Abstract
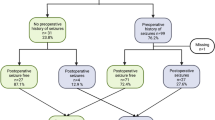
In previous studies, isocitrate dehydrogenase (IDH) mutations were associated with tumor-associated epilepsy (TAE) and venous thromboembolism (VTE). We examined the relationship between IDH mutations in grade II/III astrocytomas and TAE/VTE according to the 2016 World Health Organization classification. The clinical data of patients with newly diagnosed grade II/III gliomas who were treated at Tohoku University Hospital from January 2010 to December 2018 were reviewed. Associations between TAE or VTE and the clinical/biological characteristics, histology, and IDH1/2 mutational status in patients with grade II/III gliomas were evaluated. Of the initial 137 patients (290 hospitalizations), 117 patients (203 hospitalizations) were included in the TAE group and 124 patients (213 hospitalizations) were included in the VTE group. Seventy-eight patients (66.7%) in the TAE group were diagnosed with astrocytoma and 38/78 (48.3%) presented with TAE. According to the multivariable analysis, the IDH mutational status and male sex were associated independently with an increased risk of TAE (p < 0.05). Eighty-five patients (68.5%) in the VTE group were diagnosed with astrocytoma. VTE was observed in 16/161 (9.9%) hospitalizations. According to the multivariable analysis, age, diffuse astrocytoma histology, and resection were associated independently with an increased risk of VTE. The decision tree analysis showed that TAE was more frequent in younger patients while VTE was more frequent in older patients. This study demonstrated that the IDH mutational status was associated with TAE but not with VTE. Therefore, a future large-scale study is needed to provide sufficient evidence. TAE was more common in young patients, while VTE was more common in the elderly.


Similar content being viewed by others
References
Kerkhof M, Vecht CJ (2013) Seizure characteristics and prognostic factors of gliomas. Epilepsia 54(Suppl 9):12–17. https://doi.org/10.1111/epi.12437
van Breemen MS, Wilms EB, Vecht CJ (2007) Epilepsy in patients with brain tumours: epidemiology, mechanisms, and management. Lancet Neurol 6(5):421–430. https://doi.org/10.1016/S1474-4422(07)70103-5
Chen H, Judkins J, Thomas C et al (2017) Mutant IDH1 and seizures in patients with glioma. Neurology 88(19):1805–1813. https://doi.org/10.1212/WNL.0000000000003911
Chang EF, Potts MB, Keles GE et al (2008) Seizure characteristics and control following resection in 332 patients with low-grade gliomas. J Neurosurg 108(2):227–235. https://doi.org/10.3171/JNS/2008/108/2/0227
Cheruku R, Tapazoglou E, Ensley J et al (1991) The incidence and significance of thromboembolic complications in patients with high-grade gliomas. Cancer 68(12):2621–2624. https://doi.org/10.1002/1097-0142(19911215)68:12%3c2621::aid-cncr2820681218%3e3.0.co;2-7
Semrad TJ, O’Donnell R, Wun T et al (2007) Epidemiology of venous thromboembolism in 9489 patients with malignant glioma. J Neurosurg 106(4):601–608. https://doi.org/10.3171/jns.2007.106.4.601
Kawaguchi T, Kumabe T, Kanamori M et al (2011) Early detection of venous thromboembolism in patients with neuroepithelial tumor: efficacy of screening with serum D-dimer measurements and Doppler ultrasonography. J Neurooncol 101(3):495–504. https://doi.org/10.1007/s11060-010-0276-8
Watanabe J, Natsumeda M, Okada M et al (2019) Podoplanin expression and IDH-wildtype status predict venous thromboembolism in patients with high-grade gliomas in the early postoperative period. World Neurosurg 128:e982–e988. https://doi.org/10.1016/j.wneu.2019.05.049
Unruh D, Schwarze SR, Khoury L et al (2016) Mutant IDH1 and thrombosis in gliomas. Acta Neuropathol 132(6):917–930. https://doi.org/10.1007/s00401-016-1620-7
Auguste KI, Quinones-Hinojosa A, Gadkary C et al (2003) Incidence of venous thromboembolism in patients undergoing craniotomy and motor mapping for glioma without intraoperative mechanical prophylaxis to the contralateral leg. J Neurosurg 99(4):680–684. https://doi.org/10.3171/jns.2003.99.4.0680
Huberfeld G, Vecht CJ (2016) Seizures and gliomas–towards a single therapeutic approach. Nat Rev Neurol 12(4):204–216. https://doi.org/10.1038/nrneurol.2016.26
Louis DN, Perry A, Reifenberger G et al (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131(6):803–820. https://doi.org/10.1007/s00401-016-1545-1
Cancer Genome Atlas Research N, Brat DJ, Verhaak RG et al (2015) Comprehensive, integrative genomic analysis of diffuse lower-grade gliomas. N Engl J Med 372(26):2481–2498. https://doi.org/10.1056/NEJMoa1402121
Li Y, Shan X, Wu Z et al (2018) IDH1 mutation is associated with a higher preoperative seizure incidence in low-grade glioma: a systematic review and meta-analysis. Seizure 55:76–82. https://doi.org/10.1016/j.seizure.2018.01.011
Feyissa AM, Worrell GA, Tatum WO et al (2019) Potential influence of IDH1 mutation and MGMT gene promoter methylation on glioma-related preoperative seizures and postoperative seizure control. Seizure 69:283–289. https://doi.org/10.1016/j.seizure.2019.05.018
Nobusawa S, Watanabe T, Kleihues P et al (2009) IDH1 mutations as molecular signature and predictive factor of secondary glioblastomas. Clin Cancer Res 15(19):6002–6007. https://doi.org/10.1158/1078-0432.CCR-09-0715
Kanamori M, Kikuchi A, Watanabe M et al (2014) Rapid and sensitive intraoperative detection of mutations in the isocitrate dehydrogenase 1 and 2 genes during surgery for glioma. J Neurosurg 120(6):1288–1297. https://doi.org/10.3171/2014.3.JNS131505
Hatae R, Hata N, Yoshimoto K et al (2016) Precise detection of IDH1/2 and BRAF hotspot mutations in clinical glioma tissues by a differential calculus analysis of high-resolution melting data. PLoS ONE 11(8):e0160489. https://doi.org/10.1371/journal.pone.0160489
Shibahara I, Sonoda Y, Kanamori M et al (2012) IDH1/2 gene status defines the prognosis and molecular profiles in patients with grade III gliomas. Int J Clin Oncol 17(6):551–561. https://doi.org/10.1007/s10147-011-0323-2
Kanamori M, Kumabe T, Sonoda Y et al (2009) Predictive factors for overall and progression-free survival, and dissemination in oligodendroglial tumors. J Neurooncol 93(2):219–228. https://doi.org/10.1007/s11060-008-9762-7
Kawaguchi T, Kumabe T, Kanamori M et al (2013) Risk assessment for venous thromboembolism in patients with neuroepithelial tumors: pretreatment score to identify high risk patients. Neurol Med Chir 53(7):467–473. https://doi.org/10.2176/nmc.53.467
Sorensen HT, Mellemkjaer L, Olsen JH et al (2000) Prognosis of cancers associated with venous thromboembolism. N Engl J Med 343(25):1846–1850. https://doi.org/10.1056/NEJM200012213432504
van den Bent MJ, Smits M, Kros JM et al (2017) Diffuse infiltrating oligodendroglioma and astrocytoma. J Clin Oncol 35(21):2394–2401. https://doi.org/10.1200/JCO.2017.72.6737
Bardella C, Al-Dalahmah O, Krell D et al (2016) Expression of Idh 1(R132H) in the murine subventricular zone stem cell niche recapitulates features of early gliomagenesis. Cancer Cell 30(4):578–594. https://doi.org/10.1016/j.ccell.2016.08.017
Hesdorffer DC, Logroscino G, Benn EK et al (2011) Estimating risk for developing epilepsy: a population-based study in Rochester, Minnesota. Neurology 76(1):23–27. https://doi.org/10.1212/WNL.0b013e318204a36a
Kim DW, Lee SY, Chung SE et al (2014) Clinical characteristics of patients with treated epilepsy in Korea: a nationwide epidemiologic study. Epilepsia 55(1):67–75. https://doi.org/10.1111/epi.12469
Reddy DS (2017) The neuroendocrine basis of sex differences in epilepsy. Pharmacol Biochem Behav 152:97–104. https://doi.org/10.1016/j.pbb.2016.07.002
Hosie AM, Wilkins ME, da Silva HM et al (2006) Endogenous neurosteroids regulate GABAA receptors through two discrete transmembrane sites. Nature 444(7118):486–489. https://doi.org/10.1038/nature05324
Unruh D, Mirkov S, Wray B et al (2019) Methylation-dependent tissue factor suppression contributes to the reduced malignancy of IDH1-mutant gliomas. Clin Cancer Res 25(2):747–759. https://doi.org/10.1158/1078-0432.CCR-18-1222
Khorana AA, Francis CW, Culakova E et al (2007) Frequency, risk factors, and trends for venous thromboembolism among hospitalized cancer patients. Cancer 110(10):2339–2346. https://doi.org/10.1002/cncr.23062
Agnelli G, Bolis G, Capussotti L et al (2006) A clinical outcome-based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project. Ann Surg 243(1):89–95. https://doi.org/10.1097/01.sla.0000193959.44677.48
Stichel D, Ebrahimi A, Reuss D et al (2018) Distribution of EGFR amplification, combined chromosome 7 gain and chromosome 10 loss, and TERT promoter mutation in brain tumors and their potential for the reclassification of IDHwt astrocytoma to glioblastoma. Acta Neuropathol 136(5):793–803. https://doi.org/10.1007/s00401-018-1905-0
Brat DJ, Aldape K, Colman H et al (2018) cIMPACT-NOW update 3: recommended diagnostic criteria for “Diffuse astrocytic glioma, IDH-wildtype, with molecular features of glioblastoma, WHO grade IV.” Acta Neuropathol 136(5):805–810. https://doi.org/10.1007/s00401-018-1913-0
Riedl J, Preusser M, Nazari PM et al (2017) Podoplanin expression in primary brain tumors induces platelet aggregation and increases risk of venous thromboembolism. Blood 129(13):1831–1839. https://doi.org/10.1182/blood-2016-06-720714
Acknowledgements
The authors would like to thank Enago (http://www.enago.jp) for the English review.
Funding
This research was supported in part by AMED under Grant Number JP19lk0201070, JP20lm0203119.
Author information
Authors and Affiliations
Contributions
Conceptualization and design were performed by RS. Acquisition of data was performed by YO, RS, and MW. Statistical analysis was performed by YO and SM. Manuscript writing was performed by YO, RS, and SM. The final editing was performed by YS, TK, and TT. The first draft of the manuscript was written by YO and RS and all the authors commented on the various versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All of the authors declare no conflict of interest pertaining to this work.
Ethical approval
This study was conducted after obtaining the necessary ethical clearance from the institutional ethics board for study on human subjects of Tohoku University hospital and Sendai Medical Center.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Osada, Y., Saito, R., Miyata, S. et al. Association between IDH mutational status and tumor-associated epilepsy or venous thromboembolism in patients with grade II and III astrocytoma. Brain Tumor Pathol 38, 218–227 (2021). https://doi.org/10.1007/s10014-021-00406-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10014-021-00406-1