Abstract
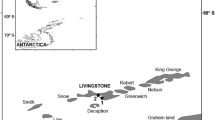
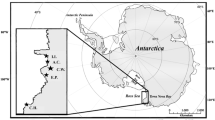
For top consumers in marine environments, trophic discrimination factors (∆13C and ∆15N) between food and consumers’ tissues are expected to be similar among related species. However, few studies conducted in the laboratory indicate a large variability among species, which should be potentially higher in free-ranging animals. Here, we test for differences in tissue-specific ∆13C and ∆15N values of two wild penguin species (Chinstrap Pygoscelis antarctica and Gentoo P. papua) breeding in sympatry at Livingston Island, Antarctica. A total of 41 adults and 28 chicks, and food items comprised exclusively by Antarctic krill (Euphausia superba, n = 22) in Chinstraps and almost exclusively in Gentoos, were sampled for stable isotope analyses. Overall, Δ13C values varied between -1.8 and 4.0 ‰ and Δ15N values ranged from 1.2 to 6.1 ‰, and these differed between species, tissues and age-classes. Δ13C in adult penguins differed between species for feather and blood. Species-specific differences in Δ13C and Δ15N were seen in chick nail and muscle, while only Δ13C values differed between species in feathers. Our results show that trophic discrimination factors can differ substantially between closely related species consuming similar prey, especially in Δ13C value. Variation in Δ13C was driven by species, tissue and age-class, while variation in Δ15N was mostly driven by tissue type. Trophic discrimination factors may be associated to physiological and/or stress factors which may fluctuate in the wild, and this was particularly evident on chicks. This study highlights the use of diet-specialised species for the determination of trophic discrimination factors in the wild.


Similar content being viewed by others
References
Aguilar A, Giménez J, Gómez-Campos E, Cardona L, Borrell A (2014) δ15N Value does not reflect fasting in mysticetes. PLoS ONE 9:e92288. https://doi.org/10.1371/journal.pone.0092288
Auerswald K, Wittmer MHOM, Zazzo A, Scha R, Schnyder H (2010) Biases in the analysis of stable isotope discrimination in food webs. J Appl Ecol 47:936–941. https://doi.org/10.1111/j.1365-2664.2009.01764.x
Barnes C, Sweeting CJ, Jennings S, Barry JT, Polunin NVC (2007) Effect of temperature and ration size on carbon and nitrogen stable isotope trophic fractionation. Funct Ecol 21:356–362. https://doi.org/10.1111/j.1365-2435.2006.01224.x
Barnes C, Jennings S, Polunin NVC, Lancaster JE (2008) The importance of quantifying inherent variability when interpreting stable isotope field data. Oecologia 155:227–235. https://doi.org/10.1007/s00442-007-0904-y
Barquete V, Strauss V, Ryan PG (2013) Stable isotope turnover in blood and claws: A case study in captive African Penguins. J Exp Mar Bio Ecol 448:121–127. https://doi.org/10.1016/j.jembe.2013.06.021
Barton MB, Litvin SY, Vollenweider JJ, Heintz RA, Norcross BL, Boswell KM (2019) Implications of trophic discrimination factor selection for stable isotope food web models of low trophic levels in the Arctic nearshore. Mar Ecol Prog Ser 613:211–216
Boecklen WJ, Yarnes CT, Cook BA, James AC (2011) On the use of stable isotopes in trophic ecology. Annu Rev Ecol Evol Syst 42:411–440. https://doi.org/10.1146/annurev-ecolsys-102209-144726
Bond A, Diamond A (2011) Recent Bayesian stable-isotope mixing models are highly sensitive to variation in discrimination factors. Ecol Appl 21:1017–1023
Bond AL, Jones IL (2009) A practical introduction to stable-isotope analysis for seabird biologists: approaches, cautions and caveats. Mar Ornithol 37:183–188
Borrell A, Abad-Oliva N, Gómez-Campos E, Giménez J, Aguilar A (2012) Discrimination of stable isotopes in fin whale tissues and application to diet assessment in cetaceans. Rapid Commun Mass Spectrom 26:1596–1602. https://doi.org/10.1002/rcm.6267
Carravieri A, Bustamante P, Churlaud C, Fromant A, Cherel Y (2014) Moulting patterns drive within-individual variations of stable isotopes and mercury in seabird body feathers: implications for monitoring of the marine environment. Mar Biol 161:963–968. https://doi.org/10.1007/s00227-014-2394-x
Caut S, Angulo E, Courchamp F (2009) Variation in discrimination factors (Δ15N and Δ13C): the effect of diet isotopic values and applications for diet reconstruction. J Appl Ecol 46:443–453. https://doi.org/10.1111/j.1365-2664.2009.01620.x
Caut S, Laran S, Garcia-Hartmann E, Das K (2011) Stable isotopes of captive cetaceans (killer whales and bottlenose dolphins). J Exp Biol 214:538–545. https://doi.org/10.1242/jeb.045104
Cherel Y (2008) Isotopic niches of emperor and Adélie penguins in Adélie Land, Antarctica. Mar Biol 154:813–821. https://doi.org/10.1007/s00227-008-0974-3
Cherel Y, Hobson K (2007) Geographical variation in carbon stable isotope signatures of marine predators: a tool to investigate their foraging areas in the Southern Ocean. Mar Ecol Prog Ser 329:281–287. https://doi.org/10.3354/meps329281
Cherel Y, Hobson KA, Weimerskirch H (2000) Using stable-isotope analysis of feathers to distinguish moulting and breeding origins of seabirds. Oecologia 122:155–162. https://doi.org/10.1007/PL00008843
Cherel Y, Hobson KA, Hassani S (2005a) Isotopic discrimination between food and blood and feathers of captive penguins: implications for dietary studies in the wild. Physiol Biochem Zool 78:106–115. https://doi.org/10.1086/425202
Cherel Y, Hobson KA, Bailleul F, Groscolas R (2005b) Nutrition, physiology, and stable isotopes: New information from fasting and molting penguins. Ecology 86:2881–2888. https://doi.org/10.1890/05-0562
Cherel Y, Jaquemet S, Maglio A, Jaeger A (2014) Differences in δ13C and δ15N values between feathers and blood of seabird chicks: implications for non-invasive isotopic investigations. Mar Biol 161:229–237. https://doi.org/10.1007/s00227-013-2314-5
Ciancio JE, Righi C, Faiella A, Frere E (2016) Blood-specific isotopic discrimination factors in the Magellanic penguin (Spheniscus magellanicus). Rapid Commun Mass Spectrom 30:1865–1869. https://doi.org/10.1002/rcm.7661
Costantini D (2008) Oxidative stress in ecology and evolution: lessons from avian studies. Ecol Lett 11:1238–1251. https://doi.org/10.1111/j.1461-0248.2008.01246.x
Dimitrijević D, Paiva VH, Ramos JA, Seco J, Ceia FR, Chipev N, Valente T, Barbosa A, Xavier JC (2018) Isotopic niches of sympatric Gentoo and Chinstrap Penguins: evidence of competition for Antarctic krill? Polar Biol 41:1655–1669. https://doi.org/10.1007/s00300-018-2306-5
Giménez J, Ramírez F, Almunia J, G. Forero M, de Stephanis R, (2016) From the pool to the sea: Applicable isotope turnover rates and diet to skin discrimination factors for bottlenose dolphins (Tursiops truncatus). J Exp Mar Bio Ecol 475:54–61. https://doi.org/10.1016/j.jembe.2015.11.001
Healy K, Guillerme T, Kelly SBA, Inger R, Bearhop S, Jackson AL (2018) SIDER: an R package for predicting trophic discrimination factors of consumers based on their ecology and phylogenetic relatedness. Ecography (cop) 41:1393–1400. https://doi.org/10.1111/ecog.03371
Hinke JT, Polito MJ, Goebel ME, Jarvis S, Reiss CS, Thorrold SR, Trivelpiece WZ, Watters GM (2015) Spatial and isotopic niche partitioning during winter in chinstrap and Adélie penguins from the South Shetland Islands. Ecosphere 6:1–32
Hobson KA, Clark RG (1992) Assessing avian diets using stable isotopes I: turnover of 13C in tissues. Condor 94:181–188
Hoeinghaus DJ, Zeug SC (2008) Can stable isotope ratios provide for community-wide measures of trophic structure? Comment. Ecology 89:2353–2357
Hussey NE, Macneil MA, Mcmeans BC, Olin JA, Dudley SFJ, Cliff G, Wintner SP, Fennessy ST, Fisk AT (2014) Rescaling the trophic structure of marine food webs. Ecol Lett 17:239–250. https://doi.org/10.1111/ele.12226
Lesage V, Hammill MO, Kovacs KM (2002) Diet-tissue fractionation of stable carbon and nitrogen isotopes in phocid seals. Mar Mammal Sci 18:182–193. https://doi.org/10.1111/j.1748-7692.2002.tb01027.x
McCutchan JH, Lewis WM, Kendall C, Mcgrath CC (2003) Variation in trophic shift for stable isotope ratios of carbon, nitrogen, and sulfur. Oikos 102:378–390
McKenzie KFR (2011) The foraging ecology of little penguin (Eudyptula minor) on Tiritiri Matangi Island. MSc Thesis, Massey University, Auckland
McMahon KW, Hamady LL, Thorrold SR (2013) A review of ecogeochemistry approaches to estimating movements of marine animals. Limnol Oceanogr 58:697–714. https://doi.org/10.4319/lo.2013.58.2.0697
Miller AK, Kappes MA, Trivelpiece SG, Trivelpiece WZ (2010) Foraging-niche separation of breeding Gentoo and Chinstrap Penguins, South Shetland Islands, Antarctica. Condor 112:683–695. https://doi.org/10.1525/cond.2010.090221
Mizutani H, Fukuda M, Kabaya Y (1992) 13C and 15N enrichment factors of feathers of 11 species of adult birds. Ecology 73:1391–1395
Newsome SD, Rio CM, Bearhop S, Phillips DL (2007) A niche for isotopic ecology. Front Ecol Environ 5:429–436. https://doi.org/10.1890/060150.01
Panasiuk A, Wawrzynek-Borejko J, Musiał A, Korczak-Abshire M (2020) Pygoscelis penguin diets on King George Island, South Shetland Islands, with a special focus on the krill Euphausia superba. Antarct Sci 32:21–28. https://doi.org/10.1017/S0954102019000543
Parnell AC, Inger R, Bearhop S, Jackson AL (2010) Source partitioning using stable isotopes: coping with too much variation. PLoS ONE 5:e9672. https://doi.org/10.1371/journal.pone.0009672
Peterson BJ, Fry B (1987) Stable isotopes in ecosystem studies. Annu Rev Ecol Syst 18:293–320. https://doi.org/10.1146/annurev.es.18.110187.001453
Petta JC, Shipley ON, Wintner SP, Cliff G, Dicken ML, Hussey NE (2020) Are you really what you eat? Stomach content analysis and stable isotope ratios do not uniformly estimate dietary niche characteristics in three marine predators. Oecologia 192:1111–1126. https://doi.org/10.1007/s00442-020-04628-6
Phillips DL, Inger R, Bearhop S, Jackson AL, Moore JW, Parnell AC, Semmens BX, Ward EJ (2014) Best practices for use of stable isotope mixing models in food-web studies. Can J Zool 92:823–835. https://doi.org/10.1139/cjz-2014-0127
Polito MJ, Fisher S, Tobias CR, Emslie SD (2009) Tissue-specific isotopic discrimination factors in gentoo penguin (Pygoscelis papua) egg components: Implications for dietary reconstruction using stable isotopes. J Exp Mar Bio Ecol 372:106–112. https://doi.org/10.1016/j.jembe.2009.02.014
Polito MJ, Abel S, Tobias CR, Emslie SD (2011a) Dietary isotopic discrimination in gentoo penguin (Pygoscelis papua) feathers. Polar Biol 34:1057–1063. https://doi.org/10.1007/s00300-011-0966-5
Polito MJ, Trivelpiece WZ, Karnovsky NJ, Ng E, Patterson WP, Emslie SD (2011b) Integrating stomach content and stable isotope analyses to quantify the diets of Pygoscelid penguins. PLoS ONE 6:e26642. https://doi.org/10.1371/journal.pone.0026642
Polito MJ, Lynch H, Naveen R, Emslie S (2011c) Stable isotopes reveal regional heterogeneity in the pre-breeding distribution and diets of sympatrically breeding Pygoscelis spp. penguins. Mar Ecol Prog Ser 421:265–277. https://doi.org/10.3354/meps08863
Polito MJ, Reiss CS, Trivelpiece WZ, Patterson WP, Emslie SD (2013) Stable isotopes identify an ontogenetic niche expansion in Antarctic krill (Euphausia superba) from the South Shetland Islands, Antarctica. Mar Biol 160:1311–1323. https://doi.org/10.1007/s00227-013-2182-z
Polito MJ, Trivelpiece W, Patterson W, Karnovsky N, Reiss C, Emslie S (2015) Contrasting specialist and generalist patterns facilitate foraging niche partitioning in sympatric populations of Pygoscelis penguins. Mar Ecol Prog Ser 519:221–237. https://doi.org/10.3354/meps11095
Post DM, Layman CA, Arrington DA, Takimoto G, Quattrochi J, Montaña CG (2007) Getting to the fat of the matter: models, methods and assumptions for dealing with lipids in stable isotope analyses. Oecologia 152:179–189. https://doi.org/10.1007/s00442-006-0630-x
Reid K (1996) A guide to the use of otoliths in the study of predators at South Georgia. British Antarctic Survey, Cambridge
Reiss C, Cossio A, Santora J, Dietrich K, Murray A, Mitchell B, Walsh J, Weiss E, Gimpel C, Jones C, Watters G (2017) Overwinter habitat selection by Antarctic krill under varying sea-ice conditions: implications for top predators and fishery management. Mar Ecol Prog Ser 568:1–16. https://doi.org/10.3354/meps12099
Robbins CT, Felicetti LA, Sponheimer M (2005) The effect of dietary protein quality on nitrogen isotope discrimination in mammals and birds. Oecologia 144:534–540. https://doi.org/10.1007/s00442-005-0021-8
Trivelpiece WZ, Trivelpiece SG, Volkman NJ (1987) Ecological segregation of Adelie, gentoo and chinstrap penguins at King George Island, Antarctica. Ecology 68:351–361. https://doi.org/10.2307/1939266
Vander Zanden HB, Bjorndal KA, Mustin W, Ponciano JM, Bolten AB (2012) Inherent variation in stable isotope values and discrimination factors in two life stages of green turtles. Physiol Biochem Zool 85:431–441. https://doi.org/10.1086/666902
Vander Zanden MJ, Rasmussen JB (2001) Variation in δ15N and δ13C trophic fractionation: implications for aquatic food web studies. Limnol Oceanogr 46:2061–2066. https://doi.org/10.4319/lo.2001.46.8.2061
Vander Zanden MJ, Clayton MK, Moody EK, Solomon CT, Weidel BC (2015) Stable isotope turnover and half-life in animal tissues: a literature synthesis. PLoS ONE 10:e0116182. https://doi.org/10.1371/journal.pone.0116182
Vanderklift MA, Ponsard S (2003) Sources of variation in consumer-diet δ15N enrichment: a meta-analysis. Oecologia 136:169–182. https://doi.org/10.1007/s00442-003-1270-z
Vasil CA, Polito MJ, Patterson WP, Emslie SD (2012) Wanted: dead or alive? Isotopic analysis (δ13C and δ15N) of Pygoscelis penguin chick tissues supports opportunistic sampling. Rapid Commun Mass Spectrom 26:487–493. https://doi.org/10.1002/rcm.5340
Wolf N, Carleton SA, Del RCM (2009) Ten years of experimental animal isotopic ecology. Funct Ecol 23:17–26. https://doi.org/10.1111/j.1365-2435.2008.01529.x
Xavier JC, Hill SL, Belchier M, Bracegirdle TJ, Murphy EJ, Dias JL (2015) From ice to penguins: the role of mathematics in Antarctic research. In: Bourguignon JP, Jeltsch R, Pinto AVM (eds) Mathematics of energy and climate change, CIM series. Springer-Verlag, Switzerland, pp 389–414
Xavier JC, Cherel Y, Boxshall G, Brandt A, Coffer T, Forman J, Havermans C, Jażdżewska AM, Kouwenberg K, Schiaparelli S, Schnabel K, Siegel V, Tarling GA, Thatje S, Ward P, Gutt J (2020) Crustacean guide for predator studies in the Southern Ocean. Scientific Committee on Antarctic Research, Cambridge, UK
Acknowledgements
This work is part of SCAR AnT-ERA, ICED and SCAR-EGBAMM programs. This study benefitted from the strategic programme of MARE, financed by FCT (MARE—UID/MAR/04292/2020). FRC acknowledges the transitory norm contract (DL57/2016/CP1370/CT90) and JS the PhD Grant (SRFH/PD/BD/113487/2015) financed by the Foundation for Science and Technology (FCT; Portugal) and the European Social Fund (POPH, EU). Part of this data has been presented previously at the SCAR Open Science Conference 2020, Hobart, Australia. We would also like to thank Keith M. Hernandez, Arnaud Tarroux and four anonymous reviewers for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
Study design: FRC, YC and JCX. Fieldwork and collection of the samples: JS, AB, NC and JCX. Data analysis and processing: FRC, YC and JCX. FRC led the writing of the manuscript. All authors edited and revised the manuscript, contributed critically to revisions and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to declare.
Research involving animal rights
The fieldwork was conducted under a research permit from the project PENGUIN of the Portuguese Polar Programme PROPOLAR. The sampling methods used for this research were in accordance with recommendations from the Scientific Committee for Antarctic Research (SCAR).
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ceia, F.R., Cherel, Y., Seco, J. et al. Variability in tissue-specific trophic discrimination factors (∆13C and ∆15N) between Antarctic krill Euphausia superba and free-ranging Pygoscelis penguins. Polar Biol 44, 1541–1551 (2021). https://doi.org/10.1007/s00300-021-02889-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-021-02889-2