alveolar epithelial cells and murine lungs. American Journal of Physiology - Lung Cellular and Molecular Physiology, 301, L547–556.
He, D., Su, Y., Usatyuk, P. V., Spannhake, E. W., Kogut, P., Solway, J., Natarajan, V., & Zhao, Y. (2009). Lysophosphatidic acid enhances pulmonary epithelial barrier integrity and protects endotoxin-induced epithelial barrier disruption and lung injury. Journal of Biological Chemistry, 284, 24123–24132.
Chen, X., Walther, F. J., Laghmani, E. H., Hoogeboom, A. M., Hogen-Esch, A. C., van Ark, I., Folkerts, G., & Wagenaar, G. T. (2017). Adult Lysophosphatidic Acid Receptor 1-Deficient Rats with Hyperoxia-Induced Neonatal Chronic Lung Disease Are Protected against Lipopolysaccharide-Induced Acute Lung Injury. Frontiers in Physiology, 8, 155.
Dong, C., Filipeanu, C. M., Duvernay, M. T., & Wu, G. (2007). Regulation of G protein-coupled receptor export trafficking. Biochimics et Biophysica Acta, 1768, 853–870.
Min, C., Zheng, M., Zhang, X., Guo, S., Kwon, K. J., Shin, C. Y., Kim, H. S., Cheon, S. H., & Kim, K. M. (2015). N-linked Glycosylation on the N-terminus of the dopamine D2 and D3 receptors determines receptor association with specific microdomains in the plasma membrane. Biochimica et Biophysica Acta, 1853, 41–51.
Lanctot, P. M., Leclerc, P. C., Clement, M., Auger-Messier, M., Escher, E., Leduc, R., & Guillemette, G. (2005). Importance of N-glycosylation positioning for cell-surface expression, targeting, affinity and quality control of the human AT1 receptor. Biochemical Journal, 390, 367–376.
Zhao, J., Wei, J., Bowser, R. K., Dong, S., Xiao, S., & Zhao, Y. (2014). Molecular regulation of lysophosphatidic acid receptor 1 trafficking to the cell surface. Cell Signal, 26, 2406–2411.
Varsano, T., Taupin, V., Guo, L., Baterina, Jr, O. Y., & Farquhar, M. G. (2012). The PDZ protein GIPC regulates trafficking of the LPA1 receptor from APPL signaling endosomes and attenuates the cell’s response to LPA. PLoS ONE, 7, e49227.
Suckau, O., Gross, I., Schrotter, S., Yang, F., Luo, J., Wree, A., Chun, J., Baska, D., Baumgart, J., Kano, K., Aoki, J., & Brauer, A. U. (2019). LPA1, LPA2, LPA4, and LPA6 receptor expression during mouse brain development. Developmental Dynamics, 248, 375–395.
Tao, Y. X., & Conn, P. M. (2014). Chaperoning G protein-coupled receptors: from cell biology to therapeutics. Endocrine Reviews, 35, 602–647.
Chrencik, J. E., Roth, C. B., Terakado, M., Kurata, H., Omi, R., Kihara, Y., Warshaviak, D., Nakade, S., Asmar-Rovira, G., Mileni, M., Mizuno, H., Griffith, M. T., Rodgers, C., Han, G. W., Velasquez, J., Chun, J., Stevens, R. C., & Hanson, M. A. (2015). Crystal Structure of Antagonist Bound Human Lysophosphatidic Acid Receptor 1. Cell, 161, 1633–1643.
Ishii, S., Tsujiuchi, T., & Fukushima, N. (2017). Functional characterization of lysophosphatidic acid receptor 1 mutants identified in rat cancer tissues. Biochemical and Biophysical Research Communcations, 486, 767–773.
Probst, W. C., Snyder, L. A., Schuster, D. I., Brosius, J., & Sealfon, S. C. (1992). Sequence alignment of the G-protein coupled receptor superfamily. DNA and Cell Biology, 11, 1–20.
Wu, G., Davis, J. E., & Zhang, M. (2015). Regulation of alpha2B-Adrenerigc Receptor Export Trafficking by Specific Motifs. Progress in Molecular Biology & Translation Science, 132, 227–244.
Ragnarsson, L., Andersson, A., Thomas, W. G., and Lewis, R. J. (2019). Mutations in the NPxxY motif stabilize pharmacologically distinct conformational states of the alpha1B- and beta2-adrenoceptors. Science Signaling, 12, eaas9485.
Calebiro, D., & Godbole, A. (2018). Internalization of G-protein-coupled receptors: Implication in receptor function, physiology and diseases. Best Practice & Research Clinical Endocrinology & Metabolism, 32, 83–91.
Gonda, A., Kabagwira, J., Senthil, G. N., & Wall, N. R. (2019). Internalization of Exosomes through Receptor-Mediated Endocytosis. Molecular Cancer Research, 17, 337–347.
Zhao, J., Wei, J., Dong, S., Bowser, R. K., Zhang, L., Jacko, A. M., & Zhao, Y. (2016). Destabilization of Lysophosphatidic Acid Receptor 1 Reduces Cytokine Release and Protects Against Lung Injury. EBioMedicine, 10, 195–203.
Alcantara-Hernandez, R., Hernandez-Mendez, A., Campos-Martinez, G. A., Meizoso-Huesca, A., & Garcia-Sainz, J. A. (2015). Phosphorylation and Internalization of Lysophosphatidic Acid Receptors LPA1, LPA2, and LPA3. PLoS ONE, 10, e0140583.
Meizoso-Huesca, A., Villegas-Comonfort, S., Romero-Avila, M. T., & Garcia-Sainz, J. A. (2018). Free fatty acid receptor 4 agonists induce lysophosphatidic acid receptor 1 (LPA1) desensitization independent of LPA1 internalization and heterodimerization. FEBS Letters, 592, 2612–2623.
Nan, L., Wei, J., Jacko, A. M., Culley, M. K., Zhao, J., Natarajan, V., Ma, H., & Zhao, Y. (2016). Cross-talk between lysophosphatidic acid receptor 1 and tropomyosin receptor kinase A promotes lung epithelial cell migration. Biochimica et Biophysica Acta, 1863, 229–235.
Avendano-Vazquez, S. E., Garcia-Caballero, A., & Garcia-Sainz, J. A. (2005). Phosphorylation and desensitization of the lysophosphatidic acid receptor LPA1. Biochemical Journal, 385, 677–684.
Urs, N. M., Kowalczyk, A. P., & Radhakrishna, H. (2008). Different mechanisms regulate lysophosphatidic acid (LPA)-dependent versus phorbol ester-dependent internalization of the LPA1 receptor. Journal of Biological Chemistry, 283, 5249–5257.
Gonzalez-Arenas, A., Avendano-Vazquez, S. E., Cabrera-Wrooman, A., Tapia-Carrillo, D., Larrea, F., Garcia-Becerra, R., & Garcia-Sainz, J. A. (2008). Regulation of LPA receptor function by estrogens. Biochimica et Biophysica Acta, 1783, 253–262.
Urs, N. M., Jones, K. T., Salo, P. D., Severin, J. E., Trejo, J., & Radhakrishna, H. (2005). A requirement for membrane cholesterol in the beta-arrestin- and clathrin-dependent endocytosis of LPA1 lysophosphatidic acid receptors. Journal of Cell Science, 118, 5291–5304.
Colin-Santana, C. C., Avendano-Vazquez, S. E., Alcantara-Hernandez, R., & Garcia-Sainz, J. A. (2011). EGF and angiotensin II modulate lysophosphatidic acid LPA(1) receptor function and phosphorylation state. Biochimica et Biophysica Acta, 1810, 1170–1177.
Mettlen, M., Chen, P. H., Srinivasan, S., Danuser, G., & Schmid, S. L. (2018). Regulation of Clathrin-Mediated Endocytosis. Annual Review of Biochemistry, 87, 871–896.
Takei, K., & Haucke, V. (2001). Clathrin-mediated endocytosis: membrane factors pull the trigger. Trends in Cell Biology, 11, 385–391.
Murph, M. M., Scaccia, L. A., Volpicelli, L. A., & Radhakrishna, H. (2003). Agonist-induced endocytosis of lysophosphatidic acid-coupled LPA1/EDG-2 receptors via a dynamin2- and Rab5-dependent pathway. Journal of Cell Science, 116, 1969–1980.
Weinberg, Z. Y., & Puthenveedu, M. A. (2019). Regulation of G protein-coupled receptor signaling by plasma membrane organization and endocytosis. Traffic, 20, 121–129.
Marchese, A., Paing, M. M., Temple, B. R., & Trejo, J. (2008). G protein-coupled receptor sorting to endosomes and lysosomes. Annual Review of Pharmacology and Toxicology, 48, 601–629.
Waters, C. M., Saatian, B., Moughal, N. A., Zhao, Y., Tigyi, G., Natarajan, V., Pyne, S., & Pyne, N. J. (2006). Integrin signalling regulates the nuclear localization and function of the lysophosphatidic acid receptor-1 (LPA1) in mammalian cells. Biochemical Journal, 398, 55–62.
Moughal, N. A., Waters, C., Sambi, B., Pyne, S., & Pyne, N. J. (2004). Nerve growth factor signaling involves interaction between the Trk A receptor and lysophosphatidate receptor 1 systems: nuclear translocation of the lysophosphatidate receptor 1 and Trk A receptors in pheochromocytoma 12 cells. Cell Signal, 16, 127–136.
Zhu, T., Gobeil, F., Vazquez-Tello, A., Leduc, M., Rihakova, L., Bossolasco, M., Bkaily, G., Peri, K., Varma, D. R., Orvoine, R., & Chemtob, S. (2006). Intracrine signaling through lipid mediators and their cognate nuclear G-protein-coupled receptors: a paradigm based on PGE2, PAF, and LPA1 receptors. Canadian Journal of Physiology and Pharmacology, 84, 377–391.
Skieterska, K., Rondou, P., & Van Craenenbroeck, K. (2017) Regulation of G Protein-Coupled Receptors by Ubiquitination. International Journal of Molecular Sciences, 18, 923.
Alonso, V., & Friedman, P. A. (2013). Minireview: ubiquitination-regulated G protein-coupled receptor signaling and trafficking. Molecular Endocrinology, 27, 558–572.
Tsai, Y. C., & Weissman, A. M. (2011). Ubiquitylation in ERAD: reversing to go forward? PLoS Biology, 9, e1001038.
Popovic, D., Vucic, D., & Dikic, I. (2014). Ubiquitination in disease pathogenesis and treatment. Nature Medicine, 20, 1242–1253.
Taleb, S. J., Wei, J., Mialki, R. K., Dong, S., Li, Y., Zhao, J., & Zhao, Y. (2021). A blocking peptide stabilizes lysophosphatidic acid receptor 1 and promotes lysophosphatidic acid-induced cellular responses. Journal of Cellular Biochemistry. in press
Cai, J., Culley, M. K., Zhao, Y., & Zhao, J. (2018). The role of ubiquitination and deubiquitination in the regulation of cell junctions. Protein Cell, 9, 754–769.
Harrigan, J. A., Jacq, X., Martin, N. M., & Jackson, S. P. (2018). Deubiquitylating enzymes and drug discovery: emerging opportunities. Nature Reviews Drug Discovery, 17, 57–78.
Reyes-Turcu, F. E., Ventii, K. H., & Wilkinson, K. D. (2009). Regulation and cellular roles of ubiquitin-specific deubiquitinating enzymes. Annual Review of Biochemistry, 78, 363–397.
Paiva, S. L., & Crews, C. M. (2019). Targeted protein degradation: elements of PROTAC design. Current Opinion in Chemical Biology, 50, 111–119.
Neklesa, T. K., Winkler, J. D., & Crews, C. M. (2017). Targeted protein degradation by PROTACs. Pharmacology & Therapeutics, 174, 138–144.
Acknowledgements
This work was supported by grants from National Institutes of Health (R01HL131665, R01HL157164, HL136294 to Y.Z., R01 GM115389, R01HL151513 to J.Z.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, J., Stephens, T. & Zhao, Y. Molecular Regulation of Lysophosphatidic Acid Receptor 1 Maturation and Desensitization. Cell Biochem Biophys 79, 477–483 (2021). https://doi.org/10.1007/s12013-021-00999-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-021-00999-6