Abstract
Objective
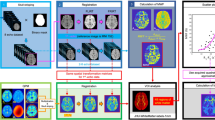
Scan time reduction is necessary for volumetric acquisitions to improve workflow productivity and to reduce motion artifacts during MRI procedures. We explored the possibility that Compressed Sensing-4 (CS-4) can be employed with 3D-turbo-field-echo T1-weighted (3D-TFE-T1W) sequence without compromising subcortical measurements on clinical 1.5 T MRI.
Materials and methods
Thirty-three healthy volunteers (24 females, 9 males) underwent imaging scans on a 1.5 T MRI equipped with a 12-channel head coil. 3D-TFE-T1W for whole-brain coverage was performed with different acceleration factors, including SENSE-2, SENSE-4, CS-4. Freesurfer, FSL’s FIRST, and volBrain packages were utilized for subcortical segmentation. All processed data were assessed using the Wilcoxon signed-rank test.
Results
The results obtained from SENSE-2 were considered as references. For SENSE-4, the maximum signal-to-noise ratio (SNR) drop was detected in the Accumbens (51.96%). For CS-4, the maximum SNR drop was detected in the Amygdala (10.55%). Since the SNR drop in CS-4 is relatively small, the SNR in all of the subcortical volumes obtained from SENSE-2 and CS-4 are not statistically different (P > 0.05), and their Pearson’s correlation coefficients are larger than 0.90. The maximum biases of SENSE-4 and CS-4 were found in the Thalamus with the mean of differences of 1.60 ml and 0.18 ml, respectively.
Conclusion
CS-4 provided sufficient quality of 3D-TFE-T1W images for 1.5 T MRI equipped with a 12-channel receiver coil. Subcortical volumes obtained from the CS-4 images are consistent among different post-processing packages.





Similar content being viewed by others
References
Giedd JN, Blumenthal J, Jeffries NO, Castellanos FX, Liu H, Zijdenbos A et al (1999) Brain development during childhood and adolescence: a longitudinal MRI study. Nat Neurosci 2:861–863
Ashburner J, Friston KJ (2000) Voxel-based morphometry - the methods. Neuroimage 11:805–821
Azevedo CJ, Cen SY, Khadka S, Liu S, Kornak J, Shi Y et al (2018) Thalamic atrophy in multiple sclerosis: a magnetic resonance imaging marker of neurodegeneration throughout disease. Ann Neurol 83(2):223–234
Pini L, Pievani M, Bocchetta M, Altomare D, Bosco P, Cavedo E et al (2016) Brain atrophy in Alzheimer’s Disease and aging. Ageing Res Rev 30:25–48
Jack CR Jr, Bernstein MA, Fox NC, Thompson P, Alexander G, Harvey D et al (2008) The Alzheimer’s Disease Neuroimaging Initiative (ADNI): MRI methods. J Magn Reson Imaging 27:685–691
Petersen RC, Aisen PS, Beckett LA et al (2010) Alzheimer’s Disease Neuroimaging Initiative (ADNI): clinical characterization. Neurology 74(3):201–209
van Erp TG, Hibar DP, Rasmussen JM et al (2016) Subcortical brain volume abnormalities in 2028 individuals with schizophrenia and 2540 healthy controls via the ENIGMA consortium. Mol Psychiatry 21(4):547–553
van Erp TG, Greve DN, Rasmussen J et al (2014) A multi-scanner study of subcortical brain volume abnormalities in schizophrenia. Psychiatry Res 222(1–2):10–16
Jovicich J, Czanner S, Han X et al (2009) MRI-derived measurements of human subcortical, ventricular and intracranial brain volumes: Reliability effects of scan sessions, acquisition sequences, data analyses, scanner upgrade, scanner vendors and field strengths. Neuroimage 46(1):177–192
Godenschweger F, Kägebein U, Stucht D, Yarach U, Sciarra A, Yakupov R et al (2016) Motion correction in MRI of the brain. Phys Med Biol 61(5):R32–R56
Havsteen I, Ohlhues A, Madsen KH, Nybing JD, Christensen H, Christensen A (2017) Are movement artifacts in magnetic resonance imaging a real problem?—a narrative review. Front Neurol 8:232
Reuter M, Tisdall MD, Qureshi A, Buckner RL, van der Kouwe AJW, Fischl B (2015) Head motion during MRI acquisition reduces gray matter volume and thickness estimates. Neuroimage 15(107):107–115
Pruessmann KP, Weiger M, Scheidegger MB, Boesiger P (1999) SENSE: sensitivity encoding for fast MRI. Magn Reson Med 42(5):952–962
Griswold MA, Jakob PM, Heidemann RM, Nittka M, Jellus V, Wang J et al (2002) Generalized autocalibrating partially parallel acquisitions (GRAPPA). Magn Reson Med 47(6):1202–1210
Sodickson DK, Griswold MA, Jakob PM (1999) SMASH imaging. Magn Reson Imaging Clin N Am 7(2):237–254
Hamilton J, Franson D, Seiberlich N (2017) Recent advances in parallel imaging for MRI. Prog Nucl Magn Reson Spectrosc 101:71–95
Lin FH, Huang T, Chen NK, Wang FN, Stufflebeam SM, Belliveau JW et al (2005) Functional MRI using regularized parallel imaging acquisition. Magn Reson Med 54(2):343–353
Leung KK, Malone IM, Ourselin S et al (2015) Effects of changing from non-accelerated to accelerated MRI for follow-up in brain atrophy measurement. Neuroimage 107:46–53
Krueger G, Granziera C, Jack CR Jr et al (2012) Effects of MRI scan acceleration on brain volume measurement consistency. J Magn Reson Imaging 36(5):1234–1240
Maclaren J, Han Z, Vos SB, Fischbein N, Bammer R (2014) Reliability of brain volume measurements: a test-retest dataset. Sci Data 1:140037
Manning EN, Leung KK, Nicholas JM et al (2017) A comparison of accelerated and non-accelerated MRI scans for brain volume and boundary shift integral measures of volume change: evidence from the ADNI dataset. Neuroinformatics 15(2):215–226
Lustig M, Donoho DL, Santos JM, Pauly JM (2008) Compressed sensing MRI. IEEE Signal Process Mag 25(2):72–82
Lustig M, Donoho D, Pauly JM (2007) Sparse MRI: the application of compressed sensing for rapid MR imaging. Magn Reson Med 58(6):1182–1195
Otazo R, Kim D, Axel L, Sodickson DK (2010) Combination of compressed sensing and parallel imaging for highly accelerated first-pass cardiac perfusion MRI. Magn Reson Med 64(3):767–776
Jaspan ON, Fleysher R, Lipton ML (2015) Compressed sensing MRI: a review of the clinical literature. Br J Radiol 88(1056):20150487
Mussard E, Hilbert T, Forman C, Meuli R, Thiran JP, Kober T (2020) Accelerated MP2RAGE imaging using Cartesian phyllotaxis readout and compressed sensing reconstruction. Magn Reson Med 84(4):1881–1894
Smith DS, Li X, Abramson RG, Quarles CC, Yankeelov TE, Welch EB (2013) Potential of compressed sensing in quantitative MR imaging of cancer. Cancer Imaging 13(4):633–644
Mair RW, Hanford LC, Mussard E, Hilbert T, Kober T, Buckner RL (2019) Towards 1 min brain morphometry—evaluating compressed-sensing MPRAGE. In: Proceedings of the International Society for Magnetic Resonance in Medicine. p. 2978
Shin DD, Rettmann D, Takei N, Banerjee S (2020) Compressed Sensed MPRAGE with Parallel Imaging: Image Quality Metrics and Morphometry Study at 3T. In: Proceedings of the International Society for Magnetic Resonance in Medicine. p. 1747
Duan Y, Zhang J, Zhuo Z, Ding J, Ju R, Wang J et al (2020) Accelerating brain 3D T1-weighted turbo field echo MRI using compressed sensing-sensitivity encoding (CS-SENSE). Eur J Radiol 131:109255
Geerts-Ossevoort L, deWeerdt E, Duijndam A, van Ijperen G, Peeters H, Doneva M, Nijenhuis M, Huang A. (2018) Compressed SENSE. Speed done right. Every time. Philips® healthcare, Netherlands. https://philipsproductcontent.blob.core.windows.net/assets/20180109/619119731f2a42c4acd4a863008a46c7.pdf. Accessed 05 Jan 2021
Falkovskiy P, Brenner D, Feiweier T et al (2016) Comparison of accelerated T1-weighted whole-brain structural-imaging protocols. Neuroimage 124:157–167
Ching CRK, Hua X, Hibar DP, Ward CP, Gunter JL, Bernstein MA et al (2012) the Alzheimers Disease Neuroimaging Initiative. P. M. T. MRI scan acceleration and power to track brain change. MICCAI NIBAD
Ching CR, Hua X, Hibar DP, Ward CP, Gunter JL, Bernstein MA et al (2015) Does MRI scan acceleration affect power to track brain change? Neurobiol Aging 1:167–177
Sartoretti T, Sartoretti E, van Smoorenburg L, Schwenk Á, Mannil M, Graf N et al (2020) Spiral 3-dimensional T1-weighted turbo field echo: increased speed for magnetization-prepared gradient echo brain magnetic resonance imaging. Invest Radiol 55(12):775–784
Sartoretti E, Sartoretti T, Binkert C, Najafi A, Schwenk Á, Hinnen M et al (2019) Reduction of procedure times in routine clinical practice with Compressed SENSE magnetic resonance imaging technique. PLoS ONE 14(4):e0214887
Morey RA, Petty CM et al (2009) A comparison of automated segmentation and manual tracing for quantifying hippocampal and amygdala volumes. Neuroimage 45(3):855–866
Chauffert N, Ciuciu P, Kahn J, Weiss P (2014) Variable density sampling with continuous trajectories. SIAM J Imag Sci 7(4):1962–1992
Puy G, Vandergheynst P, Wiaux Y (2011) On variable density compressive sampling. IEEE Signal Process Lett 18(10):595–598
Chauffert N, Weiss P, Kahn J, Ciuciu P (2014) Gradient waveform design for variable density sampling in Magnetic Resonance Imaging. arXiv: Optimization and Control
Takahashi J, Machida Y, Aoba M et al (2021) Noise power spectrum in compressed sensing magnetic resonance imaging. Radiol Phys Technol 14:93–99
Geethanath S, Reddy R, Konar AS et al (2013) Compressed sensing MRI: a review. Crit Rev Biomed Eng 41(3):183–204
Schoemaker D, Buss C, Head K et al (2016) Hippocampus and amygdala volumes from magnetic resonance images in children: assessing accuracy of FreeSurfer and FSL against manual segmentation. Neuroimage 129:1–14
Guo C, Ferreira D, Fink K, Westman E, Granberg T (2019) Repeatability and reproducibility of FreeSurfer, FSL-SIENAX and SPM brain volumetric measurements and the effect of lesion filling in multiple sclerosis. Eur Radiol 29(3):1355–1364
Manjón JV, Coupé P (2016) volBrain: an online MRI brain volumetry system. Front Neuroinform 27:10–30
Liang P, Shi L, Chen N et al (2015) Construction of brain atlases based on a multi-center MRI dataset of 2020 Chinese adults. Sci Rep 5:18216
Tang Y, Hojatkashani C, Dinov ID, Sun B, Fan L, Lin X et al (2010) The construction of a Chinese MRI brain atlas: a morphometric comparison study between Chinese and Caucasian cohorts. Neuroimage 51:33–41
Bai J, Abdul-Rahman MF, Rifkin-Graboi A, Chong YS, Kwek K, Saw SM et al (2012) Population differences in brain morphology and microstructure among Chinese, Malay and Indian neonates. PLoS ONE 7:e47816
Takahashi R, Ishii K, Kakigi T, Yokoyama K (2011) Gender and age differences in normal adult human brain: voxel-based morphometric study. Hum Brain Mapp 32:1050–1058
Acknowledgements
This work was financially supported by the Faculty of Associated Medical Sciences, Chiang Mai University. We thank Philips (Thailand) Ltd., and Philips Healthcare, Asia Pacific for providing the Compressed SENSE technique. We thank Laohawee N., Manonai N., Porploy J., and Yingmeesakul S. for data collecting. The authors wish to thank Ms. Samantha Burman for her help in editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Prapatsorn Sangpin is an employee of Philips Healthcare. All other authors have no conflict of interest to declare.
Ethical standards
The measurements on human subjects in this study have been approved by the local ethics committee and have therefore been performed following the ethical standards laid down in the Declaration of Helsinki. All involved participants have given their informed consent before recruitment in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yarach, U., Saekho, S., Setsompop, K. et al. Feasibility of accelerated 3D T1-weighted MRI using compressed sensing: application to quantitative volume measurements of human brain structures. Magn Reson Mater Phy 34, 915–927 (2021). https://doi.org/10.1007/s10334-021-00939-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-021-00939-8