Abstract
Background/purpose of the study
Laparoscopic hepatectomy (LH) has been widely used in the treatment of hepatocellular carcinoma (HCC). It is generally believed that the long-term outcomes of LH are not inferior to open hepatectomy (OH). However, the quality of evidence is low. The purpose of this study was to reconstruct time-to-event data for meta-analysis based on Kaplan–Meier curves from propensity-score matched studies and compare survival rates following LH and OH for hepatocellular carcinoma.
Methods
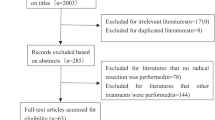
All published propensity-score matched studies reported in English that compared LH and OH for hepatocellular carcinoma with Kaplan–Meier curves were screened. Patients’ survival information was reconstructed with the aid of a computer vision program. Different models (fixed-effects model for two-stage survival analysis and Cox regression for one-stage survival analysis) were performed for sensitivity analysis. In addition to the primary meta-analysis, two specific subgroup analyses were performed on patients by types of resection, cirrhosis status.
Results
Time-to-event data were extracted from 45 propensity-score matched studies (N = 8905). According to the time-to-event data and the reconstructed Kaplan–Meier curves, the cumulative overall survival rate was 49.0% and 50.9% in the LH and OH cohorts, respectively, a log-rank test did not demonstrate statistical significance (p > 0.05). The cumulative recurrence-free survival (RFS) probability was both close to 0.0%. The median RFS time was 49.1 (95% CI 46.1 ~ 51.7) and 44.3 (95% CI 41 ~ 46.1) months. The difference in disease status was statistically significant by the Log-rank test (p < 0.05). Using the random-effects model of two-stage analysis, the minor hepatectomy subgroup (HR = 1.32, 95% CI [1.09, 1.55], I2 = 6.2%, p = 0.383) and the shared fragile model of one-stage analysis (HR = 1.44 95% CI [1.23, 1.69], p < 0.001) suggested that LH could significantly prolong RFS of patients compared with OH. This result was consistent with sensitivity analysis using different models.
Conclusion
This study was the first reconstructed time-to-event data based on a high-quality propensity-score matching study to compare the survival outcomes of LH and OH in the treatment of HCC. Results suggested that LH can improve RFS in patients with HCC undergoing minor hepatectomy and may also benefit long-term RFS in overall patients.











Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Mak LY, Cruz-Ramon V, Chinchilla-Lopez P, et al. Global epidemiology, prevention, and management of hepatocellular carcinoma. Am Soc Clin Oncol Educ Book. 2018;38:262–79.
Yang JD, Hainaut P, Gores GJ, et al. A global view of hepatocellular carcinoma: trends, risk, prevention and management. Nat Rev Gastroenterol Hepatol. 2019;16(10):589–604.
Lin S, Hoffmann K, Schemmer P. Treatment of hepatocellular carcinoma: a systematic review. Liver Cancer. 2012;1(3–4):144–58.
Gomaa AI, Waked I. Recent advances in multidisciplinary management of hepatocellular carcinoma. World J Hepatol. 2015;7(4):673–87.
Buell JF, Cherqui D, Geller DA, et al. The international position on laparoscopic liver surgery: the Louisville Statement, 2008. Ann Surg. 2009;250(5):825–30.
Morise Z, Wakabayashi G. First quarter century of laparoscopic liver resection. World J Gastroenterol. 2017;23(20):3581–8.
Nguyen KT, Gamblin TC, Geller DA. World review of laparoscopic liver resection-2,804 patients. Ann Surg. 2009;250(5):831–41.
Morise Z. Developments and perspectives of laparoscopic liver resection in the treatment of hepatocellular carcinoma. Surg Today. 2019;49(8):649–55.
Ciria R, Cherqui D, Geller DA, et al. Comparative short-term benefits of laparoscopic liver resection: 9000 cases and climbing. Ann Surg. 2016;263(4):761–77.
Xiangfei M, Yinzhe X, Yingwei P, et al. Open versus laparoscopic hepatic resection for hepatocellular carcinoma: a systematic review and meta-analysis. Surg Endosc. 2019;33(8):2396–418.
Xing L, Guo HB, Kan JL, et al. Clinical outcome of open surgery versus laparoscopic surgery for cirrhotic hepatocellular carcinoma patients: a meta-analysis. Eur J Gastroenterol Hepatol. 2020;32(2):239–45.
Goh EL, Chidambaram S, Ma S. Laparoscopic vs open hepatectomy for hepatocellular carcinoma in patients with cirrhosis: a meta-analysis of the long-term survival outcomes. Int J Surg. 2018;50:35–42.
Lim C, Osseis M, Lahat E, et al. Safety of laparoscopic hepatectomy in patients with hepatocellular carcinoma and portal hypertension: interim analysis of an open prospective study. Surg Endosc. 2019;33(3):811–20.
Wakabayashi G, Cherqui D, Geller DA, et al. Recommendations for laparoscopic liver resection: a report from the second international consensus conference held in Morioka. Ann Surg. 2015;261(4):619–29.
Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399–424.
Lonjon G, Boutron I, Trinquart L, et al. Comparison of treatment effect estimates from prospective nonrandomized studies with propensity score analysis and randomized controlled trials of surgical procedures. Ann Surg. 2014;259(1):18–25.
Guyot P, Ades AE, Ouwens MJ, et al. Enhanced secondary analysis of survival data: reconstructing the data from published Kaplan-Meier survival curves. BMC Med Res Methodol. 2012;12:9.
Saluja R, Cheng S, Delos Santos KA, et al. Estimating hazard ratios from published Kaplan-Meier survival curves: a methods validation study. Res Synth Methods. 2019;10(3):465–75.
Stroup DF, Berlin JA, Morton SC, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283(15):2008–12.
Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol. 2009;62(10):1006–12.
Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008.
Tierney JF, Stewart LA, Ghersi D, et al. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007;8:16.
Wei Y, Royston P. Reconstructing time-to-event data from published Kaplan-Meier curves. Stata J. 2017;17(4):786–802.
GA Wells BSDOC. The Newcastle-Ottawa Scale (NOS) for assessing the quality if nonrandomized studies in meta-analyses.
Ahn KS, Kang KJ, Kim YH, et al. A propensity score-matched case-control comparative study of laparoscopic and open liver resection for hepatocellular carcinoma. J Laparoendosc Adv Surg Tech A. 2014;24(12):872–7.
Andreou A, Struecker B, Raschzok N, et al. Minimal-invasive versus open hepatectomy for hepatocellular carcinoma: comparison of postoperative outcomes and long-term survivals using propensity score matching analysis. Surg Oncol. 2018;27(4):751–8.
Badawy A, Seo S, Toda R, et al. A propensity score-based analysis of laparoscopic liver resection for liver malignancies in elderly patients. J Invest Surg. 2019;32(1):75–82.
Chen K, Pan Y, Wang YF, et al. Laparoscopic right hepatectomy for hepatocellular carcinoma: a propensity score matching analysis of outcomes compared with conventional open surgery. J Laparoendosc Adv Surg Tech A. 2019;29(4):503–12.
Cheung TT, Dai WC, Tsang SH, et al. Pure laparoscopic hepatectomy versus open hepatectomy for hepatocellular carcinoma in 110 patients with liver cirrhosis: a propensity analysis at a single center. Ann Surg. 2016;264(4):612–20.
Deng ZC, Jiang WZ, Tang XD, et al. Laparoscopic hepatectomy versus open hepatectomy for hepatocellular carcinoma in 157 patients: a case controlled study with propensity score matching at two Chinese centres. Int J Surg. 2018;56:203–7.
Di Sandro S, Bagnardi V, Najjar M, et al. Minor laparoscopic liver resection for hepatocellular carcinoma is safer than minor open resection, especially for less compensated cirrhotic patients: Propensity score analysis. Surg Oncol. 2018;27(4):722–9.
Fu XT, Tang Z, Shi YH, et al. Laparoscopic versus open left lateral segmentectomy for large hepatocellular carcinoma: a propensity score-matched analysis. Surg Laparosc Endosc Percutan Tech. 2019;29(6):513–9.
Han HS, Shehta A, Ahn S, et al. Laparoscopic versus open liver resection for hepatocellular carcinoma: Case-matched study with propensity score matching. J Hepatol. 2015;63(3):643–50.
Inoue Y, Suzuki Y, Ota M, et al. Short- and long-term results of laparoscopic parenchyma-sparing hepatectomy for small-sized hepatocellular carcinoma: a comparative study using propensity score matching analysis. Am Surg. 2018;84(2):230–7.
Iwata T, Murotani K, Komatsu S, et al. Surgical outcome of laparoscopic hepatic resection for hepatocellular carcinoma: a matched case-control study with propensity score matching. J Minim Access Surg. 2018;14(4):277–84.
Jiang X, Liu L, Zhang Q, et al. Laparoscopic versus open hepatectomy for hepatocellular carcinoma: long-term outcomes. J BUON. 2016;21(1):135–41.
Kim H, Suh KS, Lee KW, et al. Long-term outcome of laparoscopic versus open liver resection for hepatocellular carcinoma: a case-controlled study with propensity score matching. Surg Endosc. 2014;28(3):950–60.
Kim JM, Kwon CHD, Yoo H, et al. Which approach is preferred in left hepatocellular carcinoma? Laparoscopic versus open hepatectomy using propensity score matching. BMC Cancer. 2018;18(1):668.
Kim WJ, Kim KH, Kim SH, et al. Laparoscopic versus open liver resection for centrally located hepatocellular carcinoma in patients with cirrhosis: a propensity score-matching analysis. Surg Laparosc Endosc Percutan Tech. 2018;28(6):394–400.
Li W, Han J, Xie G, et al. Laparoscopic versus open mesohepatectomy for patients with centrally located hepatocellular carcinoma: a propensity score matched analysis. Surg Endosc. 2019;33(9):2916–26.
Liu F, Xu H, Li Q, et al. Outcomes of pure laparoscopic Glissonian pedicle approach hepatectomy for hepatocellular carcinoma: a propensity score matching analysis. Surg Endosc. 2019;33(4):1155–66.
Meguro M, Mizuguchi T, Kawamoto M, et al. Clinical comparison of laparoscopic and open liver resection after propensity matching selection. Surgery. 2015;158(3):573–87.
Morise Z, Aldrighetti L, Belli G, et al. Laparoscopic repeat liver resection for hepatocellular carcinoma: a multicentre propensity score-based study. Br J Surg. 2020;107(7):889–95.
Noda T, Eguchi H, Iwagami Y, et al. Minimally invasive liver resection for hepatocellular carcinoma of patients with liver damage B: a propensity score-based analysis. Hepatol Res. 2018;48(7):539–48.
Peng Y, Liu F, Xu H, et al. Outcomes of laparoscopic liver resection for patients with multiple hepatocellular carcinomas meeting the milan criteria: a propensity score-matched analysis. J Laparoendosc Adv Surg Tech A. 2019;29(9):1144–51.
Rhu J, Kim SJ, Choi GS, et al. Laparoscopic versus open right posterior sectionectomy for hepatocellular carcinoma in a high-volume center: a propensity score matched analysis. World J Surg. 2018;42(9):2930–7.
Sposito C, Battiston C, Facciorusso A, et al. Propensity score analysis of outcomes following laparoscopic or open liver resection for hepatocellular carcinoma. Br J Surg. 2016;103(7):871–80.
Takahara T, Wakabayashi G, Beppu T, et al. Long-term and perioperative outcomes of laparoscopic versus open liver resection for hepatocellular carcinoma with propensity score matching: a multi-institutional Japanese study. J Hepatobiliary Pancreat Sci. 2015;22(10):721–7.
Tanaka S, Takemura S, Shinkawa H, et al. Outcomes of Pure Laparoscopic versus Open Hepatic Resection for Hepatocellular Carcinoma in Cirrhotic Patients: A Case-Control Study with Propensity Score Matching. Eur Surg Res. 2015;55(4):291–301.
Untereiner X, Cagniet A, Memeo R, et al. Laparoscopic hepatectomy versus open hepatectomy for the management of hepatocellular carcinoma: a comparative study using a propensity score matching. World J Surg. 2019;43(2):615–25.
Vega EA, Kutlu OC, Joechle K, et al. Preoperative prognosticators of safe laparoscopic hepatocellular carcinoma resection in advanced cirrhosis: a propensity score matching population-based analysis of 1799 western patients. J Gastrointest Surg. 2019;23(6):1157–65.
Wang C, Luo W, Fu Z, et al. A propensity score-matched case-control comparative study of laparoscopic and open liver resection for hepatocellular carcinoma. J BUON. 2017;22(4):936–41.
Wu L, Tsilimigras DI, Merath K, et al. Minimally invasive liver resection for early-stage hepatocellular carcinoma: inconsistent outcomes from matched or weighted cohorts. J Gastrointest Surg. 2020;24(3):560–8.
Wu X, Huang Z, Lau WY, et al. Perioperative and long-term outcomes of laparoscopic versus open liver resection for hepatocellular carcinoma with well-preserved liver function and cirrhotic background: a propensity score matching study. Surg Endosc. 2019;33(1):206–15.
Xu HW, Li HY, Liu F, et al. Laparoscopic versus open liver resection for lesions adjacent to major vessels: a propensity score matched analysis. J Laparoendosc Adv Surg Tech A. 2017;27(10):1002–8.
Xu HW, Liu F, Li HY, et al. Outcomes following laparoscopic versus open major hepatectomy for hepatocellular carcinoma in patients with cirrhosis: a propensity score-matched analysis. Surg Endosc. 2018;32(2):712–9.
Yamamoto M, Kobayashi T, Oshita A, et al. Laparoscopic versus open limited liver resection for hepatocellular carcinoma with liver cirrhosis: a propensity score matching study with the Hiroshima Surgical study group of Clinical Oncology (HiSCO). Surg Endosc. 2020;34(11):5055–61.
Yoon SY, Kim KH, Jung DH, et al. Oncological and surgical results of laparoscopic versus open liver resection for HCC less than 5 cm: case-matched analysis. Surg Endosc. 2015;29(9):2628–34.
Yoon YI, Kim KH, Cho HD, et al. Long-term perioperative outcomes of pure laparoscopic liver resection versus open liver resection for hepatocellular carcinoma: a retrospective study. Surg Endosc. 2020;34(2):796–805.
Navarro JG, Kang I, Rho SY, et al. Major laparoscopic versus open resection for hepatocellular carcinoma: a propensity score-matched analysis based on surgeons’ learning curve. Ann Surg Oncol. 2021;28(1):447–58.
Troisi RI, Berardi G, Morise Z, et al. Laparoscopic and open liver resection for hepatocellular carcinoma with Child-Pugh B cirrhosis: multicentre propensity score-matched study. Br J Surg. 2021;108(2):196–204.
Lee DH, Kim D, Park YH, et al. Long-term surgical outcomes in patients with hepatocellular carcinoma undergoing laparoscopic vs. open liver resection: a retrospective and propensity score-matched study. Asian J Surg. 2021;44(1):206–12.
Ho KM, Cheng KC, Chan FK, et al. Laparoscopic hepatectomy versus open hepatectomy for hepatocellular carcinoma: a propensity case-matched analysis of the long-term survival. Ann Hepatobiliary Pancreat Surg. 2021;25(1):1–7.
Cheung TT, Ma KW, She WH, et al. Pure laparoscopic versus open major hepatectomy for hepatocellular carcinoma with liver F4 cirrhosis without routine Pringle maneuver—a propensity analysis in a single center. Surg Oncol. 2020;35:315–20.
Chen Y, Yu L, Quan C. Laparoscopic versus open hepatectomy for elderly patients with hepatocellular carcinoma. J BUON. 2020;25(3):1404–12.
Dumronggittigule W, Han HS, Ahn S, et al. Laparoscopic versus open hepatectomy for hepatocellular carcinoma in elderly patients: a single-institutional propensity score matching comparison. Dig Surg. 2020;37(6):495–504.
Ruzzenente A, Bagante F, Ratti F, et al. Minimally invasive versus open liver resection for hepatocellular carcinoma in the setting of portal vein hypertension: results of an international multi-institutional analysis. Ann Surg Oncol. 2020;27(9):3360–71.
Delvecchio A, Conticchio M, Ratti F, et al. Laparoscopic major hepatectomy for hepatocellular carcinoma in elderly patients: a multicentric propensity scorebased analysis. Surg Endosc. 2020. https://doi.org/10.1007/s00464-020-07843-7;
Yi YW, Zhou C. Laparoscopic versus open left hemihepatectomy for hepatocellular carcinoma: A propensity score matching analysis. Translational Cancer Research. 2020;9(9):5484–92.
Riley RD, Lambert PC, Abo-Zaid G. Meta-analysis of individual participant data: rationale, conduct, and reporting. BMJ. 2010;340:c221.
de Jong VMT, Moons KGM, Riley RD, et al. Individual participant data meta-analysis of intervention studies with time-to-event outcomes: a review of the methodology and an applied example. Res Synth Methods. 2020;11(2):148–68.
Ventresca M, Schunemann HJ, Macbeth F, et al. Obtaining and managing data sets for individual participant data meta-analysis: scoping review and practical guide. BMC Med Res Methodol. 2020;20(1):113.
Nevitt SJ, Marson AG, Davie B, et al. Exploring changes over time and characteristics associated with data retrieval across individual participant data meta-analyses: systematic review. BMJ. 2017;357:j1390.
Michiels S, Piedbois P, Burdett S, et al. Meta-analysis when only the median survival times are known: a comparison with individual patient data results. Int J Technol Assess Health Care. 2005;21(1):119–25.
Fretland AA, Sokolov A, Postriganova N, et al. Inflammatory response after laparoscopic versus open resection of colorectal liver metastases: data from the Oslo-CoMet trial. Medicine (Baltimore). 2015;94(42):e1786.
Seth R, Tai LH, Falls T, et al. Surgical stress promotes the development of cancer metastases by a coagulation-dependent mechanism involving natural killer cells in a murine model. Ann Surg. 2013;258(1):158–68.
Ciria R, Gomez-Luque I, Ocana S, et al. A systematic review and meta-analysis comparing the short- and long-term outcomes for laparoscopic and open liver resections for hepatocellular carcinoma: updated results from the European guidelines meeting on laparoscopic liver surgery, Southampton, UK, 2017. Ann Surg Oncol. 2019;26(1):252–63.
Stewart LA, Clarke M, Rovers M, et al. Preferred reporting items for systematic review and meta-analyses of individual participant data: the PRISMA-IPD statement. JAMA. 2015;313(16):1657–65.
Kulik L, El-Serag HB. Epidemiology and Management of Hepatocellular Carcinoma. Gastroenterology. 2019;156(2):477–91.
Bruix J, Takayama T, Mazzaferro V, et al. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): a phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2015;16(13):1344–54.
Tabrizian P, Jibara G, Shrager B, et al. Recurrence of hepatocellular cancer after resection: patterns, treatments, and prognosis. Ann Surg. 2015;261(5):947–55.
Dai WC, Cheung TT. Strategic overview on the best treatment option for intrahepaitc hepatocellular carcinoma recurrence. Expert Rev Anticancer Ther. 2016;16(10):1063–72.
FDA. Clinical Trial Endpoints for the Approval of Cancer Drugs and Biologics: Guidance for Industry. 2018.
Kudo A, Tanaka S, Ban D, et al. Anatomic resection reduces the recurrence of solitary hepatocellular carcinoma </=5 cm without macrovascular invasion. Am J Surg. 2014;207(6):863–9.
Xu H, Liu F, Hao X, et al. Laparoscopically anatomical versus non-anatomical liver resection for large hepatocellular carcinoma. HPB (Oxford). 2020;22(1):136–43.
Garancini M, Gianotti L, Delitala A, et al. Intraoperative ultrasound: a review on its role in liver surgery for primitive and metastatic tumors. Minerva Chir. 2016;71(3):201–13.
Ishizawa T, Saiura A, Kokudo N. Clinical application of indocyanine green-fluorescence imaging during hepatectomy. Hepatobiliary Surg Nutr. 2016;5(4):322–8.
Xu Y, Chen M, Meng X, et al. Laparoscopic anatomical liver resection guided by real-time indocyanine green fluorescence imaging: experience and lessons learned from the initial series in a single center. Surg Endosc. 2020;34(10):4683–91.
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
QS designed the study, performed searches, extracted data, assessed data quality and wrote the manuscript; XDZ performed statistical analyses and reviewed the manuscript; XYG performed searches, extracted data, assessed data quality and reviewed the manuscript; ZPH assisted with statistical analyses, and reviewed the manuscript; QZ and WMH developed the models, performed statistical analyses and reviewed the manuscript; XJC, ZMH and YJC supervised the study design, analysis and interpretation, and reviewed and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Qiang Sun, Xiangda Zhang, Xueyi Gong, Zhipeng Hu, Qiao Zhang, Weiming He, Xiaojian Chang, and Zemin Huand Yajin Chen declare that there is no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sun, Q., Zhang, X., Gong, X. et al. Survival analysis between laparoscopic and open hepatectomy for hepatocellular carcinoma: a meta-analysis based on reconstructed time-to-event data. Hepatol Int 15, 1215–1235 (2021). https://doi.org/10.1007/s12072-021-10219-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-021-10219-1