Abstract
Introduction
To assess pre-therapeutic MRI-based radiomic analysis to predict the pathological complete response to neoadjuvant chemotherapy (NAC) in women with early triple negative breast cancer (TN).
Materials and methods
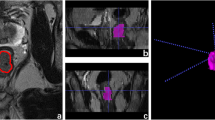
This monocentric retrospective study included 75 TN female patients with MRI (T1-weighted, T2-weighted, diffusion-weighted and dynamic contrast enhancement images) performed before NAC. For each patient, the tumor(s) and the parenchyma were independently segmented and analyzed with radiomic analysis to extract shape, size, and texture features. Several sets of features were realized based on the 4 different sequence images. Performances of 4 classifiers (random forest, multilayer perceptron, support vector machine (SVM) with linear or quadratic kernel) were compared based on pathological complete response (defined on the excised tissues), on 100 draws with 75% as training set and 25% as test.
Results
The combination of features extracted from different MR images improved the classifier performance (more precisely, the features from T1W, T2W and DWI). The SVM with quadratic kernel showed the best performance with a mean AUC of 0.83, a sensitivity of 0.85 and a specificity of 0.75 in the test set.
Conclusion
MRI-based radiomics may be relevant to predict NAC response in TN cancer. Our results promote the use of multi-contrast MRI sources for radiomics, providing enrich source of information to enhance model generalization.





Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the curve ROC
- DCE:
-
Dynamic contrast enhancement
- HER:
-
Human epidermal growth factor
- VOI:
-
Volume of interest
- MLP:
-
Multilayer perceptron
- MRI:
-
Magnetic resonance imaging
- NAC:
-
Neo-adjuvant chemotherapy
- pCR:
-
Pathological complete response
- ROC:
-
Receiver operating characteristic
- SVM:
-
Support vector machine
- TN:
-
Triple negative
- TNM:
-
Tumor, node, metastases
- T1W:
-
T1-weighted imaging
- T2W:
-
T2-weighted imaging
- DWI:
-
Diffusion weighted imaging
- GLCM:
-
Gray-level co-occurrence matrix
- GLSZM:
-
Gray-level size zone matrix
- NGTDM:
-
Neighborhood gray tone difference matrix
- SURF:
-
Speed-up robust features
- SUB3:
-
Subtraction between the image 3 min post-injection and the image pre-injection
References
Ferlay J, Colombet M, Soerjomataram I, Mathers C, Parkin DM, Piñeros M, Znaor A, Bray F (2018) Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer. https://doi.org/10.1002/ijc.31937
Billar JAY, Dueck AC, Stucky C-CH, Gray RJ, Wasif N, Northfelt DW, McCullough AE, Pockaj BA (2010) Triple-negative breast cancers: unique clinical presentations and outcomes. Ann Surg Oncol 17:384–390
Foulkes WD, Smith IE, Reis-Filho JS (2010) Triple-negative breast cancer. N Engl J Med 363:1938–1948
Derks MGM, van de Velde CJH (2018) Neoadjuvant chemotherapy in breast cancer: more than just downsizing. Lancet Oncol 19:2–3
Kong X, Moran MS, Zhang N, Haffty B, Yang Q (2011) Meta-analysis confirms achieving pathological complete response after neoadjuvant chemotherapy predicts favourable prognosis for breast cancer patients. Eur J Cancer 47:2084–2090
Liedtke C, Mazouni C, Hess KR, André F, Tordai A, Mejia JA, Symmans WF, Gonzalez-Angulo AM, Hennessy B, Green M, Cristofanilli M, Hortobagyi GN, Pusztai L (2008) Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol 26:1275–1281
Earl H, Provenzano E, Abraham J, Dunn J, Vallier A-L, Gounaris I, Hiller L (2015) Neoadjuvant trials in early breast cancer: pathological response at surgery and correlation to longer term outcomes—what does it all mean? BMC Med 13:234
For the European Society of Breast Imaging (EUSOBI), with language review by Europa Donna–The European Breast Cancer Coalition, Mann RM, Balleyguier C, Baltzer PA, Bick U, Colin C, Cornford E, Evans A, Fallenberg E, Forrai G, Fuchsjäger MH, Gilbert FJ, Helbich TH, Heywang-Köbrunner SH, Camps-Herrero J, Kuhl CK, Martincich L, Pediconi F, Panizza P, Pina LJ, Pijnappel RM, Pinker-Domenig K, Skaane P, Sardanelli F (2015) Breast MRI: EUSOBI recommendations for women’s information. Eur Radiol 25:3669–3678
Lambin P, Rios-Velazquez E, Leijenaar R, Carvalho S, van Stiphout RGPM, Granton P, Zegers CML, Gillies R, Boellard R, Dekker A, Aerts HJWL (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 48:441–446
Liu Z, Li Z, Qu J, Zhang R, Zhou X, Li L, Sun K, Tang Z, Jiang H, Li H, Xiong Q, Ding Y, Zhao X, Wang K, Liu Z, Tian J (2019) Radiomics of multiparametric MRI for pretreatment prediction of pathologic complete response to neoadjuvant chemotherapy in breast cancer: a multicenter study. Clin Cancer Res 25:3538–3547
Chamming’s F, Ueno Y, Ferré R, Kao E, Jannot AS, Chong J, Omeroglu A, Mesurolle B, Reinhold C, Gallix B (2018) Features from computerized texture analysis of breast cancers at pretreatment MR imaging are associated with response to neoadjuvant chemotherapy. Radiology 286:412–420
Fan M, Wu G, Cheng H, Zhang J, Shao G, Li L (2017) Radiomic analysis of DCE-MRI for prediction of response to neoadjuvant chemotherapy in breast cancer patients. Eur J Radiol 94:140–147
Weber JJ, Jochelson MS, Eaton A, Zabor EC, Barrio AV, Gemignani ML, Pilewskie M, Van Zee KJ, Morrow M, El-Tamer M (2017) MRI and prediction of pathologic complete response in the breast and axilla after neoadjuvant chemotherapy for breast cancer. J Am Coll Surg 225:740–746
Braman NM, Etesami M, Prasanna P, Dubchuk C, Gilmore H, Tiwari P, Plecha D, Madabhushi A (2017) Intratumoral and peritumoral radiomics for the pretreatment prediction of pathological complete response to neoadjuvant chemotherapy based on breast DCE-MRI. Breast Cancer Res 19:57
Lambin P, Leijenaar RTH, Deist TM, Peerlings J, de Jong EEC, van Timmeren J, Sanduleanu S, Larue RTHM, Even AJG, Jochems A, van Wijk Y, Woodruff H, van Soest J, Lustberg T, Roelofs E, van Elmpt W, Dekker A, Mottaghy FM, Wildberger JE, Walsh S (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749–762
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: images are more than pictures, they are data. Radiology 278:563–577
Liu Z, Wang S, Dong D, Wei J, Fang C, Zhou X, Sun K, Li L, Li B, Wang M, Tian J (2019) The applications of radiomics in precision diagnosis and treatment of oncology: opportunities and challenges. Theranostics 9:1303–1322
Limkin EJ, Sun R, Dercle L, Zacharaki EI, Robert C, Reuzé S, Schernberg A, Paragios N, Deutsch E, Ferté C (2017) Promises and challenges for the implementation of computational medical imaging (radiomics) in oncology. Ann Oncol 28:1191–1206
Agner SC, Rosen MA, Englander S, Tomaszewski JE, Feldman MD, Zhang P, Mies C, Schnall MD, Madabhushi A (2014) Computerized image analysis for identifying triple-negative breast cancers and differentiating them from other molecular subtypes of breast cancer on dynamic contrast-enhanced MR images: a feasibility study. Radiology 272:91–99
Quail DF, Joyce JA (2013) Microenvironmental regulation of tumor progression and metastasis. Nat Med 19:1423–1437
Collewet G, Strzelecki M, Mariette F (2004) Influence of MRI acquisition protocols and image intensity normalization methods on texture classification. Magn Reson Imaging 22:81–91
Suk T, Flusser J (2003) Combined blur and affine moment invariants and their use in pattern recognition. Pattern Recogn 36:2895–2907
Ming-Kuei Hu (1962) Visual pattern recognition by moment invariants. IEEE Trans Inf Theory 8:179–187
Tahmasbi A, Saki F, Shokouhi SB (2011) Classification of benign and malignant masses based on Zernike moments. Comput Biol Med 41:726–735
Saki F, Tahmasbi A, Soltanian-Zadeh H, Shokouhi SB (2013) Fast opposite weight learning rules with application in breast cancer diagnosis. Comput Biol Med 43:32–41
Aerts HJWL, Velazquez ER, Leijenaar RTH, Parmar C, Grossmann P, Carvalho S, Bussink J, Monshouwer R, Haibe-Kains B, Rietveld D, Hoebers F, Rietbergen MM, Leemans CR, Dekker A, Quackenbush J, Gillies RJ, Lambin P (2014) Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat Commun 5:4006
Kononenko I, Simec E, Robnik-Sikonja M (1997) Overcoming the myopia of inductive learning algorithms with RELIEFF. Appl Intell 7:39–55
Compton CC, Byrd DR, Garcia-Aguilar J, Kurtzman SH, Olawaiye A, Washington MK (2012) AJCC cancer staging atlas: a companion to the seventh editions of the AJCC cancer staging manual and handbook, 2nd edn. Springer, New York. https://doi.org/10.1007/978-1-4614-2080-4
Poggio F, Bruzzone M, Ceppi M, Pondé NF, La Valle G, Del Mastro L, de Azambuja E, Lambertini M (2018) Platinum-based neoadjuvant chemotherapy in triple-negative breast cancer: a systematic review and meta-analysis. Ann Oncol 29:1497–1508
Loibl S, O’Shaughnessy J, Untch M, Sikov WM, Rugo HS, McKee MD, Huober J, Golshan M, von Minckwitz G, Maag D, Sullivan D, Wolmark N, McIntyre K, Ponce Lorenzo JJ, Metzger Filho O, Rastogi P, Symmans WF, Liu X, Geyer CE (2018) Addition of the PARP inhibitor veliparib plus carboplatin or carboplatin alone to standard neoadjuvant chemotherapy in triple-negative breast cancer (BrighTNess): a randomised, phase 3 trial. Lancet Oncol 19:497–509
Schmid P, Cortes J, Pusztai L, McArthur H, Kümmel S, Bergh J, Denkert C, Park YH, Hui R, Harbeck N, Takahashi M, Foukakis T, Fasching PA, Cardoso F, Untch M, Jia L, Karantza V, Zhao J, Aktan G, Dent R, O’Shaughnessy J, KEYNOTE-522 Investigators (2020) Pembrolizumab for early triple-negative breast cancer. N Engl J Med 382:810–821
Houssami N, Macaskill P, von Minckwitz G, Marinovich ML, Mamounas E (2012) Meta-analysis of the association of breast cancer subtype and pathologic complete response to neoadjuvant chemotherapy. Eur J Cancer 48:3342–3354
Yan J, Chu-Shern JL, Loi HY, Khor LK, Sinha AK, Quek ST, Tham IWK, Townsend D (2015) Impact of image reconstruction settings on texture features in 18F-FDG PET. J Nucl Med 56:1667–1673
Fortin J-P, Parker D, Tunç B, Watanabe T, Elliott MA, Ruparel K, Roalf DR, Satterthwaite TD, Gur RC, Gur RE, Schultz RT, Verma R, Shinohara RT (2017) Harmonization of multi-site diffusion tensor imaging data. Neuroimage 161:149–170
Reuzé S, Orlhac F, Chargari C, Nioche C, Limkin E, Riet F, Escande A, Haie-Meder C, Dercle L, Gouy S, Buvat I, Deutsch E, Robert C (2017) Prediction of cervical cancer recurrence using textural features extracted from 18F-FDG PET images acquired with different scanners. Oncotarget. https://doi.org/10.18632/oncotarget.17856
Acknowledgements
This study was conducted as part of the LABEX PRIMES (ANR-11-LABX-0063) of the “Université de Lyon”, within the “Investissements d’Avenir” program (ANR-11-IDEX-0007) operated by the French National Research Agency (ANR). This study was also supported by the SIRIC LyriCAN grant (INCa_INSERM_DGOS_12563). We thank Sophie Darnis for her help with English language editing.
Author information
Authors and Affiliations
Contributions
AN: study conception and design, analysis and interpretation of data, drafting of manuscript, and critical revision. PC: study conception and design, acquisition of data, analysis and interpretation of data, and drafting of manuscript. BL: study conception and design, analysis and interpretation of data, and critical revision. P-EH: study conception and design and acquisition of data. FB: acquisition of data, and analysis and interpretation of data. OT: study conception and design. IT: study conception and design. AC: study conception and design, acquisition of data, analysis and interpretation of data, and drafting of manuscript. FP: analysis and interpretation of data and critical revision. OB: analysis and interpretation of data, and critical revision.
Corresponding author
Ethics declarations
Conflict of interests
All the authors declare that they have no conflict of interest.
Ethical approval
Ethical approval was waived by the local Ethics Committee of our institution in view of the retrospective nature of the study and all the procedures being performed were part of the routine care.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nemeth, A., Chaudet, P., Leporq, B. et al. Multicontrast MRI-based radiomics for the prediction of pathological complete response to neoadjuvant chemotherapy in patients with early triple negative breast cancer. Magn Reson Mater Phy 34, 833–844 (2021). https://doi.org/10.1007/s10334-021-00941-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-021-00941-0