Abstract
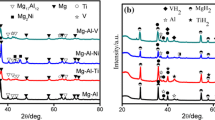
The microstructures and thermodynamic properties of MmNi5-xAlx hydrogen storage alloys are studied in this article. The structure was assessed with XRD and SEM. Pressure-composition isotherms for hydrogen absorption/desorption at 0, 25 and 50 °C were used for assessment of thermodynamic properties. XRD results show that the samples consist of single-phase CeNi5–type. The alloys exhibit second phases, porosities, and cracks. Al microsegregation is the main reason for the second phase formation. With increasing Al, the microsegregation and the amount of second phases are increased. The hydrogen storage capacities of MmNi5-xAlx (x = 0.2, 0.3, 0.4 at %) alloys are 1.40, 1.37 and 1.34 wt%, respectively. The equilibrium pressures and hysteresis (1.14, 0.96 and 0.72 for 0.2, 0.3 and 0.4 Al, respectively) for all three compositions decrease with increasing Al, that proves the formation of more stable hydrides. Furthermore, the enthalpy of hydride formation decrease to -20.770, -23.565, -26.306 kJ mol-1 H2 for the MmNi5-xAlx (x = 0.2, 0.3, 0.4 at %) alloys, respectively. This shows the improvement of thermodynamic stability and properties.






Similar content being viewed by others
References
Zhou L 2005 Progress and Problems in Hydrogen Storage Methods; Renew. Sustain. Energy Rev. 9 395–408
Hirscher M 2020 Materials for Hydrogen-Based Energy Storage Past, Recent Progress and Future Outlook; J. Alloy. Compd. 827 153548
Zareii S M, Arabi H and Pourarian F 2014 A Comprehensive Investigation of Structural, Morphological, Hydrogen Absorption and Magnetic Properties of MmNi4.22Co0.48Mn0.15Al0.15 Alloy. Int. J. Mod. Phys. B 28 1450125
Mungole M N and Balasubramaniam R 1998 Hydrogen Desorption Kinetics in MmNi4.2Al0.8-H System. Int. J. Hydrog. Energy 23 349–353
Molinas B, Pontarollo A, Scapin M, Peretti H, Melnichuk M, Corso H, Aurora A, Mirabile Gattia D and Montone A 2016 The Optimization of MmNi5−xAlx Hydrogen Storage Alloy for Sea or Lagoon Navigation and Transportation; Int. J. Hydrog. Energy 41 14484–14490
Yang F S, Chen X Y, Wu Z, Wang S M, Wang G X, Zhang Z X and Wang Y Q 2017 Experimental Studies on The Poisoning Properties of a Low-Plateau Hydrogen Storage Alloy LaNi4.3Al0.7 Against CO impurities; Int. J. Hydrog. Energy 42 16225–16234
Wijayanti I D, Denys R, Suwarno A, Volodin A, Lototsky M V, Guzik M N, Nei J, Young K, Roven H J and Yartys V 2020 Hydrides of Laves Type TiZr Alloys with Enhanced H Storage Capacity as Advanced Metal Hydride Battery Anodes; J. Alloy. Compd. 828 154354
Yang S, Han S, Li Y and Liu J 2013 Study on the Microstructure and Electrochemical Kinetic Properties of MmNi4.50−xMnxCo0.45Al0.30 (0.25≤x≤0.45) Hydrogen Storage Alloys; Mate. Sci. Eng. B. 178 39–44
Osumi Y, Suzuki H, Kato A, Oguro K, Kawai S and Kaneko M 1983 Hydrogen Absorption-Desorption Characteristics of MmNiAlM and MmNiMnM Alloys (Mm: misch metal); J. Less Common Met. 89 287–329
Iwakula C, Oura T, Inoue H and Matsuoka M 1996 Effects of Substitusion with Foreiyn Metals on the Crystallographic, Thermodynamic and Electrochemical Properties of AB5-type Hydrogen Storage Alloys; Electrochim. Acta 41 117–121
Zhang Y, Chen M, Wang X and Wang G 2004 Effect of Boron Addition on the Microstructures and Electrochemical Properties of MmNi 3.80 Co0.4 Mn0.6 Al0.2 Electrode Alloys Prepared by Casting and Rapid Quenching; J. Alloy. Compd. 373 291–297
Afhari Abolkarlou M and Amerioun M H 2019 Experimental Study of Structural and Magnetic Properties of LaNi5 and MmNi4.7Al0.3 Hydrogen Storage Alloys; J. Supercond. Nov. Mag. 32 1853–1857
Liu J, Li K, Cheng H, Yan K, Wang Y, Liu Y, Jin H and Zheng Z 2017 New Insights into the Hydrogen Storage Performance Degradation and Al Functioning Mechanism of LaNi 5–x Alx Alloys; Int. J. Hydrog. Energy 42 24904–24914
Liu J, Zheng Z, Cheng H, Li K, Yan K, Han X, Wang Y and Liu Y 2018 Long-Term Hydrogen Storage Performance and Structural Evolution of LaNi4Al Alloy; J. Alloy. Compd. 731 172–180
Sharma V K and Anil Kumar E 2014 Effect of Measurement Parameters on Thermodynamic Properties of La-Based Metal Hydrides; Int. J. Hydrog. Energy 39 5888–5898
Sharma V K, Anil Kumar E, Prakash Maiya M and Srinivasa Murthy S 2014 Experimental and Theoretical Studies on Static and Dynamic Pressure-Concentration Isotherms of MmNi5−xAlx (x = 0, 0.3, 0.5 and 0.8) Hydrides; Int. J. Hydrog. Energy 39 18940–18951
Edalati K, Shao H, Emami H and Iwaoka H 2016 Activation of Titanium-Vanadium Alloy for Hydrogen Storage by Introduction of Nanograins and Edge Dislocations Using High Pressure Torsion; Int. J. Hydrog. Energy 41 8917–8924
Pickering L A 2013 Ti-V-Mn Based Metal Hydrides for Hydrogen Storage and Compression Applications. Ph.D. thesis, University of Birmingham, Brimingham
Bowman R C, Craft B D, Attalla A, Mendelsohn M H and Gruen D M 1980 Role of Aluminum Substitution on Hydrogen Diffusion in β-LaNi5−yAlyHx; J. Less Common Met. 732 227–232
Percheron-Guégan A, Lartigue C, Achard J, Germi P and Tasset F 1980 Neutron and X-ray Diffraction Profile Analyses and Structure of LaNi5, LaNi5− xAlx and LaNi5− xMnx Intermetallics and Their Hydrides (deuterides); J. Less Common Met. 741 1–1
AlaviSadr S M, Arabi H and Pourarian F 2014 Synthesis, Characterization and Hydrogen Storage Properties of Mm (Ni Co, Mn, Al) 5 alloy; Iranian J. Hydrogen Fuel Cell. 1 83–94
Na Y S, Kim Y G and Lee J Y 1994 A study on the hydrogenation properties of the MmNi4.5Al0.5Zrx (x = 0−0.2) alloys; Int. J. Hydrog. Energy 19 899–903
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
SEIFOLLAHI, M., JOUDAKI, M., ABBASI, S.M. et al. Microstructure and thermodynamic assessment of MmNi5-xAlx hydrogen storage alloys. Sādhanā 46, 127 (2021). https://doi.org/10.1007/s12046-021-01578-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12046-021-01578-4