Abstract
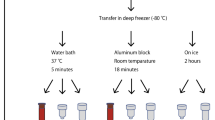
Due to degradation and limited quantities, one of the restrictions of molecular study is the inability to obtain total RNA extraction from frozen blood, while high-quality RNA is required for complementary DNA (cDNA) synthesis and gene expression analysis. To achieve optimal RNA extraction from frozen blood, the performance of three commercial RNA isolation kits, NucleoSpin RNA Blood kit, GENEzol reagent kit, and GeneJET Blood RNA kit, was examined. In addition, the effects of specific manipulation on the two of them (NucleoSpin RNA Blood kit, and GENEzol reagent kit) were evaluated. Agarose gel electrophoresis, Qubit fluorometer, and quantitative real-time PCR (qRT-PCR) were used. The mean yields of total RNA and cycle threshold (Ct) values of gene expression were investigated before manipulation and after manipulation. Three housekeeping genes, DECR1, GAPDH, ACTB, and one target gene P53, were involved. In the results, before manipulation, Ct values of gene expression and yields of RNA showed that the GeneJET, GENEzol, and NucleoSpin were the first, second, and third effective kits for RNA extraction from frozen blood, respectively. On the other hand, after manipulation, RNA yields and Ct values showed significant differences in terms of quantity and quality. Finally, as a result of the manipulation, NucleoSpin was shown to be the best and most powerful kit for isolating RNA from frozen blood. Based on these findings, the ability to obtain high-quality total RNA extraction from frozen blood was one of the major advantages of our modifications.





Similar content being viewed by others
References
Beekman JM, Reischl J, Henderson D, Bauer D, Ternes R, Peña C, Lathia C, Heubach J (2009) Recovery of microarray-quality RNA from frozen EDTA blood samples. J Pharmacol Toxicol Methods 59:44–49. https://doi.org/10.1016/j.vascn.2008.10.003
Bernard PS, Wittwer CT (2002) Real-time PCR technology for cancer diagnostics. Clin Chem 48:1178–1185. https://pubmed.ncbi.nlm.nih.gov/12142370/
Chai V, Vassilakos A, Lee Y, Wright JA, Young AH (2005) Optimization of the PAXgene blood RNA extraction system for gene expression analysis of clinical samples. J Clin Lab Anal 19:182–188. https://doi.org/10.1002/jcla.20075
Deng MY, Wang H, Ward GB, Beckham TR, McKenna TS (2005) Comparison of six RNA extraction methods for the detection of classical swine fever virus by real-time and conventional reverse transcription-PCR. J Vet Diagn Invest 17:574–578. https://doi.org/10.1177/104063870501700609
Farrell RE Jr (2006) RNA methodologies. Rev Cell Biol Molecular Med 18:17–31
Fleige S, Pfaffl MW (2006) RNA integrity and the effect on the real-time qRT-PCR performance. BMC Mol Biol 27:126–139. https://doi.org/10.1016/j.mam.2005.12.003
Haynes HR et al (2018) Evaluation of the quality of RNA extracted from archival FFPE glioblastoma and epilepsy surgical samples for gene expression assays. J Clin Pathol 71:695–701. https://doi.org/10.1136/jclinpath-2017-204969
Holley CL, Topkara VK (2011) An introduction to small non-coding RNAs: miRNA and snoRNA. Cardiovasc Drugs Ther 25:151–159. https://doi.org/10.1007/s10557-011-6290-z
Jakovljević KV, Spasić MR, Mališić EJ, Dobričić JD, Krivokuća AM, Janković R (2010) Comparison of phenol-based and alternative RNA isolation methods for gene expression analyses. J Serb Chem Soc 75:1053–1061. https://doi.org/10.2298/JSC091223084J
Kim J-H, Jin H-O, Park J-A, Chang YH, Hong YJ, Lee JK (2014a) Comparison of three different kits for extraction of high-quality RNA from frozen blood. Springerplus 3:2–5
Kim JH, Jin HO, Park JA, Chang YH, Hong YJ, Lee JK (2014b) Comparison of three different kits for extraction of high-quality RNA from frozen blood. Springerplus 3:76. http://www.springerplus.com/content/3/1/76
Kurar E, Atli MO, Guzeloglu A, Ozsensoy Y, Semacan A (2010) Comparison of five different RNA isolation methods from equine endometrium for gene transcription analysis. Kafkas Univ Vet Fak Derg 16:851–855. https://doi.org/10.9775/kvfd.2010.1829
Landolt L, Marti HP, Beisland C, Flatberg A, Eikrem OS (2016) RNA extraction for RNA sequencing of archival renal tissues. Scand J Clin Lab Invest 76:426–434. https://doi.org/10.1080/00365513.2016.1177660
Mannhalter C, Koizar D, Mitterbauer G (2000) Evaluation of RNA isolation methods and reference genes for RT-PCR analyses of rare target RNA. Clin Chem Lab Med 38:171–177. https://doi.org/10.1515/CCLM.2000.026
O’Connell J (2002) The basics of RT-PCR. Springer, Germany
O'neill M, McPartlin J, Arthure K, Riedel S, McMillan N (2011) Comparison of the TLDA with the nanodrop and the reference Qubit system. J Phys Conf Ser. vol 1. IOP Science, p 012047
Pearson G, Lago-Leston A, Valente M, Serrão E (2006) Simple and rapid RNA extraction from freeze-dried tissue of brown algae and seagrasses. Eur J Phycol 41:97–104. https://doi.org/10.1080/09670260500505011
Phongsisay V, Perera VN, Fry BN (2007) Evaluation of eight RNA isolation methods for transcriptional analysis in Campylobacter jejuni. J Microbiol Methods 68:427–429. https://doi.org/10.1016/j.mimet.2006.09.002
Rio DC, Ares M Jr, Hannon GJ, Nilsen TW (2010) Purification of RNA using TRIzol (TRI reagent). Cold Spring Harb Protoc 210:1–3. https://doi.org/10.1101/pdb.prot5439
Saleh KK, Dalkiliç S, Dalkiliç LK, Hamarashid BR, KIRBAĞ S, (2020) Targeting cancer cells: from historic methods to modern chimeric antigen receptor (CAR) T-Cell strategies. AIMS Allergy Immunol 4:32–49. https://doi.org/10.3934/Allergy.2020004
Schroeder A et al (2006) The RIN: an RNA integrity number for assigning integrity values to RNA measurements. BMC Mol Biol 7:3–3. https://doi.org/10.1186/1471-2199-7-3
Sellin Jeffries MK, Kiss AJ, Smith AW, Oris JT (2014) A comparison of commercially-available automated and manual extraction kits for the isolation of total RNA from small tissue samples. BMC Biotechnol 14:2–13. https://doi.org/10.1186/s12896-014-0094-8
Simms D, Cizdziel PE, Chomczynski P (1993) TRIzol: a new reagent for optimal single-step isolation of RNA. J Focus 15:532–535. http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.327.9742&rep=rep1&type=pdf
Szelenberger R, Kacprzak M, Saluk-Bijak J, Zielinska M, Bijak M (2019) Plasma MicroRNA as a novel diagnostic. Clin Chim Acta 499:98–107. https://doi.org/10.1016/j.cca.2019.09.005
Tan SC, Yiap BC (2009) DNA, RNA, and protein extraction: the past and the present. J Biomed Biotechnol 209:574398. https://doi.org/10.1155/2009/574398
Tavares L, Alves PM, Ferreira RB, Santos CN (2011) Comparison of different methods for DNA-free RNA isolation from SK-N-MC neuroblastoma. BMC Res Notes 4:3. https://doi.org/10.1186/1756-0500-4-3
Tesena P, Korchunjit W, Taylor J, Wongtawan T (2017) Comparison of commercial RNA extraction kits and qPCR master mixes for studying gene expression in small biopsy tissue samples from the equine gastric epithelium. J Equine Sci 28:135–141. https://doi.org/10.1294/jes.28.135
Thanh T, Omar H, Abdullah MP, Chi VT, Noroozi M, Ky H, Napis S (2009) Rapid and effective method of RNA isolation from green microalga Ankistrodesmus convolutus. Mol Biotechnol 43:148–153. https://doi.org/10.1007/s12033-009-9182-8
Thörn I, Olsson-Strömberg U, Ohlsen C, Jonsson AM, Klangby U, Simonsson B, Barbany G (2005) The impact of RNA stabilization on minimal residual disease assessment in chronic myeloid leukemia. Haematologica 90:1471–1476. https://pubmed.ncbi.nlm.nih.gov/16266893/
Funding
None.
Author information
Authors and Affiliations
Contributions
SD and LKD produced design of manipulation for RNA extraction kits. KKS and BRH collected the blood samples and prepared RNA isolation. LKD and BRH synthesized cDNA from extracted RNA. SD, LKD and SM performed real-time PCR for housekeeping genes. Additionally, SK, MK and all authors contributed to writing and controlling the manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interest.
Consent to publish
The Authors hereby consents to publication of the Work in “Iranian Journal of Science and Technology, Transaction A, Science (ISTT)”.
Consent to participate
Our manuscript had permission to perform evaluation of three commercial kits on frozen blood of human in Firat University by research ethics committee at 29/06/2020, N0. 97132852.
Animal rights
No animals were used in our study.
Data availability statement
The data associated with a paper are available, and under what conditions the data can be accessed.
Rights and permissions
About this article
Cite this article
Dalkılıç, S., Dalkılıç, L.K., Saleh, K.K. et al. The Evaluation and Manipulation of Different Kits for Isolation of High-quality RNA from Frozen Blood. Iran J Sci Technol Trans Sci 45, 1571–1578 (2021). https://doi.org/10.1007/s40995-021-01171-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-021-01171-8