Abstract
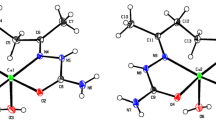
The dissolution of aqueous copper(II) acetate in coordinating bidentate 1,4-dioxane (reflux) affords anhydrous mononuclear complex Cu2(µ-OOCMe)4(dioxane)2 (I) (Cu…Cu 2.5781(3) Å). The crystal of complex I contains contacts of the α-H atom of the CH2 fragments of coordinated dioxane with the oxygen atom of the bridging anion with the formation of the 2D metal-organic framework (MOF). The anionic exchange reactions of the acetate bridges by pivalate or trifluoroacetate groups with the corresponding acids in boiling dioxane give 1D MOF {Cu2(µ-OOCR)4(dioxane)}n (R = tBu (II), Cu…Cu 2.5493(7) Å; CF3 (III), Cu…Cu 2.6391(12) Å) characterized by the step and linear geometry, respectively. A similar reaction of Cu2(µ-OOCMe)4(OH2)2 with HOOCCF3 but at room temperature in the presence of crude benzene affords mononuclear complex Cu(OOCCF3)2(OH2)3·2 dioxane (IV). Complexes I–IV are studied by elemental and X-ray diffraction analyses (CIF files CCDC nos. 2052618 (I), 2052619 (II), 2052620 (III), and 2052617 (IV)). The influence of the electronic and steric factors of substituents R on the compositions and structures of the mononuclear complexes and MOFs with 1,4-dioxane are discussed for the synthesized and known copper(II) tetracarboxylates.




Similar content being viewed by others
REFERENCES
Cotton, F.A., Wilkinson, G., Murillo, C.A., and Bochmann, M., Advanced Inorganic Chemistry, New York: Wiley, 1999.
Mehrotra, R.C. and Bohra, R., Metal Carboxylates, London: Academic, 1983.
Rakitin, Yu.V. and Kalinnikov, V.T., Sovremennaya magnetokhimiya (Modern Magnetic Chemistry), St. Petersburg: Nauka, 1984.
Lippard, S.J. and Berg, J.M., Principles of Bioinorganic Chemistry, Mill Valley: University Science Books, 1994, p. 199.
Solomon, E.I., Sundaram, U.M., and Machonkin, T.E., Chem. Rev., 1996, vol. 96, p. 2563. https://doi.org/10.1021/cr950046o
Cook, T.R., Zheng, Y.-R., and Stang, P.J., Chem. Rev., 2013, vol. 113, p. 734. https://doi.org/10.1021/cr3002824
Stock, N. and Biswas, S., Chem. Rev., 2012, vol. 112, p. 933. https://doi.org/10.1021/cr200304e
Furukawa, H., Cordova, K.E., O’Keeffe, M., et al., Science, 2013, vol. 341, p. 97. https://doi.org/10.1126/science.1230444
Stavila, V., Talin, A.A., and Allendorf, M.D., Chem. Soc. Rev., 2014, vol. 43, p. 5994. https://doi.org/10.1039/C4CS00096J
Uvarova, M., Sinelshchikova, A., Golubnichaya, M., et al., Cryst. Growth Des., 2014, no. 11, p. 5976. https://doi.org/10.1021/cg501157e
Mitrofanov, A.Yu., Rousseli, Y., Guilard, R., et al., New J. Chem., 2016, vol. 40, p. 5896. https://doi.org/10.1039/C5NJ03572D
Eremenko, I.L., Novotortsev, V.M., Sidorov, A.A., and Fomina, I.G., Ross. Khim. Zh., 2004, vol. 48, no. 1, p. 49.
Becht, M., Gerfin, T., and Dahmen, K.-H., Helv. Chim. Acta, 1994, vol. 77, p. 1288. https://doi.org/10.1002/hlca.19940770511
Pratt, R.C., Mirica, L.M., and Stack, T.D.P., Inorg. Chem., 2004, p. 8030. https://doi.org/10.1021/ic048904z
Tomkowicz, Z., Ostrovsky, S., Foro, S., et al., Inorg. Chem., 2012, vol. 51, no. 11, p. 6046. https://doi.org/10.1021/ic202529p
Mikhalitsyna, E.A., Tyurin, V.S., Nefedov, S.E., et al., Eur. J. Inorg. Chem., 2012, vol. 36, p. 5979. https://doi.org/10.1002/ejic.201200868
Hiroto, S., Furukawa, K., Shinokubo, H., and Osuka, A., J. Am. Chem. Soc., 2006, p. 12380. https://doi.org/10.1021/ja062654z
Sinelshchikova, A.A., Nefedov, S.E., Enakieva, Yu.Yu., et al., Inorg. Chem., 2013, vol. 52, no. 2, p. 999. https://doi.org/10.1021/ic302257g
Becker, H., Domshcke, G., Fanghaenel, E., and Fischer, M., Organikum. Organisch-Chemisches Grundpraktikum, Berlin: Deutscher Verlag für Wissenschäften, 1992, vol. 1.
Ingold, C.K. Structure and Mechanism in Organic Chemistry, Ithaca: Cornell University, 1969.
SMART (control) and SAINT (integration) Software. Version 5.0, Madison: Bruker AXS Inc., 1997.
SAINT. Area-Detector Integration Sofware, Madison: Bruker AXS Inc., 2012.
Sheldrick, G.M., SADABS. Program for Scaling and Correction of Area Detector Data, Göttingen: Univ. of Göttingen, 1997.
Sheldrick, G.M., Acta Crystallogr., Sect. C: Struct. Chem., 2015, vol. 71, p. 3. https://doi.org/10.1107/S2053229614024218
Uvarova, M.A. and Nefedov, S.E., Russ. J. Coord. Chem., 2020, vol. 46, p. 608. https://doi.org/10.1134/S1070328420090079
Uvarova, M.A. and Nefedov, S.E., Russ. J. Inorg. Chem., 2021, vol. 66, no. 6. https://doi.org/10.31857/S0044457X21060209
CSD. Version 5.42 (November 2020).
Smart, P., Espallargas, G.M., and Brammer, L., CrystEngComm, 2008, p. 1335. https://doi.org/10.1039/b806765a
Reck, G., Jahnig, W., and Prakt, J., Chem. Chem. Zeitung, 1979, vol. 321, p. 549. https://doi.org/10.1002/prac.19793210406
Kani, Y., Tsuchimoto, M., Ohba, S., and Tokii, T., Acta Crystallogr., Sect. C:. Cryst. Struct. Commun., 2000, vol. 56, p. e80. https://doi.org/10.1107/S0108270100002304
Borel, M.M. and Leclaire, A., Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1976, vol. 32, p. 1275.
Larionov, S.V., Glinskaya, L.A., Klevtsova, R.F., et al., Russ. J. Inorg. Chem., 1991, vol. 36, p. 2514.
ACKNOWLEDGMENTS
The XRD and IR spectral studies of the complexes were carried out using the equipment of the Center for Collective Use of Physical Methods of Investigation at the Kurnakov Institute of General and Inorganic Chemistry (Russian Academy of Sciences) functioning in terms of the state assignment of the Kurnakov Institute of General and Inorganic Chemistry (Russian Academy of Sciences) in the area of basic research.
Funding
This work was supported by the state assignment of the Kurnakov Institute of General and Inorganic Chemistry (Russian Academy of Sciences) in the area of basic research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
To blessed memory of Teacher: to the 80th birthday of Prof. A.A. Pasynskii
Translated by E. Yablonskaya
Rights and permissions
About this article
Cite this article
Uvarova, M.A., Nefedov, S.E. Correlations between the Structures of Binuclear Copper(II) Tetracarboxylates with 1,4-Dioxane and the Nature of Substituent R in the Carboxylate Anion. Russ J Coord Chem 47, 399–408 (2021). https://doi.org/10.1134/S1070328421060087
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328421060087