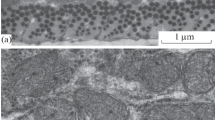
Abstract
A comparative electron microscopic and morphometric analysis of age-related transformations in the ultrastructure of the skeletal muscle mitochondrial apparatus was carried out in animal species with different aging programs: short-lived classical objects, such as C57BL/6 mice and Wistar rats, prematurely aging OXYS rats, and the longest-living rodents, naked mole-rats (Heterocephalus glaber), characterized by delayed aging. In C57BL/6 mice, age-related reorganization of the skeletal muscle mitochondrial apparatus corresponds to that reported previously for Wistar rats: the mitochondrial reticulum forms by the age of 2.5–3 months; by the age of 30 months, it undergoes a drastic reduction, due to which the number of mitochondrial cross-sections in muscle fibers decreases almost twofold, from 0.45 ± 0.074 to 0.23 ± 0.017 profiles per µm2. In C57BL/6 mice, no destructive changes in the mitochondrial ultrastructure were observed, in contrast to OXYS rats, in which age-related changes in the chondriome affect both the overall structure and internal ultrastructure of the muscle fiber mitochondrial apparatus. At the same time, in naked mole-rats, which are comparable with mice in their size, the number and size of mitochondria in skeletal muscles increase significantly by the age of five years, although no mitochondrial reticulum forms. It is hypothesized that a special organization of the mitochondrial apparatus in naked mole-rat skeletal muscles provide a proper level of redox processes in muscles, thus preventing a decline in their physical efficiency and the development of sarcopenia, whereas in C57BL/6 mice, Wistar and OXYS rats, age-related abnormalities in the structural organization of skeletal muscle mitochondrial apparatus may be one of the major causes for the development of age-related pathologies, including sarcopenia.












Similar content being viewed by others
REFERENCES
Baumann CW, Kwak D, Liu HM, Thompson VL (2016) Age-induced oxidative stress: How does it influence skeletal muscle quantity and quality? J Appl Physiol 121:1047-1052. https://doi.org/10.1152/japplphysiol.00321.2016
Brunk UT, Terman A (2002) The mitochondrial-lysosomal axis theory of aging: accumulation of damaged mitochondria as a result of imperfect autophagocytosis. Eur J Biochem 269:1996–2002. https://doi.org/10.1046/j.1432-1033.2002.02869.x
Derbré F, Gratas-Delamarche A, Gómez-Cabrera MC, Viña J (2014) Inactivity-induced oxidative stress: A central role in age-related sarcopenia? Eur J Sport Sci 14:98–108. https://doi.org/10.1080/17461391.2011.654268
Gamboa JL, Billings FT, Bojanowski MT, Gilliam LA, Yu C, Roshanravan B, Roberts LJ, Himmelfarb J, Ikizler TA, Brown NJ (2016) Mitochondrial dysfunction and oxidative stress in patients with chronic kidney disease. Physiol Rep 4(9):e12780. https://doi.org/10.14814/phy2.12780
Santos LS, Baraibar MA, Lundberg S, Eeg-Olofsson O, Larsson L, Friguet B (2015) Oxidative proteome alterations during skeletal muscle ageing. Redox Biol 5:267–274. https://doi.org/10.1016/j.redox.2015.05.006
Rygiel KA, Picard M, Turnbull DM (2016) The ageing neuromuscular system and sarcopenia: a mitochondrial perspective. J Physiol 594:4499–4512. https://doi.org/10.1113/JP271212
Dai DF, Chiao YA, Marcinek DJ, Szeto HH, Rabinovitch PS (2014) Mitochondrial oxidative stress in aging and healthspan. Longev Heal 3:6. https://doi.org/10.1186/2046-2395-3-6
Johnson ML, Robinson MM, Nair KS (2013) Skeletal muscle aging and the mitochondrion. Trends Endocrinol Metab 24:247–256. https://doi.org/10.1016/j.tem.2012.12.003
Sacktor B, Shimada Y (1972) Degenerative changes in the mitochondria of flight muscle from aging blowflies. J Cell Biol 52:465-477. https://doi.org/10.1083/jcb.52.2.465
Bakeeva LE, Saprunova VB, Pasyukova EG, Roshina NV (2007) Mitoptosis in the flight muscle of Drosophila melanogaster. Acad Sci Rep 413:1-3 (In Russ). https://doi.org/10.1134/S0012496607020111
Williams CM, Barness LA, Sawyer WH (1943) The utilization of glycogen by flies during flight and some aspects of the physiological ageing of Drosophila. Biol Bull (Woods Hole) 84:263-268. https://doi.org/10.2307/1538009
Walker DW, Benzer S (2004) Mitochondrial “swirls” induced by oxygen stress and in the Drosophila mutant hyperswirl. Proc Natl Acad Sci USA 101:10290–10295. https://doi.org/10.1073/pnas.0403767101
Anisimov VN, Bakeeva LE, Egormin PA, Filenko OF, Isakova EF, Manskikh VN, Mikhelson VM, Panteleeva AA, Pilipenko DI, Piskunova TS, Popovich IG, Saprunova VB, Samoylova TA, Semenchenko AV, Skulachev MV, Tyndyk ML, Vyssokikh MY, Yurova MN, Zabezhinsky MA, Skulachev VP (2008) Mitochondria- targeted plastoquinone derivatives as tools to interrupt execution of the aging program. 5. SkQ1 prolongs lifespan and prevents development of traits of senescence. Biochemistry (Moscow) 73:1329-1342. https://doi.org/10.1134/s0006297908120055
Beregi E, Regius O, Huttl T, Gobl Z (1988) Age-related changes in the skeletal muscle cells. Z Gerontol 21:83-86. PMID: 2968024
Bakarev MA, Nepomnyashchikh LM (2004) Structural manifestations of mitochondrial dysfunction in skeletal muscles in premature aging OXYS rats. Bull Exp Biol Med 138:674-679 (In Russ). eLIBRARY ID: 28932383
Crane JD, Devries C, Safdar A, Hamadeh MJ, Tarnopolsky MA (2010) The effect of aging on human skeletal muscle mitochondrial and intramyocellular lipid ultrastructure. J Gerontol 65:119-128. https://doi.org/10.1101/cshperspect.a029785
Orlander J, Kiessling K, Larsson L, Karlsson J, Aniansson A (1978) Skeletal muscle metabolism and ultrastructure in relation to age in sedentary men. Acta Physiol Scand 104:249-261. https://doi.org/10.1111/j.1748-1716.1978.tb06277.x
Palade GE (1956) Electron microscopy of mitochondria and other cytoplasmic structures. In: Enzymes: Units of Biological Structure and Function. Acad Press New York 185-215.
Gauthier GF, Padykula HA (1966) Cytological studies of fiber types in skeletal muscle. A comparative study of the mammalian diaphragm. J Cell Biol 28:333-354. https://doi.org/10.1083/jcb.28.2.333
Bubenzer HJ (1966) Die dunnen und die dicken Muskelfasern des Zwerchfells der Ratte. Z Zellforsch 69:520-550. https://doi.org/10.1007/BF00406300
Bakeeva LE, Skulachev VP, Chentsov YS (1978) Mitochondrial framework (reticulum mitochondriale) in rat diaphragm muscle. Biochim Biophys Acta 501:349-369. https://doi.org/10.1016/0005-2728(78)90104-4
Bakeeva LE, Chentsov YS, Skulachev VP (1981) Ontogenesis of mitochondrial reticulum in rat diaphragm muscle. Eur J Cell Biol 25:175-181. PMID: 7285951
Kirkwood SP, Munn EA, Brooks GA (1986) Mitochondrial reticulum in limb skeletal muscle. Am J Physiol 251:395-402. https://doi.org/10.1152/ajpcell.1986.251.3.C395
Glancy B, Hartnell LM, Malide D, Yu ZX, Combs CA, Connelly PS, Subramaniam S, Balaban RS (2015) Mitochondrial reticulum for cellular energy distribution in muscle. Nature 523:617-620. https://doi.org/10.1038/nature14614
Pellegrini G, Barbieri S, Moggio M, Cheldi A, Scarlato G, Minetti C (1985) A case of congenital neuromuscular disease with uniform type I fibers, abnormal mitochondrial network and jagged Z-line. Neuropediatrics 16:162-166. https://doi.org/10.1055/s-2008-1059533
Leduc-Gaudet JP, Picard M, Pelletier FSt-J, Sgarioto N, Auger MJ, Vallée J, Robitaille R, St-Pierre DH, Gouspillou G (2015) Mitochondrial morphology is altered in atrophied skeletal muscle of aged mice. Oncotarget 6:17923-17937. https://doi.org/10.18632/oncotarget.4235
Buffenstein R. (2008) Negligible senescence in the longest living rodent, the naked mole-rat: insights from a successfully aging species. Compar Physiol B 178:439–445. https://doi.org/10.1007/s00360-007-0237-5
Orr ME, Garbarino VR, Salinas A, Buffenstein R (2015) Sustained high levels of neuroprotective, high molecular weight, phosphorylated tau in the longest-lived rodent. Neurobiol Aging 36:1496–1504. https://doi.org/10.1016/j.neurobiolaging.2014.12.004
Larson J, Park TJ (2009) Extreme hypoxia tolerance of naked mole-rat brain. Neuroreport 20:1634–1637. https://doi.org/10.1097/WNR.0b013e32833370cf
Maina JN, Gebreegziabher Y, Woodley R, Buffenstein R (2001) Effects of change in environmental temperature and natural shifts in carbon dioxide and oxygen concentrations on the lungs of captive naked mole-rats (Heterocephalus glaber): a morphological and morphometric study. J Zool 253:371–382. https://doi.org/10.1017/S0952836901000346
Park TJ, Lu Y, Jüttner R, Smith EJ, Hu J, Brand A, Wetzel C, Milenkovic N, Erdmann B, Heppenstall PA, Laurito CE, Wilson SP, Lewin GR (2008) Selective Inflammatory Pain Insensitivity in the African Naked Mole-Rat (Heterocephalus glaber). PLoS Biol January 6:13. https://doi.org/10.1371/journal.pbio.0060013
Eldarov CM, Vangely IM, Kolosova NG, Bakeeva LE, Skulachev VP (2014) Antioxidant SkQ1 delays sarcopenia- associated damage of mitochondrial ultrastructure. Aging 6:140-148. https://doi.org/10.18632/aging.100636
Bakeeva LE (2015) Age-Related Changes in Ultrastructure of Mitochondria. Effect of SkQ1. Biochemistry 80:1582-8. https://doi.org/10.1134/S0006297915120068
Holtze S, Eldarov CM, Vays VB, Vangeli IM, Vysokikh MY, Bakeeva LE, Skulachev VP, Hildebrandt TB (2016) Study of Age-Dependent Structural and Functional Changes of Mitochondria in Skeletal Muscles and Heart of Naked Mole Rats (Heterocephalus glaber). Biochemistry 81: 1703-1712. https://doi.org/10.1134/S000629791612004
Skulachev VP, Holtze S, Vyssokikh MY, Bakeeva LE, Skulachev MV, Markov AV, Hildebrandt TB, Sadovnichii VA (2017) Neoteny, prolongation of youth: from naked mole rats to “naked apes” (humans). Physiol Rev 97:699–720. https://doi.org/10.1152/physrev.00040.2015
Funding
This work was supported by the Russian Foundation for Basic Research, grant No. 19-04-00578-a.
Author information
Authors and Affiliations
Contributions
Central idea and experimental design: L.E.B.; experimental studies: V.B.V., I.M.V.; mathematical data processing: Ch.M.E.; writing and editing a manuscript: V.B.V., I.M.V., Ch.M.E. and L.E.B.
Corresponding author
Ethics declarations
CONFLICT OF INTEREST
The authors declare that they have neither evident nor potential conflict of interest related to the publication of this article.
Additional information
Translated by A. Polyanovsky
Russian Text © The Author(s), 2021, published in Zhurnal Evolyutsionnoi Biokhimii i Fiziologii, 2021, Vol. 107, Nos. 6–7, pp. 876–894https://doi.org/10.31857/S0869813921060133.
Rights and permissions
About this article
Cite this article
Vays, V.B., Vangeli, I.M., Eldarov, C.M. et al. A Comparative Analysis of Age-Related Changes in the Structure of the Mitochondrial Apparatus in Skeletal Muscles of Species with Different Lifespan. J Evol Biochem Phys 57, 730–742 (2021). https://doi.org/10.1134/S0022093021030200
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022093021030200