Abstract
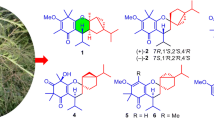
One new picrotoxane sesquiterpene (1) and one new α-pyrone derivative (4), together with nine known compounds, were isolated from the aerial parts of Dendrobium signatum. The structures of the new compounds were elucidated based on the interpretation of 1D and 2D NMR, HRESIMS and electronic circular dichroism (ECD) data. The absolute configuration of 1 was confirmed by single-crystal X-ray diffraction data. The new α-pyrone 4 exhibited promising ABTS scavenging activity with IC50 value of 0.7 μM.
Graphic abstract






Similar content being viewed by others
References
Lopo V, Patil A, Phatak A, Chandra N (2010) Free radicals, antioxidants and functional foods: impact on human health. Pharmacogn Rev 4(8):118–126
Young OI, Woodside JV (2001) Antioxidants in health and disease. J Clin Pathol 54:176–186
Cameron KM, Chase MW, Whitten WM, Kores PJ, Jarrell DC, Albert VA, Yukawa T, Hills HG, Goldman DH (1999) A phylogenetic analysis of the Orchidaceae: evidence from rbcL nucleotide. Am J Bot 86(2):208–224
Wang L, Zhang CF, Wang ZT, Zhang M, Xu LS (2009) Five new compounds from Dendrobium crystallinum. J Asian Nat Prod Res 11(11):903–911
Zhang GN, Zhong LY, Bligh SWA, Guo YL, Zhang CF, Zhang M, Wang ZT, Xu LS (2005) Bi-bicyclic and bi-tricyclic compounds from Dendrobium thyrsiflorum. Phytochemistry 66(10):1113–1120
Chanvorachote P, Kowitdamrong A, Ruanghirun T, Sritularak B, Mungmee C, Likhitwitayawuid K (2013) Cytotoxic and antimigratory activities of phenolic compounds from Dendrobium brymerianum. Nat Prod Commun 8(1):115–118
Liu XF, Zhu J, Ge SY, Xia LJ, Yang HY, Qian YT, Ren FZ (2011) Orally administered Dendrobium officinale and its polysaccharides enhance immune functions in BALB/c mice. Nat Prod Commun 6(6):867–870
Vaddhanaphuti N (2005) A field guide to the wild orchids of Thailand fourth and, expanded. Silkworm books, Chiang mai
Mittraphab A, Muangnoi C, Likhitwitayawuid K, Rojsitthisak P, Sritularak B (2016) A new bibenzyl-phenanthrene derivative from Dendrobium signatum and its cytotoxic activity. Nat Prod Commun 11(5):657–659
Okamoto T, Natsume M, Onaka T, Uchimaru F, Shimizu M (1966) The structure of dendroxine the third alkaloid from Dendrobium nobile. Chem Pharm Bull 14(6):672–675
Li Y, Wang CL, Guo SX, Yang JS, Xiao PG (2008) Two new compounds from Dendrobium candidum. Chem Pharm Bull 56(10):1477–1479
Li Y, Wang CL, Wang YJ, Wang FF, Guo SX, Yang JS, Xiao PG (2009) Four new bibenzyl derivatives from Dendrobium candidum. Chem Pharm Bull 57(9):997–999
Yang D, Cheng ZQ, Yang L, Hou B, Yang J, Li XN, Zi CT, Dong FW, Liu ZH, Zhou J, Ding ZT, Hu JM (2018) Seco-dendrobine-type alkaloids and bioactive phenolics from Dendrobium findlayanum. J Nat Prod 81(2):227–235
Kaewamatawong R, Ruangrungsi N, Likhitwitayawuid KJ (2007) Chemical constituents of Polyalthia parviflora. J Nat Med 61(3):349–350
Bi ZM, Wang ZT, Xu LS (2004) Chemical Constituents of Dendrobium moniliforme. Acta Bot Sin 46(1):124–126
Reyes-Ramirez A, Leyte-Lugo M, Figueroa M, Serrano-Alba T, Gonzalez-Andrade M, Mata R (2011) Synthesis, biological evaluation, and docking studies of gigantol analogs as calmodulin inhibitors. Eur J Med Chem 46(7):2699–2708
Katerere DR, Gray AI, Nash RJ, Waigh RD (2012) Phytochemical and antimicrobial investigations of stilbenoids and flavonoids isolated from three species of Combretaceae. Fitoterapia 83(5):932–940
Zhao WM, Ye QH, Dai JQ, Martin MT, Zhu JP (2003) Allo-aromadendrane- and picrotoxane-type sesquiterpenes from Dendrobium moniliforme. Planta Med 69(12):1136–1140
Wang P, Chen X, Wang H, Huang SZ, Cai CH, Yuan JZ, Zhu GL, Xu XL, Mei WL, Dai HF (2019) Four new picrotoxane-type sesquiterpenes from Dendrobium nobile Lindl. Front Chem 7:812
Punya J, Tachaleat A, Wattanachaisaereekul S, Haritakun R, Boonlarppradab C, Cheevadhanarak S (2013) Functional expression of a foreign gene in Aspergillus oryzae producing new pyrone compounds. Fungal Genet Biol 50(1):55–62
Snatzke G (1968) Circular dichroism and optical rotatory dispersion–principles and application to the investigation of the stereochemistry of natural products. Angew Chem Int Ed 7(1):14–25
Elvidge JA, Ralph PD (1966) Polyene acids. Part X. The conformation of hexenolactone and the configuration of the derived sorbic acid as indicated by proton magnetic resonance spectroscopy. J Chem Soc B:243–244
Mori K (1976) Absolute configuration of (−)-massoilactone as confirmed by a synthesis of its (S)-(+)-isomer. Agric Biol Chem 40(8):1617–1619
Sarigaputi C, Sommit D, Teerawatananond T, Pudhom K (2014) Weakly anti-inflammatory limonoids from the seeds of Xylocarpus rumphii. J Nat Prod 77(9):2037–2043
Lu Y, Shipton F, Khoo TJ, Wiart C (2014) Antioxidant activity determination of citronellal and crude extracts of Cymbopogon citratus by 3 different methods. Pharmacol Pharm 05:395–400
Erel O (2004) A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin Biochem 37(4):277–285
Acknowledgements
This work was supported by The 90th Anniversary of Chulalongkorn University Fund (Ratchadaphiseksomphot Endownment Fund), the Research Assistantship Fund, Faculty of Science, Chulalongkorn University, and the Center of Excellence on Petrochemical and Materials Technology. T.A. was grateful to the Ratchadapisek Sompoch Endowment Fund (CU-GR_63_161_23_27), Chulalongkorn University for the partial support of X-ray diffraction facility.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khumploy, P., Raksat, A., Choodej, S. et al. Picrotoxane sesquiterpene and α-pyrone derivative from Dendrobium signatum and their free radical scavenging potency. J Nat Med 75, 967–974 (2021). https://doi.org/10.1007/s11418-021-01547-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-021-01547-5