Abstract
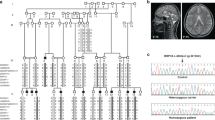
Autosomal recessive microcephaly and chorioretinopathy (MCCRP) is a neurodevelopmental disorder characterized by delayed psychomotor development, growth retardation with dwarfism, and ocular abnormalities, and its occurrence has been found to be closely related to variants of the gene encoding centrosomes. However, the association between centrosomal duplication defects and the etiology of microcephaly syndromes is poorly understood. It is well known that polo-like kinase 4 (PLK4) is a key regulator of centriole duplication, and the abnormalities of centrosomal function caused by its protein variation need to be further explored in the pathogenesis of microcephaly. In our study, we found that a patient with microcephaly and chorioretinopathy harbored compound heterozygous missense variants NM_014264.4: c.2221C > T (p.Gln741*) and NM_014264.4: c.2062 T > C (p.Tyr688His) in the PLK4 gene. Overexpression experiments of the variant PLK4 proteins then showed that the G741 variant rather than the T688H variant had lost centrosomal amplification ability, and the G741 variant but not the T688H variant induced centrosomal replication disorder, which further inhibited cell proliferation, cycle division and cytoskeleton morphology in HeLa cells. Moreover, the overexpression of the two variant proteins had inconsistent effects on the target protein PLK4 by western blot analysis, also indicating that T688H variant overexpression is not functionally equivalent to WT-PLK4 overexpression. Therefore, all data support the idea that the PLK4 mutation induces centriolar duplication disorder and reduces the efficiency of mitosis inducing cell death or cell proliferation in the etiology of microcephaly disorder.




Similar content being viewed by others
Availability of Data and Materials
All data can be uploaded if necessary
Abbreviations
- MCCRP:
-
Autosomal recessive microcephaly and chorioretinopathy
- MCPH:
-
Autosomal recessive microcephaly
- PLK4:
-
Polo-like kinase 4
- MTT:
-
3-[4,5-Dimethyl-2-thiazolyl]-2,5-diphenyl-2H-tetrazolium bromide
- siRNA:
-
Small interfering RNA
- FBS:
-
Fetal bovine serum
- DMEM:
-
Dulbecco's modified Eagle's medium
- PMSF:
-
Phenylmethanesulfonyl fluoride
References
Alieva IB, Vorobjev IA (1994) Centrosome behavior under the action of a mitochondrial uncoupler and the effect of disruption of cytoskeleton elements on the uncoupler-induced alterations. J Struct Biol 113(3):217–224
Chavali PL, Pütz M, Gergely F (2014) Small organelle, big responsibility: the role of centrosomes in development and disease. Philos Trans R Soc Lond B Biol Sci 369:20130468
Dzafic E, Strzyz P, Wilsch-Bräuninger M et al (2015) Centriole Amplification in Zebrafish Affects Proliferation and Survival but Not Differentiation of Neural Progenitor Cells. Cell Rep 13(1):168–182
Habedanck R, Stierhof YD, Wilkinson CJ et al (2005) The Polo-kinase Plk4 functions in centriole duplication. Nat Cell Biol 7:1140–1146
Hull S, Arno G, Ostergaard P et al (2019) Clinical and molecular characterization of familial exudative vitreoretinopathy associated with microcephaly. Am J Ophthalmol 207:87–98
Jean C, Tollon Y, Raynaud-Messina B et al (1999) The mammalian interphase centrosome: two independent units maintained together by the dynamics of the microtubule cytoskeleton. Eur J Cell Biol 78(8):549–560
Kaindl AM, Passemard S, Kumar P et al (2010) Many roads lead to primary autosomal recessive microcephaly. Prog Neurobiol 90(3):363–383
Katrien S, Moortgat S, Tim V et al (2018) Biallelic mutations in RTTN are associated with microcephaly, short stature and a wide range of brain malformations. Eur J Med Genet 61(12): 733–737
Kim M, O’Rourke B, Soni RK et al (2016) Promotion and Suppression of Centriole Duplication Are Catalytically Coupled through PLK4 to Ensure Centriole Homeostasis. Cell Rep 16(5):1195–1203
Kleylein-Sohn J, Westendorf J, Clech ML et al (2007) Plk4-Induced Centriole Biogenesis in Human Cells. Dev Cell 13(2):190–202
Luk HY, Mcfarlin BK, Vingren JL (2016) Using image-based flow cytometry to monitor satellite cells proliferation and differentiation in vitro. Methods 112:175–181
Martin CA, Ahmad I, Klingseisen A et al (2014) Mutations in PLK4, encoding a master regulator of centriole biogenesis, cause microcephaly, growth failure and retinopathy. Nat Genet 46:1283–1292
Marthiens V, Rujano MA, Pennetier C et al (2013) Centrosome amplification causes microcephaly. Nat Cell Biol 15:731–740
Meitinger F, Ohta M, Lee KY et al (2020) TRIM37 controls cancer-specific vulnerability to PLK4 inhibition. Nature 585:440–446
Morris-Rosendahl DJ, Kaindl AM (2015) What next-generation sequencing (NGS) technology has enabled us to learn about primary autosomal recessive microcephaly (MCPH). Mol Cell Probes 29:271–281
Muhammad IN, Angham AA, Mohammed MJ et al (2020) Next generation sequencing reveals novel homozygous frameshift in PUS7 and splice acceptor variants in AASS gene leading to intellectual disability, developmental delay, dysmorphic feature and microcephaly. Saudi J Biol Sci 27(11):3125–3131
Scheidecker S, Etard C, Haren L et al (2015) Mutations in TUBGCP4 Alter Microtubule Organization via the γ-Tubulin Ring Complex in Autosomal-Recessive Microcephaly with Chorioretinopathy. Am J Hum Genet 96(4):666–674
Soden SE, Saunders CJ, Willig LK et al (2014) Effectiveness of exome and genome sequencing guided by acuity of illness for diagnosis of neurodevelopmental disorders. Sci Transl Med 6(625):265ra168
Tsutsumi M, Yokoi S, Miya F et al (2016) Novel compound heterozygous variants in PLK4 identified in a patient with autosomal recessive microcephaly and chorioretinopathy. Eur J Hum Genet 24:1702–1706
Yokoi S, Ishihara N, Miya F et al (2015) TUBA1A mutation can cause a hydranencephaly-like severe form of cortical dysgenesis. Sci Rep 5:15165
Zhang SD, Yu L, Wang P et al (2019) Inotodiol inhibits cells migration and invasion and induces apoptosis via p53-dependent pathway in HeLa cells. Phytomedicine 60:152957
Acknowledgements
The authors gratefully acknowledge the financial support from the Xiamen Municipal Medical and Health Guidance Project (3502Z20209200).
Funding
The authors gratefully acknowledge the financial support from the Xiamen Municipal Medical and Health Guidance Project (3502Z20209200).
Author information
Authors and Affiliations
Contributions
Qi-Chang Wu, Ya-Song Xu, Zhi-Ying Su, Yi-Zhen Ji and Su-Qing Zhang conceived and performed the study. Li Sun, Xiao-Mei Yang, Shi-Yu Sun and Xiao-Fei Ji analyzed data. Ya-Song Xu and Zhi-Ying Su wrote the paper. Jie Tian and Qi-Chang Wu reviewed and edited the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics Statement
The genetic testing used in this study was approved by the ethical committees of Xiamen Maternal and Child Health Hospital in accordance with the Declaration of Helsinki. All patients provided informed consent, and all protocols were approved by the institutional committee of Xiamen Maternal and Child Health Hospital.
Consent for Publication
The work described has not been submitted elsewhere for publication, in whole or in part, and all the authors listed have approved the submission of the manuscript.
Conflict of Interest Statement
No conflict of interest exits in the submission of this manuscript, and the manuscript is approved by all authors for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, YS., Su, ZY., Sun, L. et al. Overexpression of the PLK4 Gene as a Novel Strategy for the Treatment of Autosomal Recessive Microcephaly by Improving Centrosomal Dysfunction. J Mol Neurosci 71, 2618–2627 (2021). https://doi.org/10.1007/s12031-021-01881-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-021-01881-z