Abstract
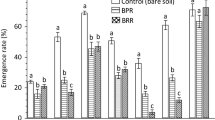
This study aimed to better understand the impact of different bauxite residues (BR) on the germination and the development of Sinapis alba (white mustard). Unamended BR from Provence, France (PRO), and Guinea (GUI) bauxite were selected, and modified bauxite residues from PRO and GUI (MBR-PRO and MBR-GUI) were obtained by gypsum application and repeated leaching, in order to reduce their pH, electrical conductivity (EC), and exchangeable sodium percentage (ESP). Germination rates were monitored in soil-BR mixtures with increasing concentrations of BR. A rhizotron experiment was done, where Sinapis alba was allowed to develop in a layer of soil on the top of a layer of bauxite residue. To assess the impact of the residue on root development, the WhinRhizo® software was used to measure the architectural traits of roots. Peroxidase and fluorescein hydrolase activities were also assessed on the roots grown either in the soil or in the residue layers. Results showed that (i) bauxite residue origin, (ii) bauxite residue modification by gypsum, and (iii) bauxite residue concentration had significant effects on the germination, the root development and architecture, and the enzymatic activities of the roots of Sinapis alba. The PRO residue had a far stronger phytotoxic effect compared to the GUI residue on every measured variable. The toxic effect was strongly modulated by the origin of bauxite residue. The gypsum application efficiently reduced the phytotoxic effect of the residues, but significant negative effects on the different variables were still recorded.




Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Alarcón-Gutiérrez, E., Floch, C., Ruaudel, F., & Criquet, S. (2008). Non-enzymatic hydrolysis of fluorescein diacetate (FDA) in a Mediterranean oak (Quercus ilex L.) litter. European Journal of Soil Science, 59(2), 139–146. https://doi.org/10.1111/j.1365-2389.2007.00963.x
Bojović, B., Đelić, G., Topuzović, M., & Stanković, M. (2010). Effects of NaCl on seed germination in some species from families Brassicaceae and Solanaceae. Kragujevac Journal of Science, 32, 83–87.
Bradshaw, A. D., & Chadwick, M. J. (1980). The restoration of land: the ecology and reclamation of derelict and degraded land. University of California Press.
Bray, A. W., Stewart, D. I., Courtney, R., Rout, S. P., Humphreys, P. N., Mayes, W. M., & Burke, I. T. (2018). Sustained bauxite residue rehabilitation with gypsum and organic matter 16 years after initial treatment. Environmental Science & Technology, 52(1), 152–161. https://doi.org/10.1021/acs.est.7b03568
Cao, Z., Geng, B., Xu, S., Xuan, W., Nie, L., Shen, W., et al. (2011). BnHO1, a haem oxygenase-1 gene from Brassica napus, is required for salinity and osmotic stress-induced lateral root formation. Journal of Experimental Botany, 62(13), 4675–4689. https://doi.org/10.1093/jxb/err190
Chaoui, A., & El Ferjani, E. (2005). Effects of cadmium and copper on antioxidant capacities, lignification and auxin degradation in leaves of pea (Pisum sativum L.) seedlings. Comptes Rendus Biologies, 328(1), 23–31. https://doi.org/10.1016/j.crvi.2004.10.001
Choudhury, S., Panda, P., Sahoo, L., & Panda, S. K. (2013). Reactive oxygen species signaling in plants under abiotic stress. Plant Signaling & Behavior, 8(4), e23681. https://doi.org/10.4161/psb.23681
Clark, L. J., Whalley, W. R., & Barraclough, P. B. (2003). How do roots penetrate strong soil? Plant and Soil, 255(1), 93–104. https://doi.org/10.1023/A:1026140122848
Correa, J., Postma, J. A., Watt, M., & Wojciechowski, T. (2019). Soil compaction and the architectural plasticity of root systems. Journal of Experimental Botany, 70(21), 6019–6034. https://doi.org/10.1093/jxb/erz383
Courtney, R., & Mullen, G. (2009). Use of germination and seedling performance bioassays for assessing revegetation strategies on bauxite residue. Water, Air, and Soil Pollution, 197(1–4), 15–22. https://doi.org/10.1007/s11270-008-9787-8
Courtney, R. G., & Timpson, J. P. (2005). Nutrient status of vegetation grown in alkaline bauxite processing residue amended with gypsum and thermally dried sewage sludge - A two year field study. Plant and Soil, 266(1–2), 187–194. https://doi.org/10.1007/s11104-005-0872-0
Courtney, Ronan, & Kirwan, L. (2012). Gypsum amendment of alkaline bauxite residue – plant available aluminium and implications for grassland restoration. Ecological Engineering, 42, 279–282. https://doi.org/10.1016/j.ecoleng.2012.02.025
Courtney, R., Feeney, E., & O’Grady, A. (2014). An ecological assessment of rehabilitated bauxite residue. Ecological Engineering, 73, 373–379. https://doi.org/10.1016/j.ecoleng.2014.09.064
Criquet, S., Joner, E. J., & Leyval, C. (2001). 2,7-Diaminofluorene is a sensitive substrate for detection and characterization of plant root peroxidase activities. Plant Science, 161(6), 1063–1066. https://doi.org/10.1016/S0168-9452(01)00491-5
Di Carlo, E., Boullemant, A., & Courtney, R. (2019). A field assessment of bauxite residue rehabilitation strategies. Science of The Total Environment, 663, 915–926. https://doi.org/10.1016/j.scitotenv.2019.01.376
Di Carlo, E., Boullemant, A., & Courtney, R. (2020). Ecotoxicological risk assessment of revegetated bauxite residue: Implications for future rehabilitation programmes. Science of The Total Environment, 698, 134344. https://doi.org/10.1016/j.scitotenv.2019.134344
Eleftheriou, E. P., Adamakis, I.-D.S., Panteris, E., & Fatsiou, M. (2015). Chromium-induced ultrastructural changes and oxidative stress in roots of Arabidopsis thaliana. International Journal of Molecular Sciences, 16(7), 15852–15871. https://doi.org/10.3390/ijms160715852
Feigl, G., Molnár, Á., Szőllősi, R., Ördög, A., Törőcsik, K., Oláh, D., et al. (2019). Zinc-induced root architectural changes of rhizotron-grown B. napus correlate with a differential nitro-oxidative response. Nitric Oxide, 90, 55–65. https://doi.org/10.1016/j.niox.2019.06.003
Finngean, G., O’Grady, A., & Courtney, R. (2018). Plant assays and avoidance tests with collembola and earthworms demonstrate rehabilitation success in bauxite residue. Environmental Science and Pollution Research, 25, 2157–2166. https://doi.org/10.1007/s11356-017-0632-8
Fourrier, C., Luglia, M., Hennebert, P., Foulon, J., Ambrosi, J.-P., Angeletti, B., et al. (2020). Effects of increasing concentrations of unamended and gypsum modified bauxite residues on soil microbial community functions and structure – a mesocosm study. Ecotoxicology and Environmental Safety, 201, 110847. https://doi.org/10.1016/j.ecoenv.2020.110847
Fuller, R. D., & Richardson, C. J. (1986). Aluminate toxicity as a factor controlling plant growth in bauxite residue. Environmental Toxicology and Chemistry, 5, 905–915.
Gräfe, M., & Klauber, C. (2011). Bauxite residue issues: IV. Old obstacles and new pathways for in situ residue bioremediation. Hydrometallurgy, 108(1), 46–59. https://doi.org/10.1016/j.hydromet.2011.02.005
Hackenberger, D. K., Feigl, V., Lončarić, Ž, & Hackenberger, B. K. (2019). Biochemical and reproductive effects of red mud to earthworm Eisenia fetida. Ecotoxicology and Environmental Safety, 168, 279–286. https://doi.org/10.1016/j.ecoenv.2018.10.097
Harris, M. A. (2009). Structural improvement of age-hardened gypsum-treated bauxite red mud waste using readily decomposable phyto-organics. Environmental Geology, 56(8), 1517–1522. https://doi.org/10.1007/s00254-008-1249-5
Judd, L. A., Jackson, B. E., & Fonteno, W. C. (2015). Advancements in root growth measurement technologies and observation capabilities for container-grown plants. Plants, 4, 369–392. https://doi.org/10.3390/plants4030369
Kataoka, T., Iikura, H., & Nakanishi, T. M. (1997). Aluminum distribution and viability of plant root and cultured cells. Soil Science and Plant Nutrition, 43(sup1), 1003–1007. https://doi.org/10.1080/00380768.1997.11863707
Keren, R. (2000). Salinity. In Handbook of soil science ( pp. G3–G25), CRC Press.
Klauber, C., Gräfe, M., & Power, G. (2011). Bauxite residue issues: II. options for residue utilization. Hydrometallurgy, 108(1), 11–32. https://doi.org/10.1016/j.hydromet.2011.02.007
Klebercz, O., Mayes, W. M., Anton, Á. D., Feigl, V., Jarvis, A. P., & Gruiz, K. (2012). Ecotoxicity of fluvial sediments downstream of the Ajka red mud spill Hungary. Journal of Environmental Monitoring, 14(8), 2063–2071. https://doi.org/10.1039/C2EM30155E
Kochian, L. V., Piñeros, M. A., & Hoekenga, O. A. (2005). The physiology, genetics and molecular biology of plant aluminum resistance and toxicity. Plant and Soil, 274(1–2), 175–195. https://doi.org/10.1007/s11104-004-1158-7
McLeod, M. L., Rummel, A., & Lekberg, Y. (2018). A rapid, quantitative, and affordable fluorometric method to determine the viability of roots with potential application in fungal hyphae. Rhizosphere, 6, 1–6. https://doi.org/10.1016/j.rhisph.2018.01.008
Mišík, M., Burke, I. T., Reismüller, M., Pichler, C., Rainer, B., Mišíková, K., Mayes, W. M., & Knasmueller, S. (2014). Red mud a byproduct of aluminum production contains soluble vanadium that causes genotoxic and cytotoxic effects in higher plants. Science of The Total Environment, 493, 883–890. https://doi.org/10.1016/j.scitotenv.2014.06.052
Pagano, G., Meriç, S., De Biase, A., Iaccarino, M., Petruzzelli, D., Tünay, O., & Warnau, M. (2002a). Toxicity of bauxite manufacturing by-products in sea urchin embryos. Ecotoxicology and Environmental Safety, 51, 28–34. https://doi.org/10.1006/eesa.2001.2114.
Pagano, G., de Biase, A., Iaccarino, M., Meric, S., Petruzzelli, D., Tünay, O., et al. (2002b). Bauxite manufacturing residues from Gardanne (France) and Portovesme (Italy) exert different patterns of pollution and toxicity to sea urchin embryos. Environmental Toxicology and Chemistry, 21(6), 1272–1278. https://doi.org/10.1002/etc.5620210623
Passardi, F., Cosio, C., Penel, C., & Dunand, C. (2005). Peroxidases have more functions than a Swiss army knife. Plant Cell Reports, 24(5), 255–265. https://doi.org/10.1007/s00299-005-0972-6
Patil, N. S., Apradh, V. T., & Karadge, B. A. (2012). Effects of alkali stress on seed germination and seedlings growth of Vigna aconitifolia (Jacq.) Marechal. Pharmacognosy Journal, 4(34), 77–80. https://doi.org/10.5530/pj.2012.34.13
Potters, G., Pasternak, T. P., Guisez, Y., Palme, K. J., & Jansen, M. A. K. (2007). Stress-induced morphogenic responses: growing out of trouble? Trends in Plant Science, 12(3), 98–105. https://doi.org/10.1016/j.tplants.2007.01.004
Rékási, M., Feigl, V., Uzinger, N., Gruiz, K., Makó, A., & Anton, A. (2013). Effects of leaching from alkaline red mud on soil biota: modelling the conditions after the Hungarian red mud disaster. Chemistry and Ecology, 29(8), 709–723. https://doi.org/10.1080/02757540.2013.817568.
Rubinigg, M., Wenisch, J., Elzenga, J. T. M., & Stulen, I. (2004). NaCl salinity affects lateral root development in Plantago maritima. Functional Plant Biology, 31(8), 775–780. https://doi.org/10.1071/FP03222
Ruyters, S., Mertens, J., Vassilieva, E., Dehandschutter, B., Poffijn, A., & Smolders, E. (2011). The red mud accident in Ajka (Hungary): Plant toxicity and trace metal bioavailability in red mud contaminated soil. Environmental Science & Technology, 45(4), 1616–1622. https://doi.org/10.1021/es104000m.
Ryser, P., & Emerson, P. (2007). Growth, root and leaf structure, and biomass allocation in Leucanthemum vulgare Lam. (Asteraceae) as influenced by heavy-metal-containing slag. Plant and Soil, 301(1), 315–324. https://doi.org/10.1007/s11104-007-9451-x
Santini, T. C., & Fey, M. V. (2015). Fly ash as a permeable cap for tailings management: Pedogenesis in bauxite residue tailings. Journal of Soils and Sediments, 15(3), 552–564. https://doi.org/10.1007/s11368-014-1038-6
Santini, T. C., & Fey, M. V. (2016). Assessment of Technosol formation and in situ remediation in capped alkaline tailings. CATENA, 136, 17–29. https://doi.org/10.1016/j.catena.2015.08.006
Santini, T. C., Kerr, J. L., & Warren, L. A. (2015). Microbially-driven strategies for bioremediation of bauxite residue. Journal of Hazardous Materials, 293, 131–157. https://doi.org/10.1016/j.jhazmat.2015.03.024
Shanker, A., & Shanker, C. (2016). Abiotic and biotic stress in plants: recent advances and future perspectives. BoD – Books on Demand.
Shi, B., Qu, Y., & Li, H. (2017). Gypsum alleviated hydroxyl radical-mediated oxidative damages caused by alkaline bauxite residue in leaves of Atriplex canescens. Ecological Engineering, 98, 166–171. https://doi.org/10.1016/j.ecoleng.2016.10.008
Singh, S., Srivastava, P. K., Kumar, D., Tripathi, D. K., Chauhan, D. K., & Prasad, S. M. (2015). Morpho-anatomical and biochemical adapting strategies of maize (Zea mays L.) seedlings against lead and chromium stresses. Biocatalysis and Agricultural Biotechnology, 4(3), 286–295. https://doi.org/10.1016/j.bcab.2015.03.004
Sofo, A., Scopa, A., Nuzzaci, M., & Vitti, A. (2015). Ascorbate peroxidase and catalase activities and their genetic regulation in plants subjected to drought and salinity stresses. International Journal of Molecular Sciences, 16(6), 13561–13578. https://doi.org/10.3390/ijms160613561
Sosa, L., Llanes, A., Reinoso, H., Reginato, M., & Luna, V. (2005). Osmotic and Specific Ion Effects on the Germination of Prosopis strombulifera. Annals of Botany, 96(2), 261–267. https://doi.org/10.1093/aob/mci173
Ujaczki, É., Klebercz, O., Feigl, V., Molnár, M., Magyar, Á., Uzinger, N., & Gruiz, K. (2015). Environmental toxicity assessment of the spilled Ajka red mud in soil microcosms for its potential utilisation as soil ameliorant. Periodica Polytechnica Chemical Engineering, 59(4), 253–261. https://doi.org/10.3311/ppch.7839
Venkatachalam, P., Jayaraj, M., Manikandan, R., Geetha, N., Rene, E. R., Sharma, N. C., & Sahi, S. V. (2017). Zinc oxide nanoparticles (ZnONPs) alleviate heavy metal-induced toxicity in Leucaena leucocephala seedlings: A physiochemical analysis. Plant Physiology and Biochemistry, 110, 59–69. https://doi.org/10.1016/j.plaphy.2016.08.022
Wong, J. W. C., & Ho, G. E. (1993). Use of waste gypsum in the revegetation on red mud deposits: A greenhouse study. Waste Management & Research, 11(3), 249–256. https://doi.org/10.1006/wmre.1993.1024
Wong, J. W. C., & Ho, G. (1994). Sewage sludge as organic ameliorant for revegetation of fine bauxite refining residue. Resources, Conservation and Recycling, 11(1–4), 297–309. https://doi.org/10.1016/0921-3449(94)90097-3
Zlobin, I. E., Kartashov, A. V., & Shpakovski, G. V. (2017). Different roles of glutathione in copper and zinc chelation in Brassica napus roots. Plant Physiology and Biochemistry, 118, 333–341. https://doi.org/10.1016/j.plaphy.2017.06.029
Zolla, G., Heimer, Y. M., & Barak, S. (2010). Mild salinity stimulates a stress-induced morphogenic response in Arabidopsis thaliana roots. Journal of Experimental Botany, 61(1), 211–224. https://doi.org/10.1093/jxb/erp290
Acknowledgements
We would like to acknowledge the help of Mr. Michael Paul in improving the English of this manuscript and Mr. Virgile Calvert for his technical assistance in the laboratory and in the field.
Funding
This work was co-funded by the Labex DRIIHM, French programme Investissements d’Avenir (ANR-11-LABX-0010), managed by the ANR, Observatoire Homme-Milieux Bassin Minier de Provence (OHM BMP) and by the Initiative Structurante Ecosphère Continentale et Côtière (EC2CO CNRS).
Author information
Authors and Affiliations
Contributions
CF conceived and carried out the experiment and drafted the manuscript. ML participated in the conception and the design of the study and helped in the funding research. CK helped in the methodology and analysis of the results. PH gave logistical help (for the substrates used) and in the conception of the study. JF participated in data analyses and methodology. J-PA and BA carried out the elemental analyses. SC conceived the study and participated in its design and coordination and helped to draft the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fourrier, C., Luglia, M., Keller, C. et al. How Raw and Gypsum Modified Bauxite Residues Affect Seed Germination, Enzyme Activities, and Root Development of Sinapis alba. Water Air Soil Pollut 232, 309 (2021). https://doi.org/10.1007/s11270-021-05232-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05232-x