Abstract
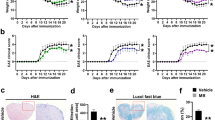
A wide range of evidence shows that Multiple sclerosis is associated with increased nociceptive and neuropathic pain affecting the quality of life in patients. There are no effective medications to relieve pain without side effects in patients with Multiple sclerosis. Hence, there is a major need to develop new effective therapies for the treatment of chronic pain in these patients. Emerging evidence suggests that β-hydroxybutyrate, the most abundant ketone body metabolite, is able to relieve pain in experimental animals. In the current study, the therapeutic effects of β-hydroxybutyrate on clinical symptoms and pain behaviors were investigated in an animal model of multiple sclerosis (experimental autoimmune encephalomyelitis; EAE). Clinical scores and body weight were recorded in EAE-induced female C57BL6 mice. To measure pain behaviors, animals were subjected to three tests including Radiant heat, Mechano-allodynia, and tail flick before and after EAE induction. The findings indicated that β-hydroxybutyrate significantly attenuated clinical scores and pain behaviors in EAE-induced mice. However, we did not any significant changes in body weight of EAE-induced mice. Overall, this study suggests that β-hydroxybutyrate could be used as a therapeutic agent for the treatment of clinical symptoms and pain behaviors in mice with EAE. Further studies are needed to validate these findings in humans.






Similar content being viewed by others
REFERENCES
Fu, W., Taylor, B.K., Neurosci. Lett., 2015, vol. 595, pp. 1–6.
Goldenberg, M.M., Pharm. Ther., 2012, vol. 37, pp. 175–184.
Foley, P.L., Vesterinen, H.M., Laird, B.J., Sena, E.S., Colvin, L.A., Chandran, S., MacLeod, M.R., Fallon, M.T., Pain, 2013, vol. 154, pp. 632–642.
Österberg, A., Boivie, J., Thuomas, K.-Å., Eur. J. Pain, 2005, vol. 9, pp. 531–542.
Truini, A., Barbanti, P., Pozzilli, C., Cruccu, G., J. Neurol., 2013, vol. 260, pp. 351–367.
Clifford, D.B., Trotter, J.L., Arch. Neurol., 1984, vol. 41, pp. 1270–1272.
Stenager, E., Knudsen, L., Jensen, K., Acta Neurol., 1991, vol. 84, pp. 197–200.
Lynch, J.L., Gallus, N.J., Ericson, M.E., Beitz, A.J., Pain, 2008, vol. 136, pp. 293–304.
Olechowski, C.J., Truong, J.J., Kerr, B.J., Pain, 2009, vol. 141, pp. 156–164.
Olechowski, C.J., Parmar, A., Miller, B., Stephan, J., Tenorio, G., Tran, K., Leighton, J., Kerr, B.J., Pain, 2010, vol. 149, pp. 565–572.
Ritter, C., Dalenogare, D.P., de Almeida, A.S., Pereira, V.L., Pereira, G.C., Fialho, M.F.P., Lückemeyer, D.D., Antoniazzi, C.T., Kudsi, S.Q., Ferreira, J., Oliveira, S.M., Trevisan, G., Mol. Neurobiol., 2020, vol. 57, pp. 2420–2435.
Goldberg, E.L., Asher, J.L., Molony, R.D., Shaw, A.C., Zeiss, C.J., Wang, C., Morozova-Roche, L.A., Herzog, R.I., Iwasaki, A., Dixit, V.D., Cell Rep., 2017, vol. 18, pp. 2077–2087.
Qian, J., Zhu, W., Lu, M., Ni, B., Yang, J., Br. J. Pharmacol., 2017, vol. 174, pp. 1961–1971.
Cahill Jr, G.F., Annu. Rev. Nutr., 2006, vol. 26, pp. 1–22.
Newman, J.C., Verdin, E., Trends Endocrinol. Metab., 2014, vol. 25, pp. 42–52.
Youm, Y.-H., Nguyen, K.Y., Grant, R.W., Goldberg, E.L., Bodogai, M., Kim, D., D’agostino, D., Planavsky, N., Lupfer, C., Kanneganti, T.D., Nat. Med., 2015, vol. 21, pp. 263–269.
Rahman, M., Muhammad, S., Khan, M.A., Chen, H., Ridder, D.A., Müller-Fielitz, H., Pokorná, B., Vollbrandt, T., Stölting, I., Nadrowitz, R., Nat. Commun., 2014, vol. 5, pp. 1–11.
Xie, G., Tian, W., Wei, T., Liu, F., Free Radic. Res., 2015, vol. 49, pp. 139–150.
Tieu, K., Perier, C., Caspersen, C., Teismann, P., Wu, D.-C., Yan, S.-D., Naini, A., Vila, M., Jackson-Lewis, V., Ramasamy, R., J. Clin. Invest., 2003, vol. 112, pp. 892–901.
Lim, S., Chesser, A.S., Grima, J.C., Rappold, P.M., Blum, D., Przedborski, S., Tieu, K., PLoS One, 2011, vol. 6, p. e24620.
Zeraati, M., Enayati, M., Kafami, L., Shahidi, S.H., Salari, A.-A., Neuropharmacology., 2019, vol. 157, p. 107685.
Mami, S., Yeganeh, F., Salari, A.-A., Anissian, A., Azizi, M., Hajimollahoseini, M., Int. Immunopharmacol., 2020, vol. 84, p. 106536.
Jahan-Abad, A.J., Karima, S., Shateri, S., Baram, S.M., Rajaei, S., Morteza-Zadeh, P., Borhani-Haghighi, M., Salari, A.-A., Nikzamir, A., Gorji, A., Neuropathology., 2020, vol. 40, pp. 84–92.
Aghaie, T., Jazayeri, M.H., Avan, A., Anissian, A., Salari, A.-A., IUBMB Life., 2019, vol. 71, pp. 1313–1321.
Kamarehei, M., Pejman, S., Ardestani, S.K., Zahednasab, H., Firouzi, M., Harirchian, M.H., Int. Immunopharmacol., 2020, vol. 82, p. 106286.
Karampoor, S., Zahednasab, H., Amini, R., Esghaei, M., Sholeh, M., Keyvani, H., Int. Immunopharmacol., 2020, vol. 80, p. 106138.
Ayatollahi, A.M., Haji Molla Hoseini, M., Ghanadian, S.M., Kosari-Nasab, M., Mami, F., Yazdiniapoure, Z., Zolfaghari, B., Salari, A.-A., Neurol. Res., 2017, vol. 39, pp. 1056–1065.
D’amour, F.E., Smith, D.L., J. Pharmacol. Exp. Ther., 1941, vol. 72, pp. 74–79.
Decosterd, I., Woolf, C.J., Pain, 2000, vol. 87, pp. 149–158.
Lutfy, K., Cai, S.X., Woodward, R.M., Weber, E., Pain, 1997, vol. 70, pp. 31–40.
Choi, I.Y., Piccio, L., Childress, P., Bollman, B., Ghosh, A., Brandhorst, S., Suarez, J., Michalsen, A., Cross, A.H., Morgan, T.E., Cell Rep., 2016, vol. 15, pp. 2136–2146.
Esquifino, A.I., Cano, P., Jimenez-Ortega, V., Fernández-Mateos, M.P., Cardinali, D.P., J. Neuroinflammation, 2007, vol. 4, p. 6.
Kafami, L., Raza, M., Razavi, A., Mirshafiey, A., Movahedian, M., Khorramizadeh, M.R., Avicenna J. Med. Biotechnol., 2010, vol. 2, pp. 47–52.
Hao, J., Liu, R., Turner, G., Shi, F.-D., Rho, J.M., PLoS One., 2012, vol. 7, pp. e35476.
Piccio, L., Stark, J.L., Cross, A.H., J. Leukoc. Biol., 2008, vol. 84, pp. 940–948.
Duffy, S.S., Perera, C.J., Makker, P.G.S., Lees, J.G., Carrive, P., Moalem-Taylor, G., Front. Immunol., 2016, vol. 7, p. 369.
Lu, J., Kurejova, M., Wirotanseng, L.N., Linker, R.A., Kuner, R., Tappe-Theodor, A., J. Neuroinflammation, 2012, vol. 9, pp. 233.
Aicher, S.A., Silverman, M.B., Winkler, C.W., Bebo, B.F., Jr., Pain., 2004, vol. 110, pp. 560–570.
Włodarek, D., Nutrients., 2019, vol. 11, pp. 169.
Lee, J.E., Titcomb, T.J., Bisht, B., Rubenstein, L.M., Louison, R., Wahls, T.L., J. Am. Coll. Nutr., 2020, pp. 1–13.
Storoni, M., Plant, G.T., Mult. Scler. Int., 2015, vol. 2015.
ACKNOWLEDGMENTS
The authors would like to thank the “Salari Institute of Cognitive and Behavioral Disorders (SICBD)” and the “Neurophysiology Research Center of Shahid University of Medical Sciences” for their valuable support.
Funding
No external funding was received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest. The authors declare that they have no conflict of interest.
Ethical approval. All experimental procedures were approved by the Research and Ethics Committee of Science and Research Branch, Islamic Azad University.
Rights and permissions
About this article
Cite this article
Vahideh Mirzaei, Eidi, A., Manaheji, H. et al. β-Hydroxybutyrate Attenuates Clinical Symptoms and Pain Behaviors in MOG-Induced Encephalomyelitis. Neurochem. J. 15, 181–186 (2021). https://doi.org/10.1134/S1819712421020100
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1819712421020100