Abstract
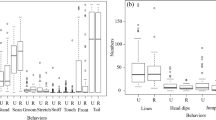
Variation in group spread can affect communication between individuals, synchrony of activity and collective decision-making. Most studies have measured this variation in large time scales, but the variables that influence group spread change as the social unit ranges over different areas throughout the day. Thus, our aim was to analyze the diurnal variation in group spread of a group of black capuchin monkeys (Sapajus nigritus) inhabiting an area within the southeastern Brazilian Atlantic Forest, the Carlos Botelho State Park. We measured group spread as the distance between the coordinates of the location of the two most distant group members every 5 min during entire days. Using time series analysis, we found that the group spread varied within short periods of time (15 min) throughout the day, revolving around the average group spread distance of 36 m. Group members tended to decrease the interindividual distance over time when they were more than 36 m apart, and to increase the distance when they were at a distance of less than 36 m. In addition, group spread was higher when monkeys were eating fruit than during other activities. We conclude that the variation in group spread may reflect competition for discrete food resources and that increasing cohesion several times throughout the day can be a way of resuming social contact between individuals.




Similar content being viewed by others
References
Ab’Sáber A (2003) Os domínios de natureza no Brasil: potencialidades paisagísticas, vol 1. Ateliê Editorial
Aguiar O (2003) Comparação entre os métodos de quadrantes e parcelas na caracterização da composição floristica e fitossociológica de um trecho de floresta ombrófila densa no Parque Estadual “Carlos Botelho.” São Miguel Arcanjo, São Paulo
Altmann J (1974) Observational study of behavior: sampling methods. Behaviour 49:227–266. https://doi.org/10.1163/156853974X00534
Archie E, Moss C, Alberts S (2006) The ties that bind: genetic relatedness predicts the fission and fusion of social groups in wild African elephants. Proc Royal Soc b: Biol Sci 273:513–522. https://doi.org/10.1098/rspb.2005.3361
Asensio N, Korstjens A, Aureli F (2009) Fissioning minimizes ranging costs in spider monkeys: a multiple-level approach. Behav Ecol Sociobiol 63:649–659. https://doi.org/10.1007/s00265-008-0699-9
Aureli F, Schaffner C, Boesch C et al (2008) Fission–fusion dynamics: new research frameworks. Currenty Anthropology 49(4):627–654. https://doi.org/10.1086/586708
Aureli F, Schaffner C, Asensio N, Lusseau D (2012) What is a subgroup? How socioecological factors influence interindividual distance. Behav Ecol. https://doi.org/10.1093/beheco/ars122
Broom M, Koenig A, Borries C (2009) Variation in dominance hierarchies among group-living animals: modeling stability and the likelihood of coalitions. Behav Ecol. https://doi.org/10.1093/beheco/arp069
Chancellor R, Isbell L (2009) Female grooming markets in a population of gray-cheeked mangabeys (Lophocebus albigena). Behav Ecol 20(1):79–86. https://doi.org/10.1093/beheco/arn117
Chapman C, Rothman J (2009) Within-species differences in primate social structure: evolution of plasticity and phylogenetic constraints. Primates 50(1):12–22. https://doi.org/10.1007/s10329-008-0123-0
Chapman C, Chapman L, Wrangham R (1995) Ecological constraints on group size: an analysis of spider monkey and chimpanzee subgroups. Behav Ecol End Sociobiol 36(1):59–70. https://doi.org/10.1007/s002650050125
Chapman C, Gillespie T, Goldberg T (2005) Primates and the ecology of their infectious diseases: how will anthropogenic change affect host-parasite interactions? Evolut Anthropol: Issues, News, Rev 14(4):134–144. https://doi.org/10.1002/evan.20068
Connor R, Wells R, Mann J, Read A (2000) The bottlenose dolphin: social relationships in fission-fusion society. In: Mann J, Connor R, Tyack P, Whitehead H (eds) Cetaceansocieties: field studies of whales and dolphins. University of Chicago Press, Chigado IL, pp 91–126
Conradt L, Roper T (2005) Consensus decision making in animals. Trends Ecol Evol 20(8):449–456. https://doi.org/10.1016/j.tree.2005.05.008
Couzin I, Laidre M (2009) Fission–fusion populations. Curr Biol 19(15):R633–R635. https://doi.org/10.1016/j.cub.2009.05.034
Crofoot M, Gilby I, Wikelski M, Kays R (2008) Interaction location outweighs the competitive advantage of numerical superiority in Cebus capucinus intergroup contests. Proc Natl Acad Sci 105(2):577–581. https://doi.org/10.1073/pnas.0707749105
Cropp S, Sughrue K, Selvaggi L, Quatrone R, Boinski S, Henry M (2002) An expanded test of the ecological model of primate social evolution: competitive regimes and female bonding in three species of squirrel monkeys (Saimiri oerstedii, S. boliviensis and S. sciureus). Behaviour 139(2):227–261. https://doi.org/10.1163/156853902760102663
Di Bitetti M (2001) Home-range use by the tufted capuchin monkey (Cebus apella nigritus) in a subtropical rainforest of Argentina. J Zool 253(01):33–45. https://doi.org/10.1017/s0952836901000048
Di Bitetti M (2003) Food-associated calls of tufted capuchin monkeys (Cebus apella nigritus) are functionally referential signals. Behaviour 140(5):565–592. https://doi.org/10.1163/156853903322149441
Di Bitetti M (2005) Food-associated calls and audience effects in tufted capuchin monkeys Cebus Apella Nigritus. Anim Behav 69(4):911–919. https://doi.org/10.1016/j.anbehav.2004.05.021
Di Bitetti M, Janson C (2001) Social foraging and the finder’s share in capuchin monkeys Cebus Apella. Animal Beh 62(1):47–56. https://doi.org/10.1006/anbe.2000.1730
Dias A, do Couto H (2005) Comparação de métodos de amostragem na Floresta Ombrófila Densa-Parque Estadual Carlos Botelho/SP-Brasil. Comparison of sampling methods in the dense Ombrophylous forest. Revista do Instituto Florestal, Carlos Botelho State Park, São Paulo, Brazil
Dias A, Custodio Filho A, Franco G, Couto H (1995) Estrutura do componente arbóreo em um trecho de floresta pluvial atlântica secundária—Parque Estadual Carlos Botelho. Revista Do Instituto Florestal 7(2):125–155
Domingues E, Silva D (1988) Geomorfologia do Parque Estadual de Carlos Botelho (SP). Boletim Técnico Do Inst Florest 42:71–105
Durlak J (2009) How to select, calculate, and interpret effect sizes. J Pediatr Psychol 34(9):917–928. https://doi.org/10.1093/jpepsy/jsp004
Eckhardt N, Polansky L, Boesch C (2015) Spatial cohesion of adult male chimpanzees (Pan troglodytes verus) in Taï National Park, Côte D’ivoire. Am J Primat 77(2):125–134. https://doi.org/10.1002/ajp.22316
Ehlers R (2007) Análise de séries temporais. Laboratório de Estatística e Geoinformação. Universidade Federal do Paraná
Fan P, Jiang X (2010) Maintenance of multifemale social organization in a group of Nomascus concolor at Wuliang Mountain, Yunnan, China. Int J Primatol 31(1):1–13. https://doi.org/10.1007/s10764-009-9375-9
Fogaça D, Izar P (2017) Percepção de predação por macaco-prego: evidência de uso de palmeira para proteção. Soc Bras De Mastozool 79:47–49
Florestal I (2008) Parque Estadual Carlos Botelho: plano de manejo. Instituto Florestal, São Paulo, Brazil
Hill RA, Lee PC (1998) Predation risk as an influence on group size in cercopithecoid primates: implications for social structure. J Zool 245(4):447–456. https://doi.org/10.1111/j.1469-7998.1998.tb00119.x
Hirsch B (2002) Social monitoring and vigilance behavior in brown capuchin monkeys (Cebus apella). Behav Ecol Sociobiol 52(6):458–464
Holekamp K, Cooper S, Katona C, Berry N, Frank L, Smale L (1997) Patterns of association among female spotted hyenas (Crocuta crocuta). J Mammal 78(1):55–64. https://doi.org/10.1007/s002650100356
Isbell L (1991) Contest and scramble competition: patterns of female aggression and ranging behavior among primates. Behav Ecol 2(2):143–155. https://doi.org/10.1093/beheco/2.2.143
Itoh N, Nishida T (2007) Chimpanzee grouping patterns and food availability in Mahale Mountains National Park, Tanzania. Primates 48(2):87–96. https://doi.org/10.1007/s10329-006-0031-0
Izar P (2004) Female social relationships of Cebus apella nigritus in a southeastern Atlantic forest: an analysis through ecological models of primate social evolution. Behaviour 141(1):71–99. https://doi.org/10.1163/156853904772746619
Izar P, Verderane M, Peternelli-dos-Santos L et al (2012) Flexible and conservative features of social systems in tufted capuchin monkeys: comparing the socioecology of Sapajus libidinosus and Sapajus nigritus. Am J Primatol 74(4):315–331. https://doi.org/10.1002/ajp.20968
Jacobs A (2010) Group cohesiveness during collective movements: travelling apart together: collective movements: decision-making process within groups. Behav Proc 84(3):678–680. https://doi.org/10.1016/j.beproc.2010.03.004
Janson C (1988) Food competition in brown capuchin monkeys (Cebus apella): quantitative effects of group size and tree productivity. Behaviour 105(1):53–76. https://doi.org/10.1163/156853988x00449
Janson C (1990) Social correlates of individual spatial choice in foraging groups of brown capuchin monkeys, Cebus apella. Anim Behav 40(5):910–921. https://doi.org/10.1016/S0003-3472(05)80993-5
Janson C, Di Bitetti M (1997) Experimental analysis of food detection in capuchin monkeys: effects of distance, travel speed, and resource size. Behav Ecol Sociobiol 41(1):17–24. https://doi.org/10.1007/s002650050359
Jordano P, Galetti M, Pizo M, Silva W (2006) Ligando frugivoria e dispersão de sementes à biologia da conservação. In: Rocha C, Bergallo H, Van Sluys M, Alves M (eds) Biologia da Conservação: Essências. Rima Editora, São Carlos, SP, pp 411–436
Kappeler P, van Schaik C (2002) Evolution of primate social systems. Int J Primatol 23(4):707–740. https://doi.org/10.1007/978-1-4684-6694-2_13
Kazahari N (2014) Maintaining social cohesion is a more important determinant of patch residence time than maximizing food intake rate in a group-living primate, Japanese macaque (Macaca fuscata). Primates 55(2):179–184. https://doi.org/10.1007/s10329-014-0410-x
Kerth G, Konig B (1999) Fission, fusion and nonrandom association in female Bechstein’s bats (Myotis bechsteinii). Behaviour 136(9):1187–1202. https://doi.org/10.1163/156853999501711
Kerth G, Perony N, Schweitzer F (2011) Bats are able to maintain long-term social relationships despite the high fission–fusion dynamics of their groups. Proc Royal Soc London b: Biol Sci 278(1719):2761–2767. https://doi.org/10.1098/rspb.2010.2718
Koenig A, Borries C (2006) The predictive power of socioecological models: a reconsideration of resource characteristics, agonism, and dominance hierarchies. In: Hohmann G, Robbins M, Boesch C (eds) Feeding ecology in apes and other primates. Cambridge University Press, Cambridge, pp 263–284
Lappan S (2007) Social relationships among males in multimale siamang groups. Int J Primatol 28(2):369–387. https://doi.org/10.1007/s10764-007-9122-z
Leca J, Gunst N, Thierry B, Petit O (2003) Distributed leadership in semifree-ranging white-faced capuchin monkeys. Anim Behav 66(6):1045–1052. https://doi.org/10.1006/anbe.2003.2276
Lehmann J, Korstjens A, Dunbar R (2007a) Fission–fusion social systems as a strategy for coping with ecological constraints: a primate case. Evol Ecol 21(5):613–634. https://doi.org/10.1007/s10682-006-9141-9
Lehmann J, Korstjens A, Dunbar R (2007b) Group size, grooming and social cohesion in primates. Anim Behav 74(6):1617–1629. https://doi.org/10.1016/j.anbehav.2006.10.025
Lynch J, Rímoli J (2000) Demography and social structure of group of Cebus apella nigritus (Goldfuss, 1809, Primates/Cebidae) at Estação Biológica de Caratinga Minas Gerais. Neotrop Primates 8(2):44–49
Lynch-Alfaro J (2007) Subgrouping patterns in a group of wild Cebus apella nigritus. Int J Primatol 28(2):271–289. https://doi.org/10.1007/s10764-007-9121-0
Marlowe F (2005) Hunter-gatherers and human evolution. Evolut Anthropol: Issues, News, Rev 14(2):54–67. https://doi.org/10.1002/evan.20046
Meunier H, Leca J, Deneubourg J, Petit O (2006) Group movement decisions in capuchin monkeys: the utility of an experimental study and a mathematical model to explore the relationship between individual and collective behaviours. Behaviour 143(12):1511–1527. https://doi.org/10.1163/156853906779366982
Miramontes O, Boyer D, Ramos-Fernandez G (2016) Understanding fission-fusion dynamics in social animals through agent-based modelling. In: Proceedings of the Artificial Life Conference, Cambridge pp 338–339. https://doi.org/10.7551/978-0-262-33936-0-ch056
Nakai É (2007) Fissão-fusão em Cebus nigritus: flexibilidade social como estratégia de ocupação de ambientes limitantes. Dissertation, Universidade de São Paulo. https://doi.org/10.11606/d.47.2007.tde-17012008-110418
Negreiros O, Custódio Filho A, Dias A, Franco G et al (1995) Análise estrutural de um trecho de floresta pluvial tropical, Parque Estadual de Carlos Botelho, Núcleo Sete Barras (SP-Brasil), vol 7. Revista do Instituto Florestal, São Paulo
Noë R, Laporte M (2014) Socio-spatial cognition in vervet monkeys. Anim Cogn 17(3):597–607. https://doi.org/10.1007/s10071-013-0690-3
Parr N, Melin A, Fedigan L (2011) Figs are more than fallback foods: the relationship between Ficus and Cebus in a tropical dry forest. Int J Zool 2011:1–10. https://doi.org/10.1155/2011/967274
Petit O, Bon R (2010) Decision-making processes: the case of collective movements. Behav Proc 84(3):635–647. https://doi.org/10.1016/j.beproc.2010.04.009
Presotto A, Izar P (2010) Spatial reference of black capuchin monkeys in Brazilian Atlantic Forest: egocentric or allocentric? Anim Behav 80(1):125–132. https://doi.org/10.1016/j.anbehav.2010.04.009
Ramos-Fernández G, Morales J (2014) Unraveling fission-fusion dynamics: how subgroup properties and dyadic interactions influence individual decisions. Behav Ecol Sociobiol 68(8):1225–1235. https://doi.org/10.1007/s00265-014-1733-8
Ramos-Fernández G, Pinacho-Guendulain B, Miranda-Pérez A, Boyer D (2011) No evidence of coordination between different subgroups in the fission–fusion society of spider monkeys (Ateles geoffroyi). Int J Primatol 32(6):1367–1382. https://doi.org/10.1007/s10764-011-9544-5
Rose L (1998) Behavioral ecology of white-faced capuchins (Cebus capucinus) in Costa Rica. Dissertation, Washington University
Rubenstein D (1978) On predation, competition, and the advantages of group living. In: Bateson P, Klopfer P (eds) Social behavior. Springer, US, pp 205–231
Sakura O (1994) Factors affecting party size and composition of chimpanzees (Pan troglodytes verus) Bossou, Guinea. Int J Primat 15(2):167–183. https://doi.org/10.1007/bf02735272
Santos L (2015) Parâmetros nutricionais da dieta de duas populações de macacos-prego: Sapajus libidinosus no ecótono cerrado/caatinga e Sapajus nigritus na Mata Atlântica. Dissertation, Universidade de São Paulo, São Paulo. https://doi.org/10.11606/t.47.2015.tde-10082015-110633
Schino G (2001) Grooming, competition and social rank among female primates: a meta-analysis. Anim Behav 62(2):265–271. https://doi.org/10.1006/anbe.2001.1750
Schreier A, Swedell L (2012) Ecology and sociality in a multilevel society: ecological determinants of spatial cohesion in hamadryas baboons. Am J Phys Anthropol 148(4):580–588. https://doi.org/10.1002/ajpa.22076
Shumway R, Stoffer S, Stoffer S (2000) Time series analysis and its applications, vol 3. Springer, New York
Smith J, Kolowski J, Graham K, Dawes S, Holekamp K (2008) Social and ecological determinants of fission–fusion dynamics in the spotted hyaena. Anim Behav 76(3):619–636. https://doi.org/10.1016/j.anbehav.2008.05.001
Snyder-Mackler N, Beehner J, Bergman T (2012) Defining higher levels in the multilevel societies of geladas (Theropithecus gelada). Int J Primatol 33(5):1054–1068. https://doi.org/10.1007/s10764-012-9584-5
Sorato E, Gullett R, Griffith C, Russell AF (2012) Effects of predation risk on foraging behaviour and group size: adaptations in a social cooperative species. Anim Behav 84(4):823–834. https://doi.org/10.1016/j.anbehav.2012.07.003
Stanford C (2002) Avoiding predators: expectations and evidence in primate antipredator behavior. Int J Primatol 23(4):741–757
Stevenson P, Quiñones M, Ahumada J (1998) Effects of fruit patch availability on feeding subgroup size and spacing patterns in four primate species at Tinigua National Park, Colombia. Int J Primatol 19(2):313–324
Sugiura H, Shimooka Y, Tsuji Y (2011) Variation in spatial cohesiveness in a group of Japanese macaques (Macaca fuscata). Int J Primatol 32(6):1348–1366. https://doi.org/10.1007/s10764-011-9533-8
Sumpter D, Pratt S (2009) Quorum responses and consensus decision making. Philos Transac Royal Soc B: Biol Sci 364(1518):743–753. https://doi.org/10.1098/rstb.2008.0204
Tabachnick G, Fidell S, Ullman B (2007) Using multivariate statistics 5: 481–498. Pearson, Boston, MA
Takahata SS, Okayasu N, Hill D (1994) Troop extinction and fusion in wild Japanese macaques of Yakushima Island, Japan. Am J Primatol 33(4):317–322. https://doi.org/10.1002/ajp.1350330406
Teichroeb J, Sicotte P (2012) Cost-free vigilance during feeding in folivorous primates? Examining the effect of predation risk, scramble competition, and infanticide threat on vigilance in ursine colobus monkeys (Colobus vellerosus). Behav Ecol Sociobiol 66(3):453–466. https://doi.org/10.1007/s00265-011-1292-1
Tokuda M, Martins M, Izar P (2018) Socio-genetic correlates of unbiased sex dispersal in a population of black capuchin monkeys (Sapajus nigritus). Acta Ethol 21(1):1–11. https://doi.org/10.1007/s10211-017-0277-0
van Schaik C (1999) The socioecology of fission–fusion sociality in orangutans. Primates 40(1):69–86. https://doi.org/10.1007/bf02557703
Verdolin J (2006) Meta-analysis of foraging and predation risk trade-offs in terrestrial systems. Behav Ecol Sociobiol 60(4):457–464. https://doi.org/10.1007/s00265-006-0172-6
Vogel E (2005) Rank differences in energy intake rates in white-faced capuchin monkeys, Cebus capucinus: the effects of contest competition. Behav Ecol Sociobiol 58(4):333–344. https://doi.org/10.1007/s00265-005-0960-4
Vogel E, Munch S, Janson C (2007) Understanding escalated aggression over food resources in white-faced capuchin monkeys. Anim Behav 74(1):71–80. https://doi.org/10.1016/j.anbehav.2007.02.003
Wada K, Ogawa H (2009) Identifying inter-individual social distances in Japanese monkeys. Mammalia 73(2):81–84. https://doi.org/10.1515/mamm.2009.023
Watve M, Jog M (1997) Epidemic diseases and host clustering: an optimum cluster size ensures maximum survival. J Theor Biol 184(2):165–169. https://doi.org/10.1006/jtbi.1996.0267
Watve M, Sukumar R (1995) Parasite abundance and diversity in mammals: correlates with host ecology. PNAS 92(19):8945–8949. https://doi.org/10.1073/pnas.92.19.8945
Wiszniewski J, Allen S, Möller L (2009) Social cohesion in a hierarchically structured embayment population of Indo-Pacific bottlenose dolphins. Anim Behav 77(6):1449–1457. https://doi.org/10.1016/j.anbehav.2009.02.025
Wittemyer G, Douglas-Halmilton I, Getz M (2005) The socioecology of elephants: analysis of the processes creating multitiered social structures. Anim Behav 69(6):1357–1371. https://doi.org/10.1016/j.anbehav.2004.08.018
Acknowledgements
We thank our field assistant, Edipo Fernandes, as well as José Siqueira for help with statistical analyses. We thank the Instituto Florestal de São Paulo, especially the director of the PECB, José Maia, who granted us permission to conduct this research.
Funding
The research was funded by Fundação de Amparo a Pesquisa do Estado de São Paulo grant to P.I. (14/13237-1) and a Conselho Nacional de Desenvolvimento Científico e Tecnológico fellowship to V.R.L. (130156/2014-0).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by VL. The first draft of the manuscript was written by VL, and all authors commented on previous versions of the manuscript. Writing—review and editing: PI. Funding acquisition: VL. Resources: PI. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest and no relevant financial or non-financial interests to disclose.
Ethical approval
This research was approved by the Committee on Ethical Research on Animals (CEPA) of the Institute of Psychology, Universidade de São Paulo, certification number 009.2014.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Luccas, V., Izar, P. Black capuchin monkeys dynamically adjust group spread throughout the day. Primates 62, 789–799 (2021). https://doi.org/10.1007/s10329-021-00926-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10329-021-00926-6