Abstract
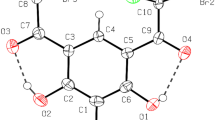
The spectroscopic characterization (1H, 13C{1H} NMR, UV–Vis) and single-crystal X-ray diffraction (scXRD) analysis accomplished by inspection of the Hirshfeld surface of bis(3-thienoyl) disulfide (1) is described. The title compound 1 crystallizes in the monoclinic space group P21/n. The unit cell parameters are a = 7.9959(3) Å, b = 6.4348(3) Å, c = 22.4924(9) Å, β = 100.108(4)°, V = 1139.32(8) Å3, Z = 4, Rgt(F) = 0.0278, wRref(F2) = 0.0667. The packing of 1 is dominated by S⋯O and S⋯S interactions, giving a 2D layer structure parallel to (101). The X‐ray crystal structure analysis revealed the packing of 1 is dominated by S⋯O and S⋯S interactions, giving a 2D layer structure parallel to (101). The intermolecular interactions in 1 were analyzed using the Hirshfeld surface method including 2D fingerprint plots and enrichment ratios (E), which shows that the most favored intermolecular contacts are the O⋯H and C⋯S indicated by E values above 1.30. The interaction energies between molecular pairs revealed the importance of the weak O⋯H and C⋯S interactions in stabilizing the molecular structure of 1.
Graphic Abstract
Single crystal X-ray structure analysis, DFT calculations and Hirshfeld surface analysis to identify intermolecular interactions within the solid state structure of bis(3-thienoyl) disulfide (1).







Similar content being viewed by others
References
Bagi N, Kaizer J, Speier G (2015) Oxidation of thiols to disulfides by dioxygen catalyzed by a bioinspired organocatalyst. RSC Adv 5:45983–45986
Lobo V, Patil A, Phatak A, Chandra N (2010) Free radicals, antioxidants and functional foods: impact on human health. Pharmacogn Rev 4:118–126
Regino CAS, Richardson DE (2007) Bicarbonate-catalyzed hydrogen peroxide oxidation of cysteine and related thiols. Inorg Chim Acta 360:3971–3977
Sanz R, Aguado R, Pedrosa MR, Arnáiz FJ (2002) Simple and selective oxidation of thiols to disulfides with dimethylsulfoxide catalyzed by dichlorodioxomolybdenum(VI). Synthesis (Stuttg) 7:856–858
Silveira CC, Mendes SR (2007) Catalytic oxidation of thiols to disulfides using iodine and CeCl3·7H2O in graphite. Tetrahedron Lett 48:7469–7471
Hekmatshoar R, Sajadi S, Heravi MM, Bamoharram FF (2007) H14[NaP5W30O110] as a heterogeneous recyclable catalyst for the air oxidation of thiols under solvent free conditions. Molecules 12:2223–2228
Supale A, Gokavi G (2008) Oxidation of thiols to disulfides using H2O2 catalyzed by recyclable chromic potassium sulphate at room temperature. React Kinet Catal Lett 93:141–148
Leite SLS, Pardini VL, Viertler H (1990) Synthetic communications: an international journal for rapid communication of synthetic organic chemistry. Synth Commun 20:393–397
Salimi A, Hallaj R (2005) Catalytic oxidation of thiols at preheated glassy carbon electrode modified with abrasive immobilization of multiwall carbon nanotubes: applications to amperometric detection of thiocytosine, l-cysteine and glutathione. Talanta 66:967–975
Sridhar M, Kumara Vadivel S, Bhalerao UT (1998) Novel method for preparation of symmetric disulfides from thiols using enzyme catalysis. Synth Commun 28:1499–1502
Khalyfeh KAL, Taher D, Helal W et al (2020) Synthesis and characterization of 1,4-chalcogenesters bearing 5-membered heterocycles. J Chem Sci 132(117):1–16
Patai S, Rappoport Z (2006) The chemistry of peroxides, 1st edn. Wiley, New York
Niyomura O, Kato S, Inagaki S (2000) An unusual planar diacyl ditelluride (2-MeOC6H4COTe)2: the origin of its planarity. J Am Chem Soc 122:2132–2133
Niyomura O, Tani K, Kato S (1999) A facile synthesis of potassium selenocarboxylates and their oxidation with XeF2 to diacyl diselenides. Heteroat Chem 10:373–379
Chou JH, Rauchfuss TB, Szczepura LF (1998) Studies on the electroactive heterocycles C6S8(0/2-) and C6S6O2(0/2-) and related metal complexes. J Am Chem Soc 120:1805–1811
Rout GC, Seshasayee M, Subrahmanyan T, Aravamudan G (1983) Structure of dibenzoyldisulphane, C14H10O2S2. Acta Crystallogr C 39:1387–1389
Paul C, Srikrishnan T (2004) Crystal and molecular structure of dibenzoyl disulfide. J Chem Crystallogr 34:211–217
Singh S, Chaturvedi J, Bhattacharya S, Nöth H (2011) Silver(I) catalyzed oxidation of thiocarboxylic acids into the corresponding disulfides and synthesis of some new Ag(I) complexes of thiophene-2-thiocarboxylate. Polyhedron 30:93–97
Vandavasi JK, Hu WP, Chen CY, Wang JJ (2011) Efficient synthesis of unsymmetrical disulfides. Tetrahedron 67:8895–8901
Wang D, Wu SY, Li HP, Yang Y, Roesky HW (2017) Synthesis and characterization of copper complexes with the N-(2,6-diisopropylphenyl)-N′-acylthiourea ligands. Eur J Inorg Chem 2017:1406–1413
Niyomura O, Kitoh Y, Nagayama K-I, Kato S (1999) Di (acyl) and di (thioacyl) disulfides: a facile synthesis and structural analysis. Sulfur Lett 22:195–207
Bereman RD, Baird DM, Bordner J, Dorfman JR (1983) Resonance induced properties in monothiocarbamates derived from aromatic amines: comparison of the coordination chemistry of indole and indoline monothiocarbamates. Ployhedron 2:25–30
Beyer L, Richter R, Seidelmann O (1999) Ferrocensubstituierte 1,3-bidentate Liganden und ihre heteronuklearen Übergangsmetallchelate. J für Prakt Chemie 341:704–726
Imrie C, Cook L, Levendis DC (2001) An investigation of the chemistry of ferrocenoyl derivatives. The synthesis and reactions of ferrocenoyl imidazolide and its derivatives. J Organomet Chem 637–639:266–275
Wang Q, Shu Y, Hou X (2011) (Furan-2-yl)[(furan-2-yl)carbonyldisulfanyl]methanone. Acta Crystallogr. https://doi.org/10.1107/S160053681104356X
Mukhlall JA, Noll BC, Hersh WH (2011) Synthesis of chiral disulfides: potential reagents for enantioselective sulfurization. J Sulfur Chem 32:199–212
Erben MF, Della Védova CO, Willner H, Trautner F, Oberhammer H, Boese R (2005) Fluoroformyl trifluoroacetyl disulfide, FC(O)SSC(O)CF 3: synthesis, structure in solid and gaseous states, and conformational properties. Inorg Chem 44:7070–7077
Erben MF, Della Védova CO, Willner H, Boese R (2006) Synthesis, structure and conformational properties of fluoroformylchlorodifluoroacetyl disulfide, FC(O)SSC(O)CF2Cl: conformational transferability in -C(O)SSC(O)- compounds. Eur J Inorg Chem 2006:4418–4425
Li F, Yin H-D, Hong M, Zhai J, Wang D-Q (2006) Bis(N,N-dicyclohexylthiocarbamoyl) disulfide. Acta Crystallogr Sect E E62:1417–1418
Zysman-Colman E, Harpp DN (2004) Comparison of the structural properties of compounds containing the XSSX moiety (X = H, Me, R, Cl, Br, F, OR). J Sulfur Chem 25:291–316
Fulmer GR, Miller AJM, Sherden NH, Gottlieb HE, Nudelman A, Stoltz BM, Bercaw JE, Goldberg KI (2010) NMR chemical shifts of trace impurities: common laboratory solvents, organics, and gases in deuterated solvents relevant to the organometallic chemist. Organometallics 29:2176–2179
Sheldrick GM (1990) Phase annealing in SHELX-90: direct methods for larger structures. Acta Crystallogr Sect A A46:467–473
Zhu Y, Wolf MO (2000) Charge transfer and delocalization in conjugated (ferrocenylethynyl)oligothiophene complexes. J Am Chem Soc 122:10121–10125
Sheldrick GM (2008) A short history of SHELX. Acta Crystallogr Sect A A64:112–122
Farrugia LJ (2012) WinGX and ORTEP for windows: an update. J Appl Crystallogr 45:849–854
Neese F (2012) The ORCA program system. WIREs Comput Mol Sci 2:73–78
Neese F (2017) Software update: the ORCA program system, version 4.0. WIREs Comput Mol Sci e1327:1–6
Grimme S (2006) Semiempirical hybrid density functional with perturbative second-order correlation. J Chem Phys. https://doi.org/10.1063/1.2148954
Weigend F, Ahlrichs R (2005) Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys Chem Chem Phys 7:3297–3305
Barone V, Cossi M (1998) Quantum calculation of molecular energies and energy gradients in solution by a conductor solvent mode. J Phys Chem A 102:1995–2001
Bauernschmitt R, Ahlrichs R (1996) Treatment of electronic excitations within the adiabatic approximation of time dependent density functional theory. Chem Phys Lett 256:454–464
Allouche A (2011) Software news and updates Gabedit—a graphical user interface for computational chemistry softwares. J Comput Chem 32:174–182
Turner MJ, McKinnon JJ, Wolff SK, Grimwood DJ, Spackman PR, Jayatilaka D, Spackman MA (2017) CrystalExplorer17. University of Western Australia
Pauling L (1960) The nature of the chemical bond by Linus Pauling, 3rd edn. Cornell University Press, Ithaca, NY
Mayer I (1983) Charge, bond order and valence in the AB initio SCF theory. Chem Phys Lett 97:270–274
Mayer I (1984) Bond order and valence: relations to Mulliken’s population analysis. Int J Quantum Chem 26:151–154
Mayer I (1985) Bond orders and valences in the SCF theory: a comment. Theor Chim Acta 67:315–322
Van Wart HE, Shipman LL, Scheraga HA (1975) Theoretical and experimental evidence for a nonbonded 1,4 carbon-sulfur interaction in organosulfur compounds. J Phys Chem 79:1436–1447
Poppitz EA, Hildebrandt A, Korb M, Schaarschmidt D, Lang H (2014) Synthesis, properties, and electron transfer studies of ferrocenyl thiophenes. Zeitschrift fur Anorg und Allg Chemie 640:2809–2816
Poppitz EA, Korb M, Lang H (2014) Crystal structure of 3-{1’-[3,5-bis(trifluoromethyl)phenyl]ferrocenyl}-4-bromothiophene. Acta Crystallogr E70:238–241
Taher D, Awwadi FF, Speck JM et al (2017) Heterocyclic-based ferrocenyl carboselenolates: synthesis, solid-state structure and electrochemical investigations. J Organomet Chem 845:55–62
Rowland RS, Taylor R (1996) Intermolecular nonbonded contact distances in organic crystal structures: comparison with distances expected from van der Waals Radii. J Phys Chem 100:7384–7391
Alvarez S (2013) A cartography of the van der Waals territories. Dalton Trans 42:8617–8636
Hirshfeld FL (1977) Bonded-atom fragments for describing molecular charge densities. Theor Chim Acta (Berl) 44:129–138
McKinnon JJ, Spackman MA, Mitchell AS (2004) Novel tools for visualizing and exploring intermolecular interactions in molecular crystals. Acta Crystallogr B 60:627–668
Spackman MA, McKinnon JJ (2002) Fingerprinting intermolecular interactions in molecular crystals. CrystEngComm 4:378–392
Liu X, Du L, Zhang W, Li R, Feng F, Feng X (2019) Syntheses, structures and Hirshfeld surface analyses of two 3D supramolecules based on nitrogen-heterocyclic tricarboxylate ligand. J Mol Struct 1194:138–143
Jelsch C, Ejsmont K, Huder L (2014) The enrichment ratio of atomic contacts in crystals, an indicator derived from the Hirshfeld surface analysis. IUCrJ 1:119
Tan SL, Jotani MM, Tiekink ERT (2019) Utilizing Hirshfeld surface calculations, non-covalent interaction (NCI) plots and the calculation of interaction energies in the analysis of molecular packing. Acta Crystallogr E 75:308
Acknowledgements
We are grateful to the Deutsche Forschungsgemeinschaft (DFG) for a visiting fellowship of Al Khalyfeh, Kahled.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Al Khalyfeh, K., Taher, D., Helal, W. et al. Crystal Structure and Hirshfeld Surface Analysis of Bis(3-thienoyl) Disulfide. J Chem Crystallogr 52, 113–121 (2022). https://doi.org/10.1007/s10870-021-00896-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-021-00896-z