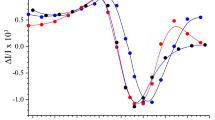
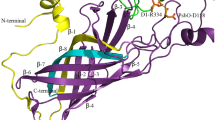
Abstract
Photosystem II (PS II) of oxygenic photosynthesis is found in the thylakoid membranes of plastids and cyanobacteria. The mature PS II complex comprises a central core of four membrane proteins that bind the majority of the redox-active cofactors. In cyanobacteria the central core is surrounded by 13 low-molecular-weight (LMW) subunits which each consist of one or two transmembrane helices. Three additional hydrophilic subunits known as PsbO, PsbU and PsbV are found associated with hydrophilic loops belonging to the core proteins protruding into the thylakoid lumen. During biogenesis the majority of the LMW subunits are known to initially associate with individual pre-assembly complexes consisting of one or more of the core proteins; however, the point at which the PsbJ LMW subunit binds to PS II is not known. The majority of models for PS II biogenesis propose that the three extrinsic proteins and PsbJ bind in the final stages of PS II assembly. We have investigated the impact of creating the double mutants ∆PsbJ:∆PsbO, ∆PsbJ:∆PsbU and ∆PsbJ:∆PsbV to investigate potential cooperation between these subunits in the final stages of biogenesis. Our results indicate that PsbJ can bind to PS II in the absence of any one of the extrinsic proteins. However, unlike their respective single mutants, the ∆PsbJ:∆PsbO and ∆PsbJ:∆PsbV strains were not photoautotrophic and were unable to support oxygen evolution suggesting a functional oxygen-evolving complex could not assemble in these strains. In contrast, the PS II centers formed in the ∆PsbJ:∆PsbU strain were capable of photoautotrophic growth and could support oxygen evolution when whole-chain electron transport was supported by the addition of bicarbonate.




Similar content being viewed by others
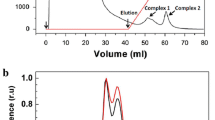
References
Ago H, Adachi H, Umena Y et al (2016) Novel features of eukaryotic photosystem II revealed by its crystal structure analysis from a red alga. J Biol Chem 291:5676–5687. https://doi.org/10.1074/jbcM115.711689
Bentley FK, Eaton-Rye JJ (2008) The effect of removing Photosystem II extrinsic proteins on dimer formation and recovery from photodamage in Synechocystis sp. PCC 6803. In: Allen JF, Gantt E, Golbeck JH, Osmond B (Eds) Photosynthesis. Energy from the sun: 14th International Congress on Photosynthesis, Springer, Dordrecht, pp 715–717. https://doi.org/10.1007/978-1-4020-6709-9_159
Bricker TM, Roose JL, Fagerlund RD et al (2012) The extrinsic protiens of photosystem II. Biochim Biophys Acta 1817:121–142. https://doi.org/10.1016/j.bbabio.2011.07.006
Burnap RL, Sherman LA (1991) Deletion mutagenesis in Synechocystis sp. PCC 6803 indicates that the Mn-stabilizing protein of photosystem II is not essential for oxygen evolution. Biochemistry 30:440–446. https://doi.org/10.1021/bi00216a020
Cardona T (2016) Reconstructing the origin of oxygenic photosynthesis: do assembly and photoactivation recapitulate evolution? Front Plant Sci 7:257. https://doi.org/10.3389/fpls.2016.00257
Chamovitz D, Sandmann G, Hirschberg J (1993) Molecular and biochemical characterization of herbicide-resistant mutants of cyanobacteria reveals that phytoene desaturation is a rate-limiting step in carotenoid biosynthesis. J Biol Chem 268:17348–17353. https://doi.org/10.1016/S0021-9258(19)85341-3
Eaton-Rye JJ (2011) Construction of gene interruptions and gene deletions in the cyanobacterium Synechocystis sp. strain PCC 6803. Meth Mol Biol 684:295–312. https://doi.org/10.1007/978-1-60761-925-3_22
Eaton-Rye JJ, Sobotka R (2017) Editorial: assembly of the photosystem II membrane-protein complex of oxygenic photosynthesis. Front Plant Sci 8:884. https://doi.org/10.3389/fpls.2017.00884
Eaton-Rye JJ, Shand JA, Nicoll WS (2003) pH-dependent photoautotrophic growth of specific Photosystem II mutants lacking lumenal extrinsic polypeptides in Synechocystis PCC 6803. FEBS Lett 543:148–153. https://doi.org/10.1016/s0014-5793(03)00432-0
Fagerlund RD, Forsman JA, Biswas S et al (2020) Stabilization of photosystem II by the PabT protein impacts photodamage, repair amd biogenesis. Biochim Biophys Acta Bioenerg 1861:148234. https://doi.org/10.1016/j.bbabio.2020.148234
Ferreira KN, Iverson TM, Maghlaoui K et al (2004) Architecture of the photosynthetic oxygen-evolving center. Science 303:1831–1838. https://doi.org/10.1126/science.1093087
Forsman JA, Eaton-Rye JJ (2021) The interaction between PsbT and the DE loop of D1 in Photosystem II stabilizes the quinone-iron electron acceptor complex. Biochemistry 60:53–63. https://doi.org/10.1021/acs.biochem.0c00668
Garcia-Cerdán JG, Furst AL, McDonald KL et al (2019) A thylakoid membrane-bound and redox-active rubredoxin (RBD1) functions in de novo assembly and repair of photosystem II. Proc Natl Acad Sci USA 116:16631–16640. https://www.pnas.org/content/116/33/16631
Govindjee (1995) Sixty-three years since Kautsky: chlorophyll a fluorescence. Aust J Plant Physiol 22:131–160. https://doi.org/10.1071/PP9950131
Guskov A, Kern J, Gabdulkhakov A et al (2009) Cyanobacterial photosystem II at 2.9-Å resolution and the role of quinones, lipids, channels and chloride. Nat Struct Mol Biol 16:334–342. https://doi.org/10.1038/nsmb.1559
Hager M, Hermann M, Biehler K et al (2002) Lack of the small plastid-encoded PsbJ polypeptide results in a defective water-splitting apparatus of Photosystem II, reduced Photosystem I levels, and hypersensitivity to light. J Biol Chem 277:14031–14039. https://doi.org/10.1074/jbc.M112053200
Heinz S, Liauw P, Nickelsen J, Nowaczyk M (2016) Analysis of photosystem II biogenesis in cyanobacteria. Biochim Biophys Acta 1857:274–287. https://doi.org/10.1016/j.bbabio.2015.11.007
Ho FM, Styring S (2008) Access channels and methanol binding to the CaMn4 cluster in photosystem II based on solvent accessibility simulations, with implications for substrate water access. Biochim Biophys Acta 1777:140–153. https://doi.org/10.1016/j.bbabio.2007.08.009
Huang G, Xiao Y, Pi X et al (2021) Structural insights into a dimeric Psb27-photosystem II complex from a cyanobacterium Thermosynechococcus vulcanus. Proc Natl Acad Sci USA 118(5):e2018053118. https://doi.org/10.1073/pnas.2018053118
Jackson SA, Eaton-Rye JJ (2015) Characterization of a Synechocystis sp. PCC 6803 double mutant lacking the CyanoP and Ycf48 proteins of photosystem II. Photosynth Res 124:217–229. https://doi.org/10.1007/s11120-015-0122-0
Jackson SA, Hervey JRD, Dale AJ, Eaton-Rye JJ (2014) Removal of both Ycf48 and Psb27 in Synechocystis sp. PCC 6803 disrupts photosystem II assembly and alters QA- oxidation in the mature complex. FEBS Lett 588:3751–3760. https://doi.org/10.1016/j.febslet.2014.08.024
Kern J, Chatterjee R, Young ID et al (2018) Structures of the intermediates of Kok’s photosynthetic water oxidation clock. Nature 563:421–425. https://doi.org/10.1038/s41586-018-0681-2
Komenda J, Knoppová J, Krynická V et al (2010) Role of FtsH2 in the repair of photosystem II in mutants of the cyanobacterium Synechocystis PCC 6803 with impaired assembly or stability of the CaMn4 cluster. Biochim Biophys Acta 1797:566–575. https://doi.org/10.1016/j.bbabio.2010.02.006
Komenda J, Sobotka R, Nixon PJ (2012) Assembling and maintaining the photosystem II complex in chloroplasts and cyanobacteria. Curr Opin Plant Biol 15:245–251. https://doi.org/10.1016/j.pbi.2012.01.017
Lea-Smith DJ, Bombelli P, Vasudevan R, Howe CJ (2016) Photosynthetic, respiratory and extracellular electron transport pathways in cyanobacteria. Biochim Biophys Acta 1857:247–255. https://doi.org/10.1016/j.bbabio.2015.10.007
Luo H, Jackson SA, Fagerlund RD et al (2014) The importance of the hydrophilic region of PsbL for the plastoqunone electron acceptor complex of photosystem II. Biochim Biophys Acta 1837:1435–1446. https://doi.org/10.1016/j.bbabio.2014.02.015
Mabbitt PD, Wilbanks SM, Eaton-Rye JJ (2014) Structure and function of the hydrophilic Photosystem II assembly proteins: Psb27, Psb28 and Ycf48. Plant Physiol Biochem 81:96–107. https/doi.org/https://doi.org/10.1016/j.plaphy.2014.02.013
MacKinney G (1941) Absorption of light by chlorophyll solutions. J Biol Chem 140:315–322. https://doi.org/10.1016/S0021-9258(18)51320-X
Morris JN, Crawford TS, Jeffs A et al (2014) Whole genome re-sequencing of two ‘wild-type’ strains of the model cyanobacterium Synechocystis sp. PCC 6803. N Z J Bot 52:36–47. https://doi.org/10.1080/0028825X.2013.846267
Morris JN, Eaton-Rye JJ, Summerfield TC (2016) Environmental pH and the requirement for the extrinsic proteins of Photosystem II in the function of cyanobacterial photosynthesis. Front Plant Sci 7:1135. https://doi.org/10.3389/fpls.2016.01135
Morris JN, Kovács S, Vass I et al (2019) Environmental pH and a Glu364 to Gln mutation in the chlorophyll-binding CP47 protein affect reox-active TyrD and charge recombination in Photosystem II. FEBS Lett 593:163–174. https://doi.org/10.1002/1873-3468.13307
Müh F, Renger T, Zouni A (2008) Crystal structure of cyanobacterial photosystem II at 3.0 Å resolution: a closer look at the antenna system and the small membrane-intrinsic subunits. Plant Physiol Biochem 46:238–264. https://doi.org/10.1016/j.plaphy.2008.01.003
Müh F, Zouni A (2016) Cytochrome b559 in photosystem II. In: Cramer WA, Kallas T (eds) Cytochrome complexes: evolution, structures, energy transduction, and signaling, Advances in Photosynthesis and Respiration, Vol 22, Springer, Dordrecht, pp 143–175. https://doi.org/10.1007/978-94-017-7481-9_8
Nickelsen J, Rengstl B (2013) Photosystem II assembly: from cyanobacteria to plants. Annu Rev Plant Biol 64:609–635. https://doi.org/10.1146/annurev-arplant-050312-120124
Nowaczyk MM, Krause K, Mieseler M et al (2012) Deletion of psbJ leads to accumulation of Psb27–Psb28 Photosystem II complexes in Thermosynechococcus elongatus. Biochim Biophys Acta 1817:1339–1343. https://doi.org/10.1016/j.bbabio.2012.02.017
Paerl HW (1984) Cyanobacterial carotenoids: their roles in maintaining optimal production among aquatic bloom forming genera. Oecologia 61:143–149. https://doi.org/10.1007/BF00396752
Regel RE, Ivleva NB, Zer H et al (2001) Deregulation of electron flow within photosystem II in the absence of the PsbJ protein. J Biol Chem 276:41473–41478. https://doi.org/10.1074/jbc.M102007200
Roose JL, Frankel LK, Mummadisetti MP, Bricker TM (2016) The extrinsic proteins of photosystem II: update. Planta 243:889–908. https://doi.org/10.1007/s00425-015-2462-6
Shen J-R (2015) The structure of photosystem II and the mechanism of water oxidation in photosynthesis. Annu Rev Plant Biol 66:23–48. https://doi.org/10.1146/annurev-arplant-050312-120129
Shen J-R, Vermaas W, Inoue Y (1995a) The role of cytochrome c-550 as studied through reverse genetics and mutant characterization in Synechocystis sp. PCC 6803. J Biol Chem 270:6901–6907. https://doi.org/10.1074/jbc.270.12.6901
Shen J-R, Burnap RL, Inoue Y (1995b) An independent role of cytochrome c-550 in cyanobacterial Photosystem II as revealed by double-deletion mutagenesis of the psbO and psbV genes in Synechocystis sp. PCC 6803. Biochemistry 34:12661–12668. https://doi.org/10.1021/bi00039a023
Shen J-R, Ikeuchi M, Inoue Y (1997) Analysis of the psbU gene encoding the 12-kDa extrinsic protein of Photosystem II and studies on its role by deletion mutagenesis in Synechocystis sp. PCC 6803. J Biol Chem 272:17821–17826. https://doi.org/10.1074/jbc.272.28.17821
Shi L-X, Hall M, Funk C, Schröder WP (2012) Photosystem II, a growing complex: updates on newly discovered components and low molecular mass proteins. Biochim Biophys Acta 1817:13–25. https://doi.org/10.1016/j.bbabio.2011.08.008
Suga M, Akita F, Hirata K et al (2015) Native structure of photosystem II at 1.95 Å resolution viewed by femtosecond X-ray pulses. Nature 517:99–103. https://doi.org/10.1038/nature13991
Suga M, Akita F, Sugahara M et al (2017) Light-induced structural changes and the site of O=O bond formation in PS II caught by XFEL. Nature 543:131–135. https://doi.org/10.1038/nature21400
Summerfield TC, Eaton-Rye JJ, Sherman LA (2007) Global gene expression of a ∆PsbO:∆PsbU mutant and a spontaneous revertant in the cyanobacterium Synechocystis sp. strain PCC 6803. Photosynth Res 94:265–274. https://doi.org/10.1007/s11120-007-9237-2
Summerfield TC, Crawford TS, Young RD et al (2013) Environmental pH affects photoautotrophic growth of Synechocystis sp. PCC 6803 strains carrying mutations in the lumenal proteins of photosystem II. Plant Cell Physiol 54:859–874. https://doi.org/10.1093/pcp/pct036
Suorsa M, Regel RE, Paakkarinen V et al (2004) Protein assembly of photosystem II and accumulation of subcomplexes in the absence of low molecular mass subunits PsbL and PsbJ. Eur J Biochem 271:96–107. https://doi.org/10.1046/j.1432-1033.2003.03906.x
Swiatek M, Regel RE, Meurer J et al (2003) Effects of selective inactivation of individual genes for low-molecular-mass subunits on the assembly of Photosystem II, as revealed by chloroplast transformation: the psbEFLJ operon in Nicotiana tabacum. Mol Gen Genom 268:699–710. https://doi.org/10.1007/s00438-002-0791-1
Tóth TN, Chukhutsina V, Domonkos I et al (2015) Carotenoids are essential for the assembly of cyanobacterial photosynthetic complexes. Biochim Biophys Acta 1847:1153–1165. https://doi.org/10.1016/j.bbabio.2015.05.020
Umena Y, Kawakami K, Shen JR, Kamiya N (2011) Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9 Å. Nature 473:55–60. https://doi.org/10.1038/nature09913
Vajravel S, Kis M, Kłodawska K et al (2017) Zeaxanthin and echinenone modify the structure of photosystem I trimer in Synechocystis sp. PCC 6803. Biochim Biophys Acta Bioenerg 1858:510–518. https://doi.org/10.1016/j.bbabio.2017.05.001
Vinyard DJ, Brudvig GW (2018) Progress toward a molecular mechanism of water oxidation in photosystem II. Annu Rev Phys Chem 68:101–116. https://doi.org/10.1146/annurev-physchem-052516-044820
Wei X, Su X, Cao P et al (2016) Structure of spinach photosystem II – LHCII supercomplex at 3.2 Å resolution. Nature 534:69–74. https://doi.org/10.1038/nature18020
Williams JGK (1988) Construction of specific mutations in photosystem II photosynthetic reaction center by genetic engineering methods in Synechocystis 6803. Methods Enzymol 167:766–778
Yu J, Knoppová J, Michoux F et al (2018) Ycf48 involved in the biogenesis of the oxygen-evolving photosystem II complex is a seven-bladed beta-propeller protein. Proc Natl Acad Sci USA 115:E7824–E7833. https://doi.org/10.1073/pnas.1800609115
Zabret J, Bohn S, Schuller SK et al (2021) Structural insights into photosystem II assembly. Nat Plants. 7:524–538. https://doi.org/10.1038/s41477-021-00895
Acknowledgements
PC and JAF contributed equally to this project and are listed as joint first authors in alphabetical order. We thank Dr Sandeep Biswas for assistance with the construction of the plasmids used to delete the genes encoding the PsbO, PsbU and PsbV extrinsic proteins.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Choo, P., Forsman, J.A., Hui, L. et al. The PsbJ protein is required for photosystem II activity in centers lacking the PsbO and PsbV lumenal subunits. Photosynth Res 151, 103–111 (2022). https://doi.org/10.1007/s11120-021-00862-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-021-00862-y