Abstract
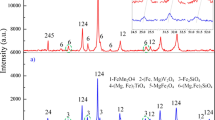
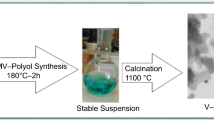
Vanadium-containing yellow pigment was deposited onto marshalite particles through solution-combustion reaction of V2O5 with salts of Bi, Zn, and Ca using citric acid as a fuel and characterized by XRD, FT-IR spectra, and SEM. Yellow color of the pigment was due to the presence of bismuth vanadate BiVO4. Minor impurity of Bi4V2O11 by-product imparted a beige hue to the pigment. The addition of Zn2+ and Ca2+ cations improved the chromatic characteristics of the pigment. Size distribution of resultant pigment is defined by that of mineral substrate powder. Our pigments were heat resistant and retained their color up to 800°С.







Similar content being viewed by others
REFERENCES
Costa, A.C.F.M., Leite, A.M.D., Ferreira, H.S., Kiminami, R.H.G.A., Cava, S., and Gama, L., Brown pigment of the nanopowder spinel ferrite prepared by combustion reaction, J. Eur. Ceram. Soc., 2008, no. 28, pp. 2033–3037. https://doi.org/10.1016/j.jeurceramsoc.2007.12.039
Ianos, R., Lazau, R., Pacurariu, C., and Barvinschi, P., Solution combustion synthesis of MgAl2O4 using fuel mixtures, Mater. Res. Bull., 2008, no. 43, pp. 3408–3415. https://doi.org/10.1016/materresbull.2008.02.003
Li, H., Wei, H.-Y., Cui, Y., Sang, R.-L., Bu, J.-L., Wei, Y.-N., Lin, J., and Zhao, J.-H., Synthesis and characterization of MgAl2O4 spinel nanopowders via nonhydrolytic sol–gel route, J. Ceram. Soc. Jpn., 2017, vol. 125, no. 3, pp. 100–104. https://doi.org/10.2109/jcersj2/16297
Zhuravlev, V.A., Minin, R.V., Itin, V.I., and Lilenko, I.Yu., Structural parameters and magnetic properties of copper ferrite nanopowders obtained by the sol–gel combustion, J. Alloys Compd., 2017, vol. 692, pp. 705–712. https://doi.org/10.1016/j.jallcom.2016.09.069
Azurdia, J., Marchal, J., and Laine, R.M., Synthesis and characterization of mixed-metal oxide nanopowders along the CoOx–Al2O3 tie line using liquid-feed flame spray pyrolysis, J. Am. Ceram. Soc., 2006, vol. 89, no. 9, pp. 2749–2756. https://doi.org/10.1111/j.1551-2916.2006.01155.x
Ahmed, I.S., Shama, S.A., Dessouki, H.A., and Ali, A.A., Synthesis, thermal and spectral characterization of nanosized NixMg1–xAl2O4 powders as new ceramic pigments via combustion route using 3-methylpyrozole-5-one as fuel, Spectrochim. Acta, Part A, 2011, vol. 81, pp. 324–333. https://doi.org/10.1016/j.saa.2011.06.019
Ianos, R., Lazau, R., and Barvinschi, P., Synthesis of Mg1–xCoxAl2O4 blue pigments via combustion route, Adv. Powder Technol., 2011, no. 22, pp. 396–400. https://doi.org/10.1016/J.apl.2010.06.006
Ran, R., McEvoy, J.G., and Zhang, Z., Synthesis and optimization of visible light active BiVO4 photocatalysts for the degradation of RhB, Int. J. Photoenergy, 2015, vol. 2015, 612857. https://doi.org/10.1155/2015/612857
Rodionov, I.A. and Zvereva, I.A., Photocatalytic activity of layered perovskite-like oxides in practically valuable chemical reactions, Russ. Chem. Rev., 2016, vol. 85, no. 3, pp. 248–279. https://doi.org/10.1070/RCR4547?Iocalt=label:RUSSIAN
Chen, S.H., Jiang, Y.S., and Lin, H.Y., Easy synthesis of BiVO4 for photocatalytic overall water splitting, ACS Omega, 2020, vol. 5, no. 15, pp. 8927–8933. https://doi.org/10.1021/acsomega.0c00699
Kendall, K.R., Navas, C., Thomas, J.K., and Zur Loye, H.C., Recent Developments in Oxide Ion Conductors: Aurivillius Phases, Chem. Mater., 1996, no. 8 (3), pp. 642–649. https://doi.org/10.1021/cm9503083
Peiwen Lv and Feng Huang, Gamma-Bi4V2O11 – A layered oxide material for ion exchange in aqueous media, RSC Adv., 2019, vol. 9, pp. 8650–8653. https://doi.org/10.1039/c8ra09565e
Mairesse, G., Roussel, P., Vannier, R.N., Anne, M., Pirovano, C., and Nowogrocki, G., Crystal structure determination of α, β and γ-Bi4V2O11 polymorphs. I: γ and β-Bi4V2O11, Solid State Sci., 2003, vol. 5, no. 6, pp. 851–859. https://doi.org/10.1016/S1293-2558(03)00015-3
Mairesse, G., Roussel, P., Vannier, R.N., Anne, M., and Nowogrocki, G., Crystal structure determination of α, β and γ-Bi4V2O11 polymorphs. II: Crystal structure of α-Bi4V2O11, Solid State Sci., 2003, vol. 5, no. 6, pp. 861–869. https://doi.org/10.1016/S1293-2558(03)00016-5
Girsova, M.A., Firstov, S.V., and Antropova, T.V., Spectral and optical properties of the bismuth-containing quartz-like glasses, J. Phys.: Conf. Ser., 2014, vol. 541, 12022. https://doi.org/10.1088/1742-6596/541/1/012022
Petrov, I.Ya. and Tryasunov, B.G., Structure and catalytic behavior of supported molybdenum, vanadium, and chromium oxide catalysts for dehydrogenation of hydrocarbons, Vestn. Kuzbasskogo Gos. Tekh. Univ., 2007, no. 5, pp. 73–80.
Nakamoto, K., Infrared and Raman Spectra of Inorganic and Coordination Compounds, Part B: Applications in Coordination, Organometallic, and Bioinorganic Chemistry, New York: Wiley, 2009.
Domoratskii, K.V., Pastukhov, V.I., Kudzin, A.Yu., Sadovskaya, L.Ya., Rizak, V.M., and Stefanovich, V.A., Raman scattering in the Bi2TeO5 single crystal, Phys. Solid State, 2000, vol. 42, no. 8, pp. 1443–1446.
Dimitriev, Y., Krupchanska, M., Ivanova, Y., and Staneva, A., Sol–gel synthesis of materials in the system Bi2O3–SiO2, J. Univ. Chem. Technol. Metall. (Sofia), 2010, vol. 45, no. 1, pp. 39–42.
Ganeshbabu, M., Kannan, N., Sundara Venkatesh, P., Paulraj, G., Jeganathan, K., and MubarakAli, D., Synthesis and characterization of BiVO4 nanoparticles for environmental applications, RSC Adv., 2020, vol. 10, 18315. https://doi.org/10.1039/D0RA01065K
Carrero, C.A., Schloegl, R., Wachs, I.E., and Schomaecker, R., Critical literature review of the kinetics for the oxidative dehydrogenation of propane over well-defined supported vanadium oxide catalysts, ACS Catal., 2014, vol. 4, no. 10, pp. 3357–3380. https://doi.org/10.1021/cs5003417
Ivanovskaya, M.I., Ovodok, E.A., and Kotsikau, D.A., Interaction of carbon monoxide with In2O3 and In2O3‒Au nanocomposite, J. Appl. Spectrosc., 2012, vol. 78, no. 6, pp. 842–847. https://doi.org/10.1007/s10812-012-9542-1
Author information
Authors and Affiliations
Corresponding authors
Additional information
Translated by Yu. Scheck
About this article
Cite this article
Radishevskaya, N.I., Nazarova, A.Y., Lvov, O.V. et al. Deposition of Vanadium-Containing Yellow Pigment onto Marshalite Particles through Solution-Combustion Reaction. Int. J Self-Propag. High-Temp. Synth. 30, 81–86 (2021). https://doi.org/10.3103/S1061386221020126
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1061386221020126